Everolimus
Everolimus is a semi-synthetic rapamycin derivative (rapalog) that inhibits the mTORC1 signaling pathway, approved for use as an immunosuppressant in organ transplantation and as an antineoplastic agent in multiple cancers, with growing research interest in its anti-aging properties.
Overview
Everolimus (trade names Afinitor, Zortress, Certican) is a 40-O-(2-hydroxyethyl) derivative of rapamycin (sirolimus) developed by Novartis to improve the oral bioavailability and pharmacokinetic profile of the parent macrolide. Like sirolimus, everolimus forms a complex with the intracellular immunophilin FKBP12, and this complex allosterically inhibits the mechanistic target of rapamycin complex 1 (mTORC1), a serine/threonine kinase that serves as a master regulator of cell growth, proliferation, metabolism, and autophagy.
By inhibiting mTORC1, everolimus suppresses phosphorylation of key downstream effectors including S6 kinase 1 (S6K1) and 4E-BP1, resulting in reduced protein synthesis, cell cycle arrest (primarily in G1 phase), and induction of autophagy. In oncology, everolimus is FDA-approved for advanced renal cell carcinoma, progressive neuroendocrine tumors of pancreatic origin, hormone receptor-positive HER2-negative breast cancer (in combination with exemestane), tuberous sclerosis complex-associated renal angiomyolipomas and subependymal giant cell astrocytomas, and certain forms of transplant rejection prophylaxis. Its anti-angiogenic effects, mediated through reduced VEGF production downstream of mTORC1, contribute to its antitumor activity.
The intersection of mTOR inhibition and aging biology has generated substantial interest in everolimus as a potential geroprotective agent. A landmark 2014 study by Mannick et al. demonstrated that low-dose everolimus enhanced immune function in elderly subjects by approximately 20% improvement in influenza vaccine response. Subsequent research has explored mTOR inhibition for age-related decline in immune function (immunosenescence), neurodegeneration, and cardiovascular aging. The key challenge remains achieving the anti-aging benefits of partial mTORC1 inhibition while avoiding the immunosuppressive and metabolic side effects (hyperglycemia, dyslipidemia, stomatitis) associated with higher doses used in oncology and transplantation.
Mechanism of Action
mTORC1 Inhibition
Everolimus is a 40-O-(2-hydroxyethyl) derivative of rapamycin (sirolimus) that functions as an allosteric inhibitor of mechanistic target of rapamycin complex 1 (mTORC1). It binds the intracellular immunophilin FKBP12 (FK506-binding protein 12), forming a binary complex that associates with the FRB domain of mTOR, preventing mTORC1 from phosphorylating its key substrates p70S6 kinase 1 (S6K1) and eukaryotic translation initiation factor 4E-binding protein 1 (4E-BP1) (PMID: 19461509).
Downstream Translational Control
Inhibition of S6K1 reduces phosphorylation of ribosomal protein S6, decreasing translation of mRNAs containing 5' terminal oligopyrimidine (5'TOP) tracts — these encode ribosomal proteins and elongation factors essential for protein synthesis capacity. Simultaneously, hypophosphorylated 4E-BP1 sequesters eIF4E, blocking cap-dependent translation initiation of oncogenic mRNAs including cyclin D1, c-Myc, HIF-1alpha, and VEGF (PMID: 22203767).
Cell Cycle Arrest & Autophagy
By suppressing mTORC1 signaling, everolimus induces G1 cell cycle arrest through downregulation of cyclin D1 and upregulation of p27Kip1. It simultaneously activates autophagy via de-repression of the ULK1/ATG13/FIP200 complex, which mTORC1 normally phosphorylates to suppress autophagosome formation. This dual mechanism — proliferative arrest plus enhanced autophagy — underpins both its antitumor and immunosuppressive activities (PMID: 25484100).
Anti-Angiogenic & Immunosuppressive Effects
Everolimus reduces VEGF secretion from tumor cells by suppressing HIF-1alpha translation, impairing tumor angiogenesis. In T lymphocytes, it blocks IL-2-driven proliferation by preventing mTORC1-mediated metabolic reprogramming from oxidative phosphorylation to glycolysis, a switch required for clonal expansion. This selective immunosuppression underlies its use in transplant medicine (PMID: 24803579).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Sustained Efficacy:: Long-term clinical data shows that beneficial effects on tumor reduction and lesion control are maintained over 4+ years.. Bioavailability:: Evidence suggests everolimus has better oral bioavailability and a shorter half-life than sirolimus, allowing for more precise dosing.. Geroprotection:: Research indicates it is a leading pharmacological agent for extending maximal lifespan and improving healthspan in mammalian models.
- Long-term clinical data shows that beneficial effects on tumor reduction and lesion control are maintained over 4+ years.
- Evidence suggests everolimus has better oral bioavailability and a shorter half-life than sirolimus, allowing for more precise dosing.
- Research indicates it is a leading pharmacological agent for extending maximal lifespan and improving healthspan in mammalian models.
Safety Profile
Safety Profile: Everolimus (FDA Approved)
Overview Everolimus (brand names Afinitor, Zortress, Certican) is an FDA-approved mTOR (mechanistic target of rapamycin) inhibitor used for multiple indications including advanced renal cell carcinoma, breast cancer, neuroendocrine tumors, tuberous sclerosis complex, and organ transplant rejection prophylaxis. It is a potent immunosuppressive and antiproliferative agent with a significant side effect profile requiring close medical monitoring.
Common Side Effects
- Stomatitis/oral mucositis (44-78%): the most frequent adverse effect, often dose-limiting; managed with dexamethasone mouthwash
- Infections (20-50%): including upper respiratory infections, urinary tract infections, and pneumonia due to immunosuppression
- Rash and skin toxicity (25-59%): acneiform rash, pruritus, dry skin
- Fatigue and asthenia (20-45%)
- Diarrhea (19-34%), nausea (16-29%), and decreased appetite
- Peripheral edema (20-45%)
- Hyperglycemia (12-57%) and hyperlipidemia (hypercholesterolemia 30-77%, hypertriglyceridemia 27-73%)
Serious Adverse Effects
- Non-infectious pneumonitis (12-19%): potentially fatal interstitial lung disease; monitor for new or worsening respiratory symptoms; may require dose reduction or discontinuation
- Severe immunosuppression: increased risk of opportunistic infections including Pneumocystis jirovecii, BK virus, CMV, and invasive fungal infections
- Hepatotoxicity: elevated transaminases, hepatic failure reported rarely
- Renal toxicity: proteinuria, increased serum creatinine; particularly when combined with calcineurin inhibitors
- Thrombotic microangiopathy/hemolytic uremic syndrome: reported in transplant patients
- Impaired wound healing: significant concern post-surgery
- Myelosuppression: anemia (45-92%), lymphopenia, thrombocytopenia, neutropenia
- Angioedema: particularly when co-administered with ACE inhibitors
Contraindications
- Known hypersensitivity to everolimus, sirolimus, or other rapamycin derivatives
- Severe hepatic impairment (Child-Pugh C) for oncology indications
- Severe uncontrolled infection
- Live vaccine administration during treatment
- Pregnancy (Category D): causes fetal harm; effective contraception required during and for 8 weeks after treatment
Drug Interactions
- Strong CYP3A4 inhibitors (ketoconazole, itraconazole, clarithromycin, ritonavir): dramatically increase everolimus levels — contraindicated or require significant dose reduction
- Strong CYP3A4 inducers (rifampin, phenytoin, carbamazepine, St. John's Wort): substantially reduce everolimus levels, potentially rendering treatment ineffective
- Calcineurin inhibitors (cyclosporine, tacrolimus): complex interaction in transplant settings; cyclosporine increases everolimus levels
- ACE inhibitors: increased angioedema risk
- Live vaccines: contraindicated due to immunosuppression; inactivated vaccines may have reduced efficacy
- Grapefruit and grapefruit juice: CYP3A4 inhibition increases everolimus exposure
Special Populations
- Hepatic impairment: dose reduction required for mild (Child-Pugh A) and moderate (Child-Pugh B) impairment
- Renal impairment: no dose adjustment needed but monitor closely
- Elderly: no specific dose adjustment; increased susceptibility to side effects
- Fertility: may cause male and female infertility; spermatogenesis may be affected
Dosage Considerations
- Oncology: 10 mg once daily (dose reductions to 5 mg or 2.5 mg for toxicity)
- Transplant: 0.75 mg twice daily initially, adjusted by therapeutic drug monitoring (target trough 3-8 ng/mL)
- Therapeutic drug monitoring is essential for transplant indications
- Take consistently with or without food at the same time daily
Pharmacokinetic Profile
Everolimus — Pharmacokinetic Curve
SubcutaneousMolecular Structure
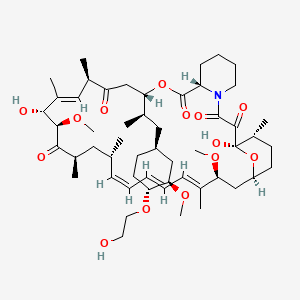
- Formula
- C53H83NO14
- Weight
- 958.2 Da
- PubChem CID
- 6442177
- Exact Mass
- 957.5814 Da
- LogP
- 5.9
- TPSA
- 205 Ų
- H-Bond Donors
- 3
- H-Bond Acceptors
- 14
- Rotatable Bonds
- 9
- Complexity
- 1810
Identifiers (SMILES, InChI)
InChI=1S/C53H83NO14/c1-32-16-12-11-13-17-33(2)44(63-8)30-40-21-19-38(7)53(62,68-40)50(59)51(60)54-23-15-14-18-41(54)52(61)67-45(35(4)28-39-20-22-43(66-25-24-55)46(29-39)64-9)31-42(56)34(3)27-37(6)48(58)49(65-10)47(57)36(5)26-32/h11-13,16-17,27,32,34-36,38-41,43-46,48-49,55,58,62H,14-15,18-26,28-31H2,1-10H3/b13-11+,16-12+,33-17+,37-27+/t32-,34-,35-,36-,38-,39+,40+,41+,43-,44+,45+,46-,48-,49+,53-/m1/s1
HKVAMNSJSFKALM-GKUWKFKPSA-NSafety Profile
Common Side Effects
- Stomatitis:: Inflammatory sores in the mouth are the most frequently reported side effect, occurring in over 40% of patients.
- Mouth Ulceration:: Small, painful ulcers that typically decrease in frequency as the body adapts to the compound.
- Dyslipidemia:: Potential for increased blood lipid and glucose levels, requiring periodic metabolic panels.
- Dermatological Issues:: While it can clear lesions, some users experience acne-like rashes or thin skin.
References (1)
- [1]Long-Term Use of Everolimus in Patients with Tuberous Sclerosis Complex: Final Results from the EXIST-1 Study
→ This study demonstrated that long-term everolimus use leads to sustained reductions in tumor volume and safely manages multi-system manifestations over several years.
Etiracetam
Etiracetam is a pyrrolidinone derivative and an analog of piracetam that functions as a nootropic agent and seizure medication. It is primarily recognized for i
Evodiamine
Evodiamine is a bioactive alkaloid extracted from Evodia rutaecarpa (Wu-Zhu-Yu), a traditional Chinese medicinal fruit. It acts as a vanilloid receptor agonist