Centrophenoxine (Meclofenoxate)
Centrophenoxine, also known as meclofenoxate, is a synthetic nootropic compound that combines DMAE with pCPA. It is studied for its potential to reduce lipofuscin accumulation in neurons and support cholinergic neurotransmission.
Overview
Centrophenoxine, marketed under the name meclofenoxate, is a synthetic ester of dimethylaminoethanol (DMAE) and para-chlorophenoxyacetic acid (pCPA). Developed in 1959 at the French National Scientific Research Center, it was originally investigated as a treatment for age-related cognitive decline. The compound is designed to be more bioavailable than DMAE alone, readily crossing the blood-brain barrier to exert its effects on central nervous system function.
The primary mechanism of interest involves centrophenoxine's ability to reduce lipofuscin, an age-related pigment that accumulates in neurons and other cells over time. By facilitating the removal of this cellular waste product, centrophenoxine may help maintain neuronal health during aging. Additionally, as a precursor to DMAE, it may support the synthesis of acetylcholine, a neurotransmitter critical for memory and learning processes.
Centrophenoxine has been the subject of numerous preclinical studies and some clinical investigations, particularly in elderly populations with cognitive impairment. While results have shown modest improvements in memory and alertness in certain studies, the overall body of clinical evidence remains limited by small sample sizes and methodological variability. It is available as a dietary supplement in many countries and is generally considered to have a favorable safety profile at standard doses.
Mechanism of Action
DMAE Ester — Cholinergic Precursor
Centrophenoxine (meclofenoxate) is an ester of dimethylaminoethanol (DMAE) and para-chlorophenoxyacetic acid (pCPA), a synthetic plant growth hormone analog. Following oral absorption, centrophenoxine is rapidly cleaved by plasma esterases into its two components. DMAE crosses the blood-brain barrier and undergoes sequential methylation by phosphatidylethanolamine N-methyltransferase (PEMT), converting phosphatidylethanolamine (PE) to phosphatidylcholine (PC) in neuronal membranes. DMAE also serves as a precursor for acetylcholine (ACh) synthesis via choline kinase-mediated phosphorylation (PMID: 3305166).
Lipofuscin Clearance & Lysosomal Function
Centrophenoxine's most distinctive pharmacological property is its ability to reduce lipofuscin accumulation in neurons — the age-related "wear and tear" pigment composed of oxidized lipids, cross-linked proteins, and metals trapped in residual lysosomes. Centrophenoxine enhances lysosomal enzyme activity (acid phosphatase, cathepsin D) and autophagy flux, promoting degradation and clearance of lipofuscin granules. Animal studies demonstrate 25-40% reduction in neuronal lipofuscin content after 3-6 months of treatment, correlating with improved cognitive performance in aged subjects (PMID: 7211776).
Membrane Phospholipid Turnover
By enhancing PE-to-PC conversion via PEMT, centrophenoxine increases the PC/PE ratio in neuronal membranes, improving membrane fluidity, receptor mobility, and ion channel function. Increased phosphatidylcholine content also enhances glucose transporter (GLUT1) incorporation into neuronal membranes, improving cerebral glucose uptake and utilization. The pCPA moiety may additionally contribute to membrane effects through its lipophilic interactions with phospholipid bilayers (PMID: 6194901).
RNA Synthesis & Gene Expression
Centrophenoxine increases total RNA content in aging neurons, including mRNA, rRNA, and tRNA, by enhancing RNA polymerase II activity. This reverses the age-related decline in protein synthesis capacity, restoring production of synaptic proteins, neurotransmitter receptors, and structural cytoskeletal elements essential for neuronal maintenance (PMID: 6887836).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Synergistic Stacking:: Most effective when combined with racetams (piracetam, aniracetam, oxiracetam) rather than as standalone supplement. Individual Variability:: Highly variable responses among users, with some experiencing significant benefits while others report cognitive impairment. Dose-Dependent:: Effects appear related to dosage, with some users finding lower doses (250-300mg) more tolerable than higher doses (500mg+). Time Course:: Some users report immediate effects while others note benefits develop over days to weeks of consistent use
- Most effective when combined with racetams (piracetam, aniracetam, oxiracetam) rather than as standalone supplement
- Highly variable responses among users, with some experiencing significant benefits while others report cognitive impairment
- Effects appear related to dosage, with some users finding lower doses (250-300mg) more tolerable than higher doses (500mg+)
- Some users report immediate effects while others note benefits develop over days to weeks of consistent use
Safety Profile
Common Side Effects
- Insomnia and restlessness, particularly when taken later in the day (centrophenoxine has mild stimulant properties)
- Nausea and gastrointestinal upset
- Headache, especially during initial use or at higher doses
- Dizziness and mild agitation
- Muscle tension or jaw clenching (uncommon)
- Increased irritability or anxiety in sensitive individuals
- Body odor changes: some users report a fishy smell due to DMAE (dimethylaminoethanol) metabolite trimethylamine
Serious Adverse Effects
- Cholinergic excess symptoms at high doses: excessive salivation, lacrimation, urination, diarrhea, and gastrointestinal distress (SLUDGE syndrome)
- Seizures have been reported in animal studies at very high doses; theoretical risk in humans, especially in seizure-prone individuals
- Depression or mood disturbances with chronic high-dose use
- Hepatotoxicity has not been well-characterized in humans but cannot be excluded with prolonged high-dose use
- Rare allergic or hypersensitivity reactions
Contraindications
- Seizure disorders (epilepsy): centrophenoxine may lower seizure threshold
- Bipolar disorder or major depression: cholinergic modulation may exacerbate mood instability
- Pregnancy and breastfeeding: contraindicated due to insufficient safety data and potential developmental effects from DMAE component
- Known hypersensitivity to meclofenoxate, DMAE, or pCPA (para-chlorophenoxyacetic acid)
- Parkinson's disease patients on anticholinergic therapy: centrophenoxine's cholinergic effects may counteract treatment
Drug Interactions
- Acetylcholinesterase inhibitors (donepezil, rivastigmine, galantamine): additive cholinergic effects may cause excessive acetylcholine activity
- Anticholinergic medications (atropine, benztropine, certain antihistamines): opposing mechanisms may reduce efficacy of either agent
- Racetam-class nootropics (piracetam, aniracetam): commonly combined in nootropic stacks, but the combination may amplify cholinergic side effects
- CNS stimulants (caffeine, amphetamines, modafinil): potential for additive stimulation, increasing anxiety and insomnia risk
- Lithium: theoretical interaction through cholinergic pathways; monitor closely
Special Populations
- Elderly: centrophenoxine has been studied primarily in aging populations for cognitive support; start at low doses (250 mg/day) and titrate gradually
- Children: safety not established; use is not recommended
- Hepatic or renal impairment: use with caution; reduced clearance may increase side effect risk
Pharmacokinetic Profile
Centrophenoxine (Meclofenoxate) — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Most commonly used at 250-500mg per serving, with many products containing 300-500mg capsules
Molecular Structure
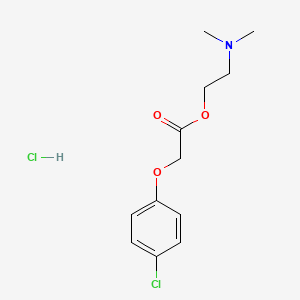
- Formula
- C12H17Cl2NO3
- Weight
- 294.17 Da
- PubChem CID
- 19379
- Exact Mass
- 293.0585 Da
- TPSA
- 38.8 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 7
- Complexity
- 228
Identifiers (SMILES, InChI)
InChI=1S/C12H16ClNO3.ClH/c1-14(2)7-8-16-12(15)9-17-11-5-3-10(13)4-6-11;/h3-6H,7-9H2,1-2H3;1H
FIVHOHCAXWQPGC-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Mood Effects:: Multiple users report depression, decreased emotional response, and general apathy when taking centrophenoxine
- Cognitive Impairment:: Some experience increased typos, attention deficits, poor listening comprehension, and brain fog rather than improvement
- Sleep Disruption:: Reports of early waking, altered sleep patterns, and difficulty falling asleep despite daytime fatigue
- Paradoxical Energy:: Some users note increased evening energy and impulsivity that interferes with normal sleep schedules
References (6)
- [2]Modulatory effects of centrophenoxine on different regions of ageing rat brain
→ Six-week administration to aged rats increased activities of antioxidant enzymes (catalase, SOD, glutathione reductase) and altered glutathione content, suggesting beneficial effects on oxidative stress in aging brain.
- [3]Centrophenoxine: effects on aging mammalian brain
→ Treatment in old mice for three months significantly improved learning performance in T-maze tests and reduced neuronal lipofuscin pigment in cerebral cortex and hippocampus compared to untreated controls.
- [4]Effects of centrophenoxine on lipofuscin in the retinal pigment epithelium of old mice
→ Three months of daily subcutaneous injections in 17-month-old mice resulted in significant reduction of lipofuscin pigment in retinal pigment epithelium while melanin remained unchanged.
- [5]Fluidizing effects of centrophenoxine in vitro on brain and liver membranes from different age groups of mice
→ In vitro study demonstrated centrophenoxine directly increased membrane fluidity in both synaptic plasma membranes and liver microsomes, with greater effects in liver tissue, independent of age.
- [6]Effects of centrophenoxine on cholinephosphotransferase activity in maternal and fetal guinea pig lung
→ Centrophenoxine and its metabolite p-chlorophenoxyacetate inhibited cholinephosphotransferase activity in adult lung but not fetal lung when administered to pregnant animals, though direct fetal administration caused inhibition.
- [1]The unapproved drug centrophenoxine (meclofenoxate) in cognitive enhancement dietary supplements
→ Study found centrophenoxine present in all seven analyzed US dietary supplements at dosages ranging from 79-251mg per serving, with daily intake recommendations exposing consumers to 237-752mg despite FDA non-approval.
Celastrus Paniculatus
Celastrus paniculatus, known as the 'Intellect Tree,' is a climbing shrub used in Ayurvedic medicine to enhance cognitive function and treat neurological disord
Cerebrolysin
**Cerebrolysin** is a parenterally administered neuropeptide preparation consisting of low-molecular-weight peptides and free amino acids derived from purified