MIF-1
A naturally occurring tripeptide (Pro-Leu-Gly-NH2) derived from oxytocin that modulates dopaminergic neurotransmission, investigated for its anti-opiate, antidepressant, and anti-Parkinsonian properties.
Overview
MIF-1 (Melanocyte-Inhibiting Factor 1, also known as PLG or Pro-Leu-Gly-NH2) is a naturally occurring tripeptide originally isolated from hypothalamic tissue and identified as the C-terminal fragment of oxytocin. Despite its historical name suggesting melanocyte-inhibiting activity, its most significant pharmacological actions involve modulation of dopaminergic neurotransmission. MIF-1 acts as a positive allosteric modulator of dopamine D2 receptors, enhancing dopamine binding and signaling without directly activating the receptor — a mechanism that distinguishes it from conventional dopamine agonists and offers potential therapeutic advantages in conditions characterized by dopaminergic dysfunction.
The clinical and preclinical research on MIF-1 spans several neurological and psychiatric domains. In Parkinson's disease, where progressive loss of dopaminergic neurons in the substantia nigra is the hallmark pathology, MIF-1 has shown the ability to potentiate the effects of L-DOPA and dopamine agonists in animal models, suggesting utility as an adjunctive therapy that could reduce the doses (and side effects) of conventional antiparkinsonian medications. In depression research, MIF-1 demonstrated antidepressant effects in clinical trials conducted in the 1970s–80s, with improvements reported in patients who were refractory to conventional treatments. The peptide also exhibits anti-opiate properties, attenuating morphine-induced analgesia and reducing physical dependence in preclinical models, which has sparked interest in its application for opioid withdrawal and addiction management.
MIF-1 is typically administered via subcutaneous or intravenous injection due to its peptide nature, though its small size (molecular weight ~285 Da) and amidated C-terminus provide some resistance to enzymatic degradation. The peptide has an excellent safety profile in published studies, with minimal reported adverse effects. MIF-1 is structurally related to other endogenous neuropeptides including Selank and Semax that modulate central nervous system function, and it is sometimes discussed alongside BPC-157 as an example of a naturally derived peptide with broad neurorestorative potential. Research interest in MIF-1 has been relatively quiet in recent decades, though renewed attention to allosteric dopamine modulation as a therapeutic strategy may revive interest in this unique tripeptide.
Mechanism of Action
MIF-1 (Melanocyte-Inhibiting Factor-1, Pro-Leu-Gly-NH2) is a tripeptide that acts as an endogenous modulator of dopaminergic and opioid neurotransmission. Its primary mechanism involves allosteric modulation of dopamine D2 receptors, where it enhances dopamine binding affinity and receptor signaling without directly activating the receptor itself. MIF-1 also functions as an anti-opioid peptide by antagonizing mu-opioid receptor signaling, which reduces opioid tolerance and dependence. It achieves this by uncoupling the mu-opioid receptor from its associated Gi/Go proteins, thereby attenuating cAMP inhibition and downstream signaling cascades.
At the molecular level, MIF-1 interacts with the allosteric sites on both D2 dopamine receptors and mu-opioid receptors, shifting receptor conformations to favor or disfavor agonist binding respectively. In dopaminergic pathways, this enhances mesolimbic and nigrostriatal dopamine transmission, increasing locomotor activity and mood-related behaviors. MIF-1 also modulates oxytocin release from the hypothalamus and influences GABA-ergic transmission, contributing to its anxiolytic properties. The peptide crosses the blood-brain barrier efficiently due to its small size and amidated C-terminus.
Therapeutically, MIF-1 has been investigated for Parkinson's disease due to its dopamine-potentiating effects, for depression through its mood-elevating properties, and for opioid addiction by reducing tolerance and withdrawal symptoms. Its dual action on dopaminergic enhancement and opioid modulation makes it a unique pharmacological tool for conditions involving dysregulated reward circuitry and movement disorders.
Reconstitution Calculator
MIF-1
MIF-1 (Melanocyte-Stimulating Hormone Release-Inhibiting Factor-1, Pro-Leu-Gly-N

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Depression Models:: Effective at 0.1-1.0 mg/kg doses in chronic stress models with activity levels comparable to imipramine. Inverted U-Shaped Curve:: Clinical evidence shows increasing doses can result in decreasing therapeutic effects, requiring careful dose optimization. Rapid Onset:: Historical clinical trials reported faster onset of antidepressant action compared to traditional medications available at the time. Regional Brain Specificity:: Effects vary by brain region with notable activity in striatum, cortex, and olfactory areas but not uniformly across all regions
- Effective at 0.1-1.0 mg/kg doses in chronic stress models with activity levels comparable to imipramine
- Clinical evidence shows increasing doses can result in decreasing therapeutic effects, requiring careful dose optimization
- Historical clinical trials reported faster onset of antidepressant action compared to traditional medications available at the time
- Effects vary by brain region with notable activity in striatum, cortex, and olfactory areas but not uniformly across all regions
Safety Profile
At high doses, MIF-1 may paradoxically increase stress and pain sensitivity. As an opioid antagonist, it can interfere with opioid-based pain medications. There is limited long-term human safety data available, with most information derived from animal studies or older, smaller clinical trials.
Pharmacokinetic Profile
Molecular Structure
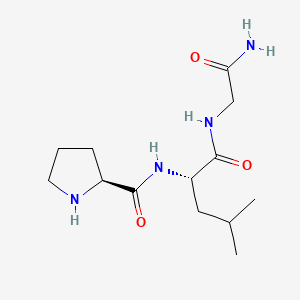
- Formula
- C13H24N4O3
- Weight
- 284.35 Da
- PubChem CID
- 92910
- Exact Mass
- 284.1848 Da
- LogP
- -1.4
- TPSA
- 113 Ų
- H-Bond Donors
- 4
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 7
- Complexity
- 371
Identifiers (SMILES, InChI)
InChI=1S/C13H24N4O3/c1-8(2)6-10(12(19)16-7-11(14)18)17-13(20)9-4-3-5-15-9/h8-10,15H,3-7H2,1-2H3,(H2,14,18)(H,16,19)(H,17,20)/t9-,10-/m0/s1
NOOJLZTTWSNHOX-UWVGGRQHSA-NSafety Profile
Common Side Effects
- High Dose Effects:: At 10 mg/kg, MIF-1 may increase stress responses and produce hyperalgesia rather than therapeutic benefits
- Regional Variability:: Effects on neurotransmitter systems vary by brain region, potentially causing unpredictable responses in some individuals
- Opioid Interactions:: As an opioid antagonist, may interfere with pain management or medications acting through opioid pathways
- Limited Safety Data:: Minimal long-term human safety data available, with most evidence from animal studies or older clinical trials
References (8)
- [2]MIF-1 is active in a chronic stress animal model of depression
→ Low doses of MIF-1 (0.1 and 1.0 mg/kg) significantly increased activity and decreased stress-related behaviors in chronically stressed rats, demonstrating antidepressant effects comparable to imipramine in this depression model.
- [3]Neurological effects of MIF-1, MSH, and opiate peptides in clinical studies
→ Clinical research documenting the neurological and behavioral effects of MIF-1 in human subjects, establishing its relevance for neuropsychiatric applications.
- [4]An extraordinary relationship involving MIF-1 and other peptides
→ Retrospective review of collaborative clinical work showing MIF-1 was effective in treating human depression with greater efficacy and faster onset of action than traditional antidepressants available at the time of clinical trials.
- [6]MIF-1 and Tyr-MIF-1 augment GABA-stimulated benzodiazepine receptor binding
→ Both MIF-1 and Tyr-MIF-1 significantly augmented GABA-stimulated benzodiazepine receptor binding at low doses, indicating interactions between peptide and GABA-benzodiazepine receptor systems relevant to anxiety modulation.
- [7]Biphasic effects of MIF-1 and Tyr-MIF-1 on apomorphine-induced stereotypy in rats
→ MIF-1 showed biphasic dose-dependent effects on dopamine-mediated behaviors, increasing responses at moderate doses but decreasing them at high doses, suggesting complex modulation of dopaminergic neurotransmission.
- [8]Effects of melanostatine (MIF-1) on focal potentials in slices of rat brain cortex
→ MIF-1 demonstrated activatory influence on CNS cellular activity, enhancing focal potential amplitudes and facilitating post-tetanic potentiation, supporting the neurophysiological basis for its antidepressant properties.
- [1]From MIF-1 to endomorphin: the Tyr-MIF-1 family of peptides
→ Comprehensive review establishing MIF-1 as the first hypothalamic peptide shown to act on the brain rather than the pituitary, demonstrating antiopiate activity and serving as a prototypic molecule for discovering endogenous opioid antagonists.
- [5]MIF-1: effects on norepinephrine, dopamine and serotonin metabolism in certain discrete brain regions
→ MIF-1 at 5 mg/kg increased striatal tyrosine hydroxylase activity by 25% and dopamine levels in multiple brain regions, demonstrating enhanced turnover of dopamine and norepinephrine neurotransmitter systems.
MGF
Non-pegylated IGF-1 splice variant produced locally in muscle tissue following mechanical stress. Has a very short half-life compared to PEG-MGF, requiring more
Milk Thistle
Milk thistle (Silybum marianum) is a medicinal herb containing silymarin, a mixture of flavonolignans with silibinin as the main active component. It is primari