Piceatannol
A naturally occurring hydroxylated analog of resveratrol found in grapes, passion fruit seeds, and berries, with superior bioavailability and enhanced anti-inflammatory, antioxidant, and metabolic benefits compared to its parent compound.
Overview
Piceatannol (3,3',4,5'-tetrahydroxy-trans-stilbene) is a stilbenoid polyphenol structurally related to resveratrol, differing by the addition of a single hydroxyl group at the 3' position. This seemingly minor modification confers substantially enhanced biological activity, including greater antioxidant capacity, improved Syk kinase inhibition, and stronger anti-inflammatory effects. Piceatannol occurs naturally in grapes, wine, passion fruit (Passiflora edulis) seeds, white tea, Japanese knotweed, and several berry species. Notably, piceatannol is also a metabolite of resveratrol — the enzyme CYP1B1 (which is overexpressed in many tumor cells) hydroxylates resveratrol to produce piceatannol, suggesting that some of resveratrol's attributed anticancer effects may actually be mediated by its piceatannol metabolite.
Piceatannol exerts anti-inflammatory effects through multiple mechanisms: it inhibits NF-kB activation, suppresses COX-2 expression, blocks Syk kinase signaling (a key mediator in immune cell activation), and attenuates JAK1/STAT3 pathways. These properties have been studied in models of allergic inflammation, autoimmunity, and metabolic syndrome. Metabolically, piceatannol activates AMPK and SIRT1 — the same longevity-associated pathways targeted by resveratrol — but with greater potency in several cell-based assays. Animal studies have demonstrated anti-obesity effects, including reduced adipogenesis through inhibition of the insulin receptor/insulin receptor substrate-1 pathway during adipocyte differentiation, improved insulin sensitivity, and decreased hepatic lipid accumulation.
Compared to resveratrol, piceatannol shows improved oral bioavailability due to greater resistance to rapid glucuronidation and sulfation metabolism. Clinical data remains limited but growing — a randomized controlled trial in overweight men found that passion fruit seed extract (standardized for piceatannol) improved markers of endothelial function and reduced serum uric acid. The compound is also being investigated for skin health benefits, including photoprotection and anti-melanogenic activity, paralleling the topical use of resveratrol and pterostilbene. Piceatannol's antioxidant profile complements other polyphenolic compounds such as fisetin, quercetin, and trans-resveratrol.
Mechanism of Action
Piceatannol (3,3',4,5'-tetrahydroxy-trans-stilbene) is a naturally occurring polyphenolic stilbene and a metabolite of resveratrol, distinguished by an additional hydroxyl group that enhances its biological activity. Its best-characterized molecular target is spleen tyrosine kinase (Syk), a non-receptor protein tyrosine kinase critical for immune cell receptor signaling. By directly inhibiting Syk autophosphorylation and kinase activity, piceatannol disrupts downstream signaling through pathways including PLC-gamma, PI3K, and NF-kB, leading to suppression of inflammatory cytokine production, reduced immune cell activation, and inhibition of osteoclastogenesis.
In cancer cells, piceatannol induces apoptosis through both intrinsic and extrinsic pathways. It activates caspases-3, -8, and -9, upregulates pro-apoptotic proteins (Bax, Bad), and downregulates anti-apoptotic proteins (Bcl-2, Bcl-xL). It also inhibits the JAK1/STAT3 signaling axis, reducing expression of survival genes such as cyclin D1, survivin, and VEGF. Additionally, piceatannol suppresses matrix metalloproteinase (MMP) expression and inhibits cancer cell migration and invasion, partly through targeting the Wnt/beta-catenin pathway.
As an antioxidant, piceatannol scavenges reactive oxygen species (ROS) and upregulates endogenous antioxidant enzymes through Nrf2/ARE pathway activation. It also demonstrates anti-inflammatory effects by inhibiting COX-2 and iNOS expression via NF-kB suppression, and by reducing production of pro-inflammatory mediators including TNF-alpha, IL-1beta, and IL-6. In fibroblast-like synoviocytes relevant to rheumatoid arthritis, piceatannol's Syk inhibition reduces TNF-alpha-induced inflammatory responses.
Research
Reported Effects
Mechanism Similarity:: Works through similar pathways as well-studied compounds like l-citrulline and l-arginine, which show consistent benefits in research. Resveratrol Analog:: As a hydroxylated analog of resveratrol, may offer similar or enhanced benefits with different bioavailability profile. Limited Human Data:: While mechanism is well-established, direct human studies on piceatannol specifically are limited compared to related compounds. Synergistic Potential:: May work well in combination with other nitric oxide precursors based on similar compound research
- Works through similar pathways as well-studied compounds like l-citrulline and l-arginine, which show consistent benefits in research
- As a hydroxylated analog of resveratrol, may offer similar or enhanced benefits with different bioavailability profile
- While mechanism is well-established, direct human studies on piceatannol specifically are limited compared to related compounds
- May work well in combination with other nitric oxide precursors based on similar compound research
Safety Profile
Piceatannol is generally well-tolerated with few reported side effects at typical doses. It may have anti-platelet and blood-thinning properties, so individuals on anticoagulant therapy should exercise caution. Long-term safety data in humans is limited, and it should be avoided during pregnancy and breastfeeding.
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Specific piceatannol dosing guidelines for humans are not well-established in current research
Molecular Structure
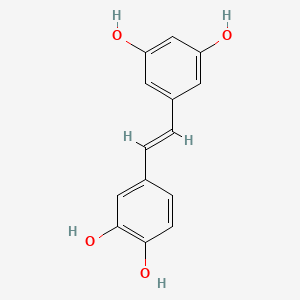
- Formula
- C14H12O4
- Weight
- 244.24 Da
- PubChem CID
- 667639
- Exact Mass
- 244.0736 Da
- LogP
- 2.9
- TPSA
- 80.9 Ų
- H-Bond Donors
- 4
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 2
- Complexity
- 282
Identifiers (SMILES, InChI)
InChI=1S/C14H12O4/c15-11-5-10(6-12(16)8-11)2-1-9-3-4-13(17)14(18)7-9/h1-8,15-18H/b2-1+
CDRPUGZCRXZLFL-OWOJBTEDSA-NSafety Profile
Common Side Effects
- Minimal Direct Reports:: Limited user experience data specific to piceatannol makes side effect profile unclear
- Related Compound Effects:: L-arginine can cause gastrointestinal discomfort at higher doses; piceatannol may have similar potential
- Blood Pressure Effects:: Compounds affecting nitric oxide production can lower blood pressure, requiring monitoring in hypotensive individuals
- Unknown Long-term Safety:: Lack of extensive human trials means long-term safety profile remains to be established
References (2)
- [1]Piceatannol, a natural hydroxylated analog of resveratrol, promotes nitric oxide release through phosphorylation of endothelial nitric oxide synthase in human endothelial cells
→ Study demonstrates that piceatannol enhances nitric oxide production by activating endothelial nitric oxide synthase, suggesting cardiovascular benefits similar to or potentially exceeding those of resveratrol.
- [2]l-Citrulline Supplementation: Impact on Cardiometabolic Health
→ Research shows l-citrulline supplementation mildly reduces blood pressure and improves cardiovascular markers through enhanced arginine availability and nitric oxide production, relevant to piceatannol's mechanism of action.
Picamilon
Picamilon is a synthetic compound combining GABA (gamma-aminobutyric acid) and niacin (vitamin B3) that was developed in Russia as a nootropic and anxiolytic dr
Pinealon
Pinealon is a short synthetic tetrapeptide (Ala-Glu-Asp-Gly) developed by the St. Petersburg Institute of Bioregulation and Gerontology as part of a class of pe