Artemisinin
A sesquiterpene lactone derived from sweet wormwood (Artemisia annua) that is the foundation of modern antimalarial therapy, recognized with the 2015 Nobel Prize in Physiology or Medicine.
Overview
Artemisinin is a sesquiterpene lactone compound (C₁₅H₂₂O₅) isolated from the plant Artemisia annua (sweet wormwood), a herb long used in Traditional Chinese Medicine known as qinghao. Its discovery in 1972 by Chinese pharmaceutical chemist Tu Youyou — as part of the secret military research initiative "Project 523" — revolutionized the treatment of malaria and earned her a share of the 2015 Nobel Prize in Physiology or Medicine. The key structural feature of artemisinin is its unusual endoperoxide bridge (1,2,4-trioxane ring), which is essential for its antimalarial activity and distinguishes it from all other classes of antimalarial drugs.
The antimalarial mechanism centers on the endoperoxide bridge, which is activated by heme iron released during hemoglobin digestion within the Plasmodium parasite. This activation generates reactive oxygen species and carbon-centered free radicals that damage parasite proteins, lipids, and membranes, rapidly killing the organism. Artemisinin and its semisynthetic derivatives — dihydroartemisinin, artemether, and artesunate — form the basis of artemisinin-based combination therapies (ACTs), which the World Health Organization recommends as the first-line treatment for Plasmodium falciparum malaria worldwide. These compounds are valued for their rapid onset of action, broad-stage parasite killing, and relatively low toxicity.
A major concern in global malaria control is the emergence of artemisinin resistance, primarily associated with mutations in the Plasmodium falciparum Kelch13 protein, first identified in Southeast Asia and now spreading to other regions. Beyond antimalarial applications, artemisinin derivatives have shown promising pharmacological activities in preclinical research, including anticancer effects through induction of apoptosis and ferroptosis, anti-inflammatory properties, and antiviral activity. Ongoing research continues to explore novel therapeutic applications and strategies to overcome emerging drug resistance.
Mechanism of Action
Endoperoxide Bridge Activation
Artemisinin is a sesquiterpene lactone derived from Artemisia annua containing a unique 1,2,4-trioxane endoperoxide bridge essential for its antimalarial activity. Inside the parasitized erythrocyte, ferrous iron (Fe²⁺) from heme — liberated during hemoglobin digestion by Plasmodium — cleaves the endoperoxide bridge via reductive scission, generating cytotoxic carbon-centered free radicals and reactive oxygen species (ROS) (PMID: 15518585).
Heme Alkylation & Protein Damage
The carbon radicals produced by endoperoxide cleavage alkylate heme and parasite proteins, disrupting essential cellular processes. Key targets include the SERCA-type calcium ATPase (PfATP6), disrupting calcium homeostasis in the parasite's endoplasmic reticulum, and phosphatidylinositol 3-kinase (PfPI3K), impairing vesicular trafficking and lipid metabolism (PMID: 12853456).
Mitochondrial & Membrane Disruption
Artemisinin-derived radicals also attack parasite mitochondrial electron transport chain components, collapsing the mitochondrial membrane potential and triggering oxidative damage to lipid membranes. This multi-target oxidative assault results in rapid parasiticidal activity across all intraerythrocytic stages, including the ring stage, which is critical for preventing progression to severe malaria (PMID: 16101480).
Anti-Cancer Mechanisms
Beyond antimalarial activity, artemisinin shows selective cytotoxicity toward iron-loaded cancer cells through the same iron-dependent radical generation. It inhibits angiogenesis via downregulation of VEGF and disrupts the NF-kB and Wnt/beta-catenin pathways, inducing apoptosis and cell cycle arrest in various tumor models (PMID: 22185819).
Anti-Inflammatory Properties
Artemisinin suppresses NLRP3 inflammasome activation and inhibits NF-kB-mediated transcription of TNF-alpha, IL-6, and IL-1beta, contributing to anti-inflammatory effects investigated in autoimmune conditions.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Limited Standalone Data:: Most user reports involve artemisinin in combination products rather than as a single supplement. Research-Practice Gap:: Strong scientific evidence for antimalarial and potential anticancer effects, but minimal consumer-level experience reports. Dosage Uncertainty:: Users express concern about appropriate dosing, particularly with liposomal formulations and combinations. Bioavailability Considerations:: Research suggests whole plant extracts may offer better absorption than isolated compounds
- Most user reports involve artemisinin in combination products rather than as a single supplement
- Strong scientific evidence for antimalarial and potential anticancer effects, but minimal consumer-level experience reports
- Users express concern about appropriate dosing, particularly with liposomal formulations and combinations
- Research suggests whole plant extracts may offer better absorption than isolated compounds
Safety Profile
Common Side Effects
- Gastrointestinal: Nausea, vomiting, abdominal pain, diarrhea (very common, dose-related)
- Dizziness and headache
- Tinnitus (ringing in the ears)
- Pruritus (itching)
- Fatigue and asthenia
- Dark urine discoloration (harmless)
Serious Adverse Reactions
- Neurotoxicity: Artemisinin and its derivatives can cause cerebellar and brainstem toxicity at high or prolonged doses. Symptoms include ataxia, slurred speech, hearing loss, and nystagmus. This is well-documented in animal studies and case reports in humans with non-malarial (off-label) use.
- QT prolongation: Artemisinin combination therapies (ACTs) have been associated with cardiac conduction changes. ECG monitoring recommended per WHO guidelines.
- Hepatotoxicity: Elevated transaminases reported; rare cases of drug-induced liver injury.
- Hemolytic anemia: Can trigger hemolysis in G6PD-deficient patients.
- Type I hypersensitivity: Anaphylaxis reported rarely.
Contraindications
- Known hypersensitivity to artemisinin or sesquiterpene lactones
- First trimester of pregnancy (embryotoxic and teratogenic in animal studies; WHO advises avoidance in first trimester unless no alternative exists for severe malaria)
- Severe hepatic impairment
- G6PD deficiency (increased hemolysis risk)
- Cardiac conduction disorders (long QT syndrome)
Drug Interactions
- CYP2B6 and CYP3A4: Artemisinin is a potent auto-inducer of its own metabolism and induces CYP2B6/3A4. May reduce levels of co-administered drugs metabolized by these enzymes.
- Anticoagulants (warfarin): May alter INR; mechanism unclear
- QT-prolonging agents: Additive risk of arrhythmia
- Antiretrovirals (efavirenz, nevirapine): Complex CYP interactions; may reduce antiretroviral efficacy
- Immunosuppressants: May alter immune function; use with caution post-transplant
- Iron supplements: Artemisinin generates reactive oxygen species via iron-mediated Fenton reaction; theoretical potentiation of both efficacy and toxicity
Special Populations
- Pediatric: WHO-approved ACTs are used in pediatric malaria; supplement-grade artemisinin is not standardized for children
- Pregnancy: Contraindicated in first trimester; second/third trimester use only for malaria under medical supervision
- Geriatric: Increased susceptibility to neurotoxicity and hepatotoxicity
- Renal impairment: Limited data; dose adjustment may be needed
Monitoring
- CBC with reticulocyte count (hemolysis surveillance)
- Liver function tests (baseline and periodic)
- ECG (QTc interval) before and during therapy
- Neurological assessment with prolonged or high-dose use
- G6PD screening before initiation
Pharmacokinetic Profile
Artemisinin — Pharmacokinetic Curve
SubcutaneousMolecular Structure
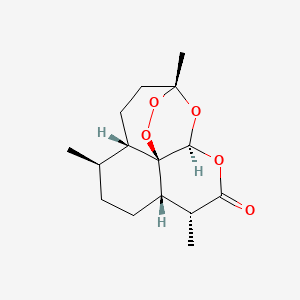
- Formula
- C15H22O5
- Weight
- 282.33 Da
- PubChem CID
- 68827
- Exact Mass
- 282.1467 Da
- LogP
- 2.8
- TPSA
- 54 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 0
- Complexity
- 452
Identifiers (SMILES, InChI)
InChI=1S/C15H22O5/c1-8-4-5-11-9(2)12(16)17-13-15(11)10(8)6-7-14(3,18-13)19-20-15/h8-11,13H,4-7H2,1-3H3/t8-,9-,10+,11+,13-,14-,15-/m1/s1
BLUAFEHZUWYNDE-NNWCWBAJSA-NSafety Profile
Common Side Effects
- Minimal User Reports:: Very few direct side effect reports in consumer forums, possibly due to limited standalone use
- Combination Concerns:: Users worry about interactions when stacking multiple liposomal antioxidants including artemisinin
- Safety Profile:: Generally considered safe based on long antimalarial use history, though long-term supplementation data is limited
- Antioxidant Overload:: Theoretical concerns about excessive antioxidant intake when combined with multiple similar compounds
References (5)
- [1]Artemisinin and Its Derivatives from Molecular Mechanisms to Clinical Applications: New Horizons Beyond Antimalarials
→ Comprehensive review demonstrating artemisinin's antitumor effects through oxidative stress induction, cell cycle arrest, and apoptosis triggering, plus immunomodulatory and metabolic regulation properties beyond antimalarial use.
- [3]Medicinal and mechanistic overview of artemisinin in the treatment of human diseases
→ Analysis of artemisinin's therapeutic potential for malaria, cancer, viral infections, bacterial infections, and cardiovascular diseases, with detailed pharmacokinetic and pharmacological properties based on in vitro and in vivo studies.
- [2]Artemisinin and its derivatives throughout the therapeutic mechanisms and clinical potential
→ Review highlighting artemisinin's broad pharmacological activities including anticancer, antiviral, anti-inflammatory, and renoprotective properties, with applications extending to metabolic syndrome, fibrosis, and SARS-CoV-2 treatment.
- [4]Artemisinin ameliorates cognitive decline by inhibiting hippocampal neuronal ferroptosis via Nrf2 activation in T2DM mice
→ Study demonstrating artemisinin (40 mg/kg) reversed cognitive impairments in diabetic mice by activating Nrf2 pathways to inhibit neuronal ferroptosis and reduce oxidative stress in the hippocampus.
- [5]Dried Leaf Artemisia Annua Improves Bioavailability of Artemisinin via Cytochrome P450 Inhibition
→ Research showing whole dried leaf Artemisia annua improves artemisinin bioavailability through cytochrome P450 enzyme inhibition, potentially enhancing therapeutic efficacy compared to isolated artemisinin.
Armodafinil
Armodafinil is the R-enantiomer of modafinil, offering longer-lasting wakefulness promotion with potentially smoother effects. It's FDA-approved for narcolepsy,
Artichoke Extract
Artichoke Extract, derived from Cynara scolymus leaves, is a supplement rich in polyphenolic compounds, particularly chlorogenic acid and cynarin. It primarily