Brivaracetam
Brivaracetam is a third-generation racetam derivative and anticonvulsant medication approved for the adjunctive treatment of partial-onset (focal) seizures in patients with epilepsy. It binds with high affinity to synaptic vesicle glycoprotein 2A (SV2A), a mechanism shared with but more selective than its predecessor levetiracetam.
Overview
Brivaracetam (marketed as Briviact) is a next-generation anticonvulsant belonging to the racetam class of compounds, developed as a structural analog of levetiracetam (Keppra) with enhanced pharmacological properties. It was approved by the U.S. Food and Drug Administration in 2016 and by the European Medicines Agency for the adjunctive treatment of focal (partial-onset) seizures in adults and adolescents aged 16 years and older, with subsequent approvals extending to younger pediatric populations. Brivaracetam is available in oral tablet, oral solution, and intravenous formulations.
The primary mechanism of action of brivaracetam involves high-affinity binding to synaptic vesicle glycoprotein 2A (SV2A), a transmembrane protein ubiquitously expressed in synaptic vesicles that modulates neurotransmitter release. Brivaracetam demonstrates approximately 15–30 times greater binding affinity for SV2A compared to levetiracetam, and it exhibits faster brain penetration and more selective engagement of its target. Unlike levetiracetam, which has additional effects on ion channels and other receptors, brivaracetam appears to act more specifically through SV2A modulation, which may contribute to its distinct tolerability profile.
In pivotal phase III clinical trials, brivaracetam demonstrated statistically significant reductions in focal seizure frequency compared to placebo when used as adjunctive therapy. The most common adverse effects include somnolence, dizziness, and fatigue, but notably, the incidence of behavioral and psychiatric adverse events (such as irritability and aggression) appears to be lower than that observed with levetiracetam, making it a potential alternative for patients who experience neuropsychiatric side effects with the older compound. Brivaracetam is metabolized primarily by CYP2C19 and amidase hydrolysis, and dosage adjustments may be warranted in patients with hepatic impairment.
Mechanism of Action
High-Affinity SV2A Ligand
Brivaracetam is a next-generation synaptic vesicle glycoprotein 2A (SV2A) ligand and chemical analog of levetiracetam, with approximately 15-30-fold higher binding affinity for SV2A (IC50 ~ 28 nM vs. ~770 nM for levetiracetam). SV2A is a 12-transmembrane domain glycoprotein ubiquitously expressed on synaptic vesicles throughout the CNS, functioning as a critical regulator of vesicle-mediated neurotransmitter release (PMID: 26332317).
SV2A-Mediated Vesicle Release Modulation
SV2A modulates neurotransmitter release by interacting with synaptotagmin-1, the primary calcium sensor for fast synchronous exocytosis. SV2A assists in the proper priming of synaptic vesicles by facilitating the conformational change of synaptotagmin-1 required for calcium-dependent SNARE complex assembly. Brivaracetam binding to SV2A alters this interaction, reducing the readily releasable pool of synaptic vesicles and attenuating excessive neurotransmitter release during high-frequency neuronal firing characteristic of epileptic seizures, without significantly affecting normal synaptic transmission (PMID: 22538469).
Differential Mechanism from Levetiracetam
Unlike levetiracetam, brivaracetam has negligible activity at AMPA receptors, GABA_A receptors, or neuronal voltage-gated sodium channels at therapeutic concentrations. Its anticonvulsant effect is attributed almost exclusively to SV2A binding. Brivaracetam also demonstrates a faster brain penetration rate due to higher lipophilicity (LogP ~ 1.04), achieving maximal brain occupancy within minutes of administration compared to hours for levetiracetam (PMID: 24519919).
Network Excitability & Epileptogenesis
Chronic SV2A modulation by brivaracetam may have anti-epileptogenic properties beyond acute seizure suppression. In kindling models, brivaracetam slows the rate of seizure progression and reduces interictal spike frequency, potentially by restoring SV2A protein levels that decline during epileptogenesis and by normalizing aberrant synaptic vesicle cycling in reorganized hippocampal circuits (PMID: 25307356).
Research
Reported Effects
Levetiracetam Comparison:: Often successful in patients who did not tolerate or respond to levetiracetam (Keppra).. Adjunctive Efficacy:: Works well both as a standalone therapy and when added to existing medication regimens.. Long-term Retention:: Shows high patient retention rates in real-world studies, indicating sustained efficacy over years.
- Often successful in patients who did not tolerate or respond to levetiracetam (Keppra).
- Works well both as a standalone therapy and when added to existing medication regimens.
- Shows high patient retention rates in real-world studies, indicating sustained efficacy over years.
Safety Profile
Safety Profile: Brivaracetam
Common Side Effects
- Somnolence/drowsiness (~15-20% of patients)
- Dizziness (~11-16%)
- Fatigue (~9-14%)
- Nausea and vomiting (~5-9%)
- Headache
- Irritability and mood changes
- Upper respiratory tract infections
- Decreased appetite
Serious Adverse Effects
- Suicidal ideation and behavior: Class warning for all antiepileptic drugs; monitor for depression, anxiety, behavioral changes
- Psychiatric symptoms: Psychotic symptoms, aggression, agitation, anger reported (lower incidence than levetiracetam)
- Hypersensitivity reactions: Bronchospasm, angioedema (rare)
- Hematologic: Leukopenia, neutropenia (rare; periodic CBC recommended)
- Hepatic: Elevated transaminases; rare cases of jaundice
- Withdrawal seizures: Abrupt discontinuation may precipitate seizures or status epilepticus; taper gradually
Contraindications
- Known hypersensitivity to brivaracetam, other pyrrolidone derivatives, or any excipients
- Severe hepatic impairment (Child-Pugh C) -- maximum recommended dose is 150 mg/day with dose adjustment
Drug Interactions
- Rifampin: Strong enzyme inducer decreases brivaracetam plasma levels by ~45%; dose adjustment needed
- Carbamazepine: Brivaracetam increases carbamazepine-epoxide (active metabolite) levels by ~25%; monitor for carbamazepine toxicity
- Phenytoin: Brivaracetam may increase phenytoin levels by inhibiting CYP2C19; monitor phenytoin levels
- Levetiracetam: No additional benefit from co-administration (same SV2A target); not recommended together
- Oral contraceptives: No clinically significant interaction at recommended doses
- Alcohol/CNS depressants: Additive sedation; avoid or use with caution
Population-Specific Considerations
- Pregnancy: Category C equivalent; crosses placenta. Registry data limited. Folic acid supplementation recommended. Enroll in pregnancy registries
- Lactation: Excreted in breast milk; weigh benefits vs. risks. Monitor infant for sedation
- Pediatric (4+ years): FDA-approved for adjunctive treatment of partial-onset seizures; weight-based dosing
- Elderly: No specific dose adjustment; however, increased sensitivity to CNS effects
- Hepatic impairment: Reduce starting dose in all stages; max 150 mg/day in severe impairment (Child-Pugh C)
- Renal impairment: No dose adjustment needed, including in dialysis patients
- Psychiatric history: Use with heightened monitoring; lower risk of behavioral AEs than levetiracetam but not negligible
Pharmacokinetic Profile
Brivaracetam — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Typically starts at 50 mg twice daily, with a therapeutic range of 50 mg to 200 mg per day.
Molecular Structure
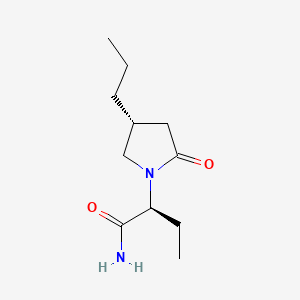
- Formula
- C11H20N2O2
- Weight
- 212.29 Da
- PubChem CID
- 9837243
- Exact Mass
- 212.1525 Da
- LogP
- 1
- TPSA
- 63.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 5
- Complexity
- 253
Identifiers (SMILES, InChI)
InChI=1S/C11H20N2O2/c1-3-5-8-6-10(14)13(7-8)9(4-2)11(12)15/h8-9H,3-7H2,1-2H3,(H2,12,15)/t8-,9+/m1/s1
MSYKRHVOOPPJKU-BDAKNGLRSA-NSafety Profile
Common Side Effects
- Somnolence:: Excessive sleepiness and fatigue are the most commonly reported neurological side effects.
- Psychiatric Symptoms:: While improved over Keppra, some users still report irritability, depression, or anxiety.
- Dermatologic Risks:: Rare but serious skin reactions can occur, necessitating immediate discontinuation.
- Dizziness:: Frequent reports of lightheadedness or balance issues, especially during initial dosing.
References (4)
- [1]Brivaracetam: Pharmacology, Clinical Efficacy, and Safety in Epilepsy
→ This review summarizes brivaracetam's role as a potent SV2A ligand with rapid brain penetration and effective seizure reduction in both clinical trials and real-world settings.
- [2]Narrative Review of Brivaracetam: Preclinical Profile and Clinical Benefits in the Treatment of Patients with Epilepsy
→ This study highlights brivaracetam's 15- to 30-fold higher affinity for SV2A compared to levetiracetam and its effectiveness even in patients who failed prior treatments.
- [3]Effectiveness and tolerability of brivaracetam in patients with epilepsy stratified by comorbidities and etiology in the real world
→ A large-scale analysis showing that brivaracetam significantly reduces seizure frequency across diverse patient populations, including those with cognitive or psychiatric comorbidities.
- [4]Brivaracetam: a novel antiepileptic drug for focal-onset seizures
→ Explains that brivaracetam slows synaptic vesicle mobilization more effectively than its predecessor, providing broad protection against focal and generalized seizures.
Bradykinin
Bradykinin is a nine-amino acid vasoactive peptide (Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg) that serves as one of the most potent endogenous mediators of vasodilat
Bromantane
Bromantane is a unique Russian compound that acts as both an actoprotector (enhancing physical performance) and an anxiolytic. It upregulates tyrosine hydroxyla