Adrafinil
Adrafinil is a synthetic eugeroic (wakefulness-promoting) prodrug that is metabolized to modafinil in the liver, historically used to promote alertness without the hyperactivity associated with traditional stimulants.
Overview
Adrafinil is a synthetic benzhydryl sulfinyl compound first developed in the late 1970s by the French pharmaceutical company Laboratoire Lafon. It functions as a prodrug of modafinil, being converted to its active metabolite primarily through hepatic amidase-mediated hydrolysis. Adrafinil was marketed in France under the brand name Olmifon for the treatment of narcolepsy and age-related drowsiness until its discontinuation in 2011, following the wider availability of modafinil itself.
The pharmacological profile of adrafinil closely mirrors that of modafinil, promoting wakefulness through mechanisms that likely involve adrenergic, dopaminergic, and histaminergic signaling, though the precise mechanism remains incompletely understood. Unlike amphetamines, adrafinil does not appear to produce significant euphoria, rebound hypersomnolence, or marked sympathomimetic effects. Its onset of action is slower than modafinil (typically 60-90 minutes) due to the additional metabolic conversion step required.
Because adrafinil undergoes hepatic metabolism to produce modafinil, chronic use raises concerns about liver enzyme elevation and hepatotoxicity. Liver function monitoring has been recommended for long-term users. Adrafinil is not a scheduled substance in most jurisdictions, unlike modafinil, which has contributed to its popularity in nootropic communities. However, it is banned by the World Anti-Doping Agency as a non-specified stimulant. Common side effects include headache, nausea, and insomnia.
Mechanism of Action
Prodrug Metabolism
Adrafinil is a prodrug that undergoes hepatic biotransformation to its active metabolite modafinil via hydrolytic cleavage of the diphenylmethyl sulfinyl hydroxamic acid moiety. This conversion is mediated by hepatic microsomal enzymes and esterases. A secondary metabolite, modafinilic acid (inactive), is also produced (PMID: 10498882).
Hypocretin/Orexin System
Modafinil, the active metabolite, activates hypothalamic orexin (hypocretin) neurons in the lateral hypothalamus. Orexin-A and orexin-B then activate OX1R and OX2R receptors on downstream wake-promoting nuclei including the tuberomammillary nucleus (histaminergic), locus coeruleus (noradrenergic), and dorsal raphe (serotonergic), stabilizing the wake state.
Dopamine Transporter Inhibition
Modafinil binds to the dopamine transporter (DAT) at the presynaptic terminal, inhibiting dopamine reuptake and increasing extracellular dopamine in the prefrontal cortex, nucleus accumbens, and striatum. This mechanism was confirmed by DAT-knockout studies showing abolished wake-promoting effects (PMID: 19037199).
Glutamate/GABA Balance
The compound enhances glutamatergic transmission while reducing GABAergic tone in cortical and subcortical regions. It increases extracellular glutamate in the medial preoptic area and posterior hypothalamus, while decreasing GABA in the cortex, medial preoptic area, and posterior hypothalamus, shifting the excitatory/inhibitory balance toward wakefulness.
Histaminergic Contribution
Adrafinil/modafinil increases histamine release from tuberomammillary nucleus neurons, contributing to cortical arousal through H1 receptor activation, though this appears secondary to orexin-mediated activation rather than direct histaminergic action.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Individual Variation:: Response varies based on liver enzyme activity and metabolism.. Tolerance Development:: Some users report diminishing effects with regular use.
- Response varies based on liver enzyme activity and metabolism.
- Some users report diminishing effects with regular use.
Safety Profile
Safety Profile: Adrafinil
Common Side Effects
- CNS effects: Headache, dizziness, insomnia (especially with late-day dosing), anxiety, irritability
- GI effects: Nausea, stomach pain, diarrhea
- Dermatologic: Skin irritation, rash (uncommon)
- Other: Dry mouth, elevated blood pressure
Serious Adverse Effects
- Hepatotoxicity: Adrafinil requires hepatic conversion to modafinil (its active metabolite); this conversion produces modafinilic acid, which may cause liver enzyme elevation with chronic use — this was the primary safety concern leading to its limited adoption
- Stevens-Johnson syndrome/Toxic epidermal necrolysis (SJS/TEN): Rare but potentially fatal; same class effect as modafinil — discontinue immediately at first sign of rash
- Psychiatric effects: Rare reports of psychosis, mania, or suicidal ideation (class effect of eugeroics)
- Orofacial dyskinesia: Reported with chronic use in early French clinical data
Contraindications
- Known hypersensitivity to adrafinil or modafinil
- Severe hepatic impairment (hepatotoxicity risk)
- History of Stevens-Johnson syndrome or drug-induced rash with modafinil/armodafinil
- Unstable angina or recent myocardial infarction
- History of psychosis or severe anxiety disorders
Drug Interactions
- CYP3A4 substrates: Modafinil (active metabolite) induces CYP3A4, potentially reducing efficacy of: hormonal contraceptives, cyclosporine, midazolam, triazolam
- CYP2C19 substrates: Modafinil inhibits CYP2C19; may increase levels of: omeprazole, phenytoin, diazepam, propranolol
- Warfarin: Monitor INR — modafinil may affect warfarin metabolism
- Stimulants/caffeine: Additive CNS stimulation; increased cardiovascular risk
- MAOIs: Potential for hypertensive crisis; avoid combination
Special Populations
- Pregnancy/Lactation: Contraindicated — modafinil (active metabolite) is associated with congenital malformations (FDA pregnancy registry data)
- Pediatric: Not recommended; modafinil was denied pediatric approval by FDA due to Stevens-Johnson syndrome risk
- Elderly: Start with reduced doses; increased hepatic and cardiovascular sensitivity
- Renal impairment: Active metabolite is renally excreted; dose adjustment may be needed
- Hepatic impairment: Contraindicated in severe impairment; use with extreme caution in mild-moderate
Monitoring Recommendations
- Liver function tests (LFTs): Baseline and every 3 months with chronic use — this is critical and non-negotiable
- Blood pressure and heart rate monitoring
- Dermatologic assessment — discontinue immediately with any rash
- Psychiatric symptom monitoring
- CBC periodically (rare reports of blood dyscrasias with modafinil class)
Regulatory Note: Adrafinil was a prescription drug in France (Olmifon, discontinued 2011). It is unscheduled in the US but not FDA-approved. It is banned by WADA as a non-specified stimulant. Modafinil (Provigil), the active metabolite, is Schedule IV in the US and generally preferred due to the hepatotoxicity concern with adrafinil.
Pharmacokinetic Profile
Adrafinil — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- 300-600mg per day, usually taken in the morning.
Molecular Structure
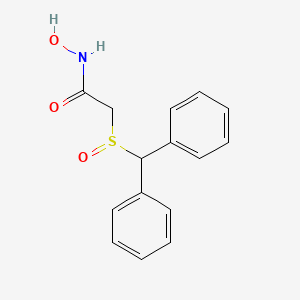
- Formula
- C15H15NO3S
- Weight
- 289.4 Da
- PubChem CID
- 3033226
- Exact Mass
- 289.0773 Da
- LogP
- 1.6
- TPSA
- 85.6 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 5
- Complexity
- 318
Identifiers (SMILES, InChI)
InChI=1S/C15H15NO3S/c17-14(16-18)11-20(19)15(12-7-3-1-4-8-12)13-9-5-2-6-10-13/h1-10,15,18H,11H2,(H,16,17)
CGNMLOKEMNBUAI-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Liver Strain:: Long-term use may stress the liver; liver function monitoring is recommended.
- Headaches:: Some users report headaches, often mitigated by staying hydrated.
- Insomnia:: Taking too late in the day can disrupt sleep.
References (9)
- [1]Behavioral Activating Effects of Adrafinil in Aged Canines
→ Study demonstrates that adrafinil increases locomotor activity and directed sniffing in aged dogs, with variable individual responses.
- [2]Identification of adrafinil and its main metabolite modafinil in human hair
→ Research shows adrafinil and modafinil can be detected in human hair following single consumption, helping distinguish trafficking cases from self-use.
- [3]Adrafinil disrupts performance on a delayed nonmatching-to-position task in aged beagle dogs
→ Study reveals adrafinil significantly impairs working memory in aged dogs, potentially via increased noradrenergic transmission in prefrontal cortex.
- [4]Adrafinil: Effects on behavior and cognition in aged canines
→ Research characterizes adrafinil as a behavioral stimulant with cognitive enhancement potential in aged dogs, though mechanisms remain unclear.
- [5]Oral Administration of Adrafinil Improves Discrimination Learning in Aged Beagle Dogs
→ Investigation demonstrates adrafinil significantly enhances discrimination learning in aged dogs, possibly through enhanced vigilance and motivation.
- [6]Fluorescent zinc and copper complexes for detection of adrafinil
→ Fluorescent complexes provide detection method for adrafinil in paper-based microfluidic devices, useful for identifying banned performance-enhancing substances.
- [7]Thermal degradation of modafinil analogs in GC injector
→ Analysis shows thermal degradation of modafinil and adrafinil compounds in gas chromatography injectors can produce unexpected byproducts.
- [9]Development and Validation of an Analytical Method to Identify Novel Modafinil Analogs
→ LC-HRMS method effectively identifies and quantifies novel modafinil analogs in dietary supplements, protecting consumers from undisclosed active ingredients.
- [8]Comparison of the effects of adrafinil, propentofylline, and nicergoline on behavior in aged dogs
→ Research indicates adrafinil treatment may improve quality of life in aged dogs by increasing exploratory behavior and alertness.
Adipotide (FTPP)
Adipotide, also known as FTPP (fat-targeted proapoptotic peptide), is an experimental peptidomimetic that selectively targets and destroys blood vessels supplyi
Adrenomedullin
Adrenomedullin (ADM) is a 52-amino acid vasoactive peptide originally isolated in 1993 by Kitamura et al. from human pheochromocytoma tissue based on its abilit