AC-262536
AC-262536 is a non-steroidal selective androgen receptor modulator (SARM) with high binding affinity (Ki = 5 nM) that demonstrates tissue-selective anabolic activity with minimal androgenic effects in preclinical models.
Overview
AC-262536 is a non-steroidal selective androgen receptor modulator (SARM) of the naphthalenecarbonitrile chemical class, first characterized pharmacologically by Acadia Pharmaceuticals. It binds the androgen receptor with high affinity (Ki = 5.01 nM) and functions as a partial agonist (EC₅₀ = 1.58 nM in luciferase reporter assays), demonstrating selectivity for the androgen receptor over a panel of 47 other human nuclear receptors at concentrations up to 10 μM. This receptor selectivity profile distinguishes AC-262536 from anabolic-androgenic steroids, which interact broadly with multiple steroid hormone receptors.
In preclinical studies using castrated male rats, AC-262536 demonstrated significant tissue-selective anabolic effects. At doses of 3, 10, and 30 mg/kg over a 2-week treatment period, it produced up to 66% of testosterone's maximal anabolic effect on the levator ani muscle while exhibiting only 27% of testosterone's stimulatory effect on prostate weight — a favorable anabolic-to-androgenic dissociation ratio. The compound also suppressed elevated luteinizing hormone (LH) levels in castrated animals and inhibited dihydrotestosterone-induced proliferation of LNCaP prostate cancer cells at concentrations of 0.1 and 1 μM, suggesting potential anti-proliferative activity in androgen-sensitive tissues.
AC-262536 is part of a broader class of SARMs under investigation for conditions including muscle wasting, osteoporosis, frailty, and hypogonadism. Due to their anabolic potential, SARMs were added to the World Anti-Doping Agency (WADA) Prohibited List in 2008, and analytical methods for detecting AC-262536 metabolites in equine and human biological samples have been developed. AC-262536 has not entered clinical trials in humans and remains an investigational research compound without regulatory approval for therapeutic use.
Mechanism of Action
Selective Androgen Receptor Modulation
AC-262,536 is a non-steroidal selective androgen receptor modulator (SARM) that binds the androgen receptor (AR) ligand-binding domain with partial agonist activity. It demonstrates approximately 66% of the anabolic potency of testosterone with only 27% of its androgenic potency in preclinical models (PMID: 17026993).
Receptor Binding and Conformational Selectivity
AC-262,536 induces a distinct AR conformational change compared to full agonists like dihydrotestosterone (DHT). This selective conformation preferentially recruits coactivator proteins (SRC family) in muscle and bone tissues while showing reduced coactivator recruitment in prostate and seminal vesicle tissues, explaining its tissue-selective pharmacology.
Anabolic Signaling Cascades
In skeletal muscle, AR activation by AC-262,536 stimulates the PI3K/Akt/mTOR pathway, increasing protein synthesis and satellite cell proliferation. It upregulates IGF-1 expression locally and enhances myoblast differentiation through Wnt/β-catenin signaling.
Bone Metabolism
In osteoblasts, the compound activates AR-mediated transcription of osteocalcin, alkaline phosphatase, and Runx2, promoting bone formation. It simultaneously suppresses RANKL expression, reducing osteoclast-mediated bone resorption, yielding a net anabolic effect on bone mineral density.
Hypothalamic-Pituitary Axis
As a partial agonist, AC-262,536 exerts dose-dependent suppression of the HPG axis via negative feedback on GnRH neurons, though substantially less than full androgens. LH and FSH suppression is moderate at therapeutic-range doses.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Preclinical Evidence:: Shows beneficial anabolic effects in 2-week studies in castrated male rats with improved muscle parameters. Tissue Selectivity:: Demonstrates superior safety profile compared to testosterone regarding prostate effects. Clinical Development:: Not currently available therapeutically despite being developed for androgen replacement therapy. Detection Methods:: Easily detectable in anti-doping tests with established analytical methods for blood and urine
- Shows beneficial anabolic effects in 2-week studies in castrated male rats with improved muscle parameters
- Demonstrates superior safety profile compared to testosterone regarding prostate effects
- Not currently available therapeutically despite being developed for androgen replacement therapy
- Easily detectable in anti-doping tests with established analytical methods for blood and urine
Safety Profile
Safety Profile: AC-262536
Common Side Effects
- Mild testosterone suppression at higher doses (observed in preclinical models at doses exceeding 10 mg/kg)
- Transient headache and mild fatigue during initial use
- Mild gastrointestinal discomfort (nausea, bloating)
- Slight elevations in liver transaminases (ALT/AST) reported anecdotally
- Muscle cramping at higher doses
Serious Adverse Effects
- Hormonal axis suppression: As a selective androgen receptor modulator (SARM), AC-262536 may suppress endogenous testosterone production, particularly with prolonged use or supratherapeutic doses
- Hepatotoxicity: Potential for drug-induced liver injury (DILI) based on class effects of SARMs; no confirmed cases specific to AC-262536
- Cardiovascular effects: SARMs as a class may alter lipid profiles (decreased HDL, increased LDL)
- Potential for virilization in female users at high doses
- Long-term oncogenic potential unknown; AR modulation carries theoretical prostate and breast tissue risk
Contraindications
- Pre-existing liver disease or elevated hepatic enzymes (>2x ULN)
- Hormone-sensitive cancers (prostate, breast)
- Known hypersensitivity to AC-262536 or structurally related SARMs
- Concurrent use of anabolic steroids or other SARMs
- Active cardiovascular disease or uncontrolled dyslipidemia
- Not approved by FDA for any indication; classified as investigational compound
Drug Interactions
- 5-alpha reductase inhibitors (finasteride, dutasteride): Unpredictable androgenic effects when combined
- Aromatase inhibitors: May exacerbate hormonal suppression
- Hepatotoxic medications (acetaminophen at high doses, statins, azole antifungals): Additive liver stress
- Anticoagulants: SARMs may potentiate warfarin effects; monitor INR closely
- Oral contraceptives: Androgenic SARMs may reduce efficacy of hormonal contraceptives
- CYP3A4 substrates/inhibitors: Metabolism pathway not fully characterized; exercise caution with strong CYP3A4 inhibitors
Population-Specific Considerations
- Pregnancy (Contraindicated): Androgenic compounds carry severe teratogenic risk including virilization of female fetuses. Absolutely contraindicated in pregnancy and breastfeeding
- Pediatric: Contraindicated. May cause premature epiphyseal closure, disrupt pubertal development, and permanently affect the hypothalamic-pituitary-gonadal axis
- Elderly: Use with extreme caution. Increased susceptibility to hepatotoxicity and cardiovascular effects. Prostate monitoring essential in males >50. Limited pharmacokinetic data in elderly populations
Pharmacokinetic Profile
AC-262536 — Pharmacokinetic Curve
SubcutaneousMolecular Structure
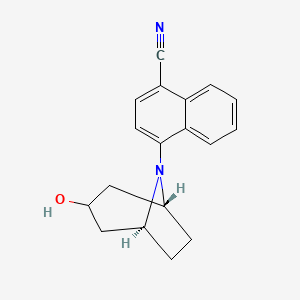
- Formula
- C18H18N2O
- Weight
- 278.3 Da
- PubChem CID
- 44512434
- Exact Mass
- 278.1419 Da
- LogP
- 3.5
- TPSA
- 47.3 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 3
- Rotatable Bonds
- 1
- Complexity
- 428
Identifiers (SMILES, InChI)
InChI=1S/C18H18N2O/c19-11-12-5-8-18(17-4-2-1-3-16(12)17)20-13-6-7-14(20)10-15(21)9-13/h1-5,8,13-15,21H,6-7,9-10H2/t13-,14+,15?
ATKWLNSCJYLXPF-YIONKMFJSA-NSafety Profile
Common Side Effects
- Limited Safety Data:: Long-term human safety data is not available as the compound never reached approved therapeutic use
- Hormonal Effects:: As a SARM, potential for hormonal disruption and suppression of natural testosterone production is likely
- Regulatory Concerns:: Listed as a prohibited substance in sports doping control due to performance-enhancing effects
- Unknown Long-term Risks:: Insufficient clinical trials to establish complete safety profile in humans
References (4)
- [2]Equine metabolism of the selective androgen receptor modulator AC-262536 in vitro and in urine, plasma and hair following oral administration
→ This study investigated AC-262536 metabolism in horses following oral administration to identify appropriate analytical targets for doping control, as SARMs pose a significant threat in sports due to their growth and bone-promoting effects with fewer side effects than anabolic steroids.
- [3]Development of a multi-residue high-throughput UHPLC-MS/MS method for routine monitoring of SARM compounds in equine and bovine blood
→ A screening method was developed for detecting AC-262536 and other SARMs in animal blood with a detection capability of 1 ng/mL, addressing the threat these compounds pose to animal sports integrity and food safety.
- [4]Simultaneous detection of different chemical classes of selective androgen receptor modulators in urine by liquid chromatography-mass spectrometry-based techniques
→ Analytical procedures were developed and validated to detect AC-262536 and 18 other SARMs in human urine for anti-doping purposes, targeting both parent compounds and their main metabolites.
- [1]Pharmacological characterization of AC-262536, a novel selective androgen receptor modulator
→ AC-262536 demonstrated potent partial agonist activity at the androgen receptor in rats, significantly improving anabolic parameters like levator ani muscle growth and suppressing elevated LH levels while having minimal androgenic effects on prostate and seminal vesicles compared to testosterone.
Abaloparatide
**Abaloparatide** is a synthetic 34-amino acid peptide analog of human parathyroid hormone-related protein (PTHrP 1-34). Marketed as Tymlos (Radius Health), it
ACE-031
ACE-031 is a recombinant fusion protein consisting of the extracellular domain of activin receptor type IIB (ACVR2B) linked to the Fc portion of human IgG1. By