Donepezil
Donepezil is an FDA-approved acetylcholinesterase inhibitor prescribed for the symptomatic treatment of mild to severe Alzheimer's disease, marketed under the brand name Aricept.
Overview
Donepezil is a centrally-acting, reversible inhibitor of acetylcholinesterase (AChE), the enzyme responsible for breaking down the neurotransmitter acetylcholine in the synaptic cleft. By inhibiting this enzyme, donepezil increases the availability of acetylcholine in the brain, partially compensating for the cholinergic neuron loss that characterizes Alzheimer's disease. First approved by the FDA in 1996 under the brand name Aricept, it remains one of the most widely prescribed medications for dementia worldwide.
Donepezil is indicated for the treatment of mild, moderate, and severe Alzheimer's disease. Clinical trials have consistently demonstrated modest but statistically significant improvements in cognitive function, global clinical status, and activities of daily living compared to placebo. While donepezil does not halt or reverse the neurodegenerative process, it can slow the rate of symptomatic decline for a period of months to years. It is also being investigated for potential benefits in vascular dementia and other cognitive disorders.
The standard dosing regimen begins at 5 mg once daily, typically taken at bedtime, with the option to increase to 10 mg daily after four to six weeks. A 23 mg extended-release formulation is available for patients with moderate to severe disease. Common side effects are cholinergic in nature and include nausea, diarrhea, insomnia, and muscle cramps. Donepezil has a long half-life of approximately 70 hours, allowing for convenient once-daily dosing and steady-state plasma levels.
Mechanism of Action
Reversible Acetylcholinesterase Inhibition
Donepezil is a piperidine-class reversible inhibitor of acetylcholinesterase (AChE) with high selectivity for AChE over butyrylcholinesterase (BuChE), exhibiting a selectivity ratio >1000-fold. It binds simultaneously to the catalytic anionic site (CAS) via its indanone moiety and the peripheral anionic site (PAS) via its benzylpiperidine group within the AChE active site gorge. This dual-site binding provides an inhibition constant (Ki) of approximately 5.7 nM, making it one of the most potent AChE inhibitors in clinical use. By blocking AChE-mediated hydrolysis of acetylcholine (ACh), donepezil increases synaptic ACh concentrations in cholinergic projections from the nucleus basalis of Meynert to the hippocampus and neocortex — regions severely depleted in Alzheimer's disease (PMID: 8876023).
Cholinergic Signaling Cascade Enhancement
Elevated synaptic ACh activates both muscarinic (M1–M5) and nicotinic (alpha4beta2, alpha7) acetylcholine receptors. M1 muscarinic receptor activation in cortical pyramidal neurons stimulates phospholipase C (PLC) → IP3/DAG → PKC signaling, which promotes non-amyloidogenic processing of amyloid precursor protein (APP) via alpha-secretase (ADAM10), reducing amyloid-beta (Abeta) production. Alpha7 nicotinic receptor activation triggers calcium-dependent PI3K/Akt signaling and MAPK/ERK cascades that support neuronal survival and synaptic plasticity. Donepezil also enhances long-term potentiation (LTP) at Schaffer collateral–CA1 synapses through augmented cholinergic modulation (PMID: 12649318).
Non-Cholinergic Neuroprotective Effects
Beyond cholinesterase inhibition, donepezil exerts direct neuroprotective effects through blockade of voltage-gated sodium channels and attenuation of glutamate-induced excitotoxicity. It inhibits mitochondrial permeability transition pore (mPTP) opening, preserving mitochondrial membrane potential and reducing cytochrome c release under oxidative stress. Donepezil also suppresses microglial activation and reduces pro-inflammatory cytokine release (TNF-alpha, IL-1beta) via sigma-1 receptor agonism, providing anti-neuroinflammatory effects independent of AChE inhibition. Clinical doses of 5–10 mg/day produce ~60–80% cortical AChE inhibition at steady state (PMID: 15094072).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Best for Impairment:: Most effective for treating cognitive decline.. Variable in Healthy:: Benefits less consistent in cognitively healthy individuals.. Long Half-Life:: Reaches steady state after several weeks.
- Most effective for treating cognitive decline.
- Benefits less consistent in cognitively healthy individuals.
- Reaches steady state after several weeks.
Safety Profile
Safety Profile: Donepezil (FDA Approved)
Overview Donepezil (brand name Aricept) is an FDA-approved reversible acetylcholinesterase inhibitor used for the treatment of mild, moderate, and severe Alzheimer disease. It is one of the most widely prescribed dementia medications globally. Its cholinergic mechanism produces predictable side effects, predominantly gastrointestinal and cardiac.
Common Side Effects
- Nausea (11-19% of patients), diarrhea (10-15%), and vomiting (5-8%) — most frequent during dose titration
- Insomnia and vivid dreams (6-14%), often necessitating morning dosing
- Fatigue and drowsiness
- Muscle cramps (6-8%), particularly nocturnal leg cramps
- Anorexia and weight loss (3-8%), concerning in elderly patients
- Headache and dizziness (8-10%)
Serious Adverse Effects
- Cardiac effects: bradycardia, heart block (first- and second-degree), and syncope due to vagotonic cholinergic stimulation; particular risk in patients with sick sinus syndrome or conduction abnormalities
- Seizures: cholinergic agents may lower seizure threshold
- GI hemorrhage: increased gastric acid secretion may exacerbate peptic ulcer disease
- Rhabdomyolysis: rare but reported, particularly in combination with statins
- Neuroleptic malignant syndrome: very rare reports
- Bladder outflow obstruction: cholinergic stimulation may exacerbate urinary retention
Contraindications
- Known hypersensitivity to donepezil or piperidine derivatives
- Severe cardiac conduction abnormalities (sick sinus syndrome without pacemaker, third-degree heart block)
- Active peptic ulcer disease or GI bleeding
- Severe hepatic impairment (limited data)
- History of rhabdomyolysis with cholinesterase inhibitors
Drug Interactions
- Anticholinergic medications (diphenhydramine, oxybutynin, tricyclic antidepressants): directly antagonize donepezil's mechanism, reducing efficacy — this is one of the most clinically significant interactions
- Beta-blockers and calcium channel blockers: additive bradycardia risk
- Succinylcholine-type muscle relaxants: enhanced neuromuscular blockade during anesthesia
- NSAIDs: increased GI bleeding risk due to combined gastric acid effects
- CYP3A4 and CYP2D6 inhibitors (ketoconazole, fluoxetine, paroxetine): may increase donepezil levels
- CYP3A4 inducers (phenytoin, carbamazepine, rifampin): may reduce donepezil efficacy
Special Populations
- Elderly patients are the primary population; dose titration should be gradual (5 mg for 4-6 weeks before increasing to 10 mg)
- Renal impairment: no dose adjustment needed
- Hepatic impairment: use with caution; metabolism may be affected
- Not indicated for pediatric use
Dosage Considerations
- Starting dose: 5 mg once daily at bedtime
- May increase to 10 mg/day after 4-6 weeks
- 23 mg/day tablet available for moderate-to-severe disease (higher GI side effect rate)
Pharmacokinetic Profile
Donepezil — Pharmacokinetic Curve
SubcutaneousMolecular Structure
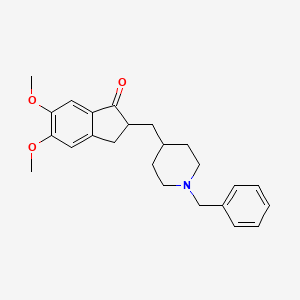
- Formula
- C24H29NO3
- Weight
- 379.5 Da
- PubChem CID
- 3152
- Exact Mass
- 379.2147 Da
- LogP
- 4.3
- TPSA
- 38.8 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 6
- Complexity
- 510
Identifiers (SMILES, InChI)
InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3
ADEBPBSSDYVVLD-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Cholinergic Effects:: Nausea, diarrhea, and muscle cramps common.
- Sleep Disturbances:: Insomnia and vivid dreams reported.
- Bradycardia:: Can slow heart rate; caution with cardiac conditions.
- GI Issues:: Digestive problems frequent, especially initially.
References (5)
- [1]Donepezil for dementia due to Alzheimer's disease
→ Evaluates donepezil's modest benefits in cognitive function and daily living activities over 12-24 weeks, noting higher doses produce more adverse events.
- [2]Clinical efficacy and safety of donepezil in the treatment of Alzheimer's disease in Chinese patients
→ Reviews donepezil effectiveness in Chinese populations, finding it generally safe with potential impacts on disease biomarkers.
- [4]Impact of Donepezil and Memantine on Behavioral and Psychological Symptoms of Alzheimer Disease
→ Study of 85 moderate Alzheimer's patients found both medications significantly reduced behavioral symptoms, with good tolerability.
- [5]Donepezil in the treatment of ischemic stroke: Review and future perspective
→ Explores donepezil's potential neuroprotective effects in ischemic stroke, noting preclinical promise but limited clinical evidence.
- [3]Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer's disease
→ Systematic review comparing three cholinesterase inhibitors through comprehensive database searches to evaluate their therapeutic effectiveness.
Doliracetam
Doliracetam is a cyclic GABA derivative and nootropic agent within the racetam family, structurally related to substances like piracetam and oxiracetam. It is p
Dong Quai
Dong Quai (Angelica sinensis) is a traditional Chinese medicinal herb containing compounds such as ligustilide, ferulic acid, and natural coumarin derivatives.