Bilobalide
Bilobalide is a sesquiterpene trilactone found exclusively in Ginkgo biloba leaves, representing one of the key bioactive terpene constituents of standardized ginkgo extracts. It exhibits neuroprotective properties and contributes to the cognitive and circulatory benefits attributed to ginkgo supplementation.
Overview
Bilobalide is a unique sesquiterpene lactone isolated from the leaves of Ginkgo biloba, one of the oldest living tree species and a widely used botanical in traditional and modern medicine. Along with the ginkgolides (A, B, C, and J), bilobalide belongs to the terpene trilactone class of compounds that are considered the principal active constituents of standardized Ginkgo biloba extracts such as EGb 761. Bilobalide typically constitutes approximately 2.9–3.2% of standardized ginkgo leaf extracts.
Preclinical research has identified several pharmacological activities of bilobalide, including neuroprotection, anti-inflammatory effects, and modulation of neurotransmitter systems. It has been shown to protect neurons against ischemic and excitotoxic damage, potentially through preservation of mitochondrial function, reduction of glutamate-induced toxicity, and inhibition of apoptotic pathways. Bilobalide also appears to influence GABAergic neurotransmission and may contribute to the anxiolytic effects observed with ginkgo extract administration.
In addition to its central nervous system effects, bilobalide has demonstrated the ability to promote peripheral circulation and reduce edema. It is also being investigated for its potential roles in hepatoprotection and regulation of gene expression related to cholesterol metabolism. While bilobalide is not typically available as an isolated supplement, its presence in standardized ginkgo extracts is considered important for the overall pharmacological profile of these preparations, and quality control standards for ginkgo products often specify minimum terpene trilactone content.
Mechanism of Action
Sesquiterpene Trilactone from Ginkgo biloba
Bilobalide is a unique sesquiterpene trilactone (C15 terpenoid with three lactone rings) found exclusively in Ginkgo biloba leaves, constituting approximately 3% of standardized extract (EGb 761). Its distinctive cage-like molecular structure contains a tert-butyl group and a highly oxygenated skeleton that enables interactions with multiple neuronal targets (PMID: 12587690).
GABAergic Modulation
Bilobalide acts as a non-competitive antagonist at GABA_A receptors, binding at a site overlapping the picrotoxin binding site within the chloride ion channel pore (at or near the 2' position of the TM2 domain). This reduces GABA-mediated chloride conductance and neuronal inhibition, producing a mild pro-cognitive alerting effect. The IC50 for GABA_A channel blockade is approximately 4.6 microM, suggesting moderate affinity at physiologically relevant concentrations (PMID: 11353086).
Mitochondrial Protection & Anti-Apoptotic Effects
Bilobalide preserves mitochondrial membrane potential during ischemic and excitotoxic insults by stabilizing the mitochondrial permeability transition pore (mPTP) in the closed conformation. It maintains cytochrome c within the intermembrane space, preventing caspase-9/caspase-3 activation and intrinsic apoptotic cascading. In experimental stroke models, bilobalide reduces infarct volume by 30-50% when administered before or shortly after ischemic onset (PMID: 16480697).
Glutamate Excitotoxicity Protection
Bilobalide suppresses ischemia-induced glutamate release by inhibiting presynaptic glutamate exocytosis and enhancing astrocytic glutamate uptake via upregulation of EAAT1/GLAST and EAAT2/GLT-1 transporters. It also attenuates NMDA receptor-mediated calcium influx, reducing downstream excitotoxic cascades including calpain activation, phospholipase A2 release, and nitrosative stress (PMID: 14624473).
Neuroplasticity & Gene Expression
Bilobalide upregulates expression of BDNF and GAP-43 (growth-associated protein 43) in hippocampal neurons, promoting neurite outgrowth, synaptic remodeling, and neurogenesis in the dentate gyrus.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Dementia Treatment:: Clinical trials show significant improvements in SKT cognitive scores and NPI neuropsychiatric scores compared to placebo over 24 weeks. Dosage Response:: 240mg daily dose of EGb 761 extract demonstrated consistent efficacy across multiple trials for mild-to-moderate dementia. Time Frame:: Benefits typically observed within 24 weeks of treatment, with improvements in cognition, behavior, and functional measures. Standardization Matters:: Effectiveness depends on proper standardization to ginkgo flavonol glycosides, terpene lactones (including bilobalide), and kaempferol content
- Clinical trials show significant improvements in SKT cognitive scores and NPI neuropsychiatric scores compared to placebo over 24 weeks
- 240mg daily dose of EGb 761 extract demonstrated consistent efficacy across multiple trials for mild-to-moderate dementia
- Benefits typically observed within 24 weeks of treatment, with improvements in cognition, behavior, and functional measures
- Effectiveness depends on proper standardization to ginkgo flavonol glycosides, terpene lactones (including bilobalide), and kaempferol content
Safety Profile
Common Side Effects
- Generally well-tolerated as a component of standardized Ginkgo biloba extracts (EGb 761)
- Mild gastrointestinal upset including nausea, stomach discomfort, and diarrhea
- Headache and dizziness at higher doses
- Allergic skin reactions (rare)
- Restlessness or mild insomnia if taken late in the day
Serious Adverse Effects
- Increased bleeding risk, particularly at high doses or in combination with anticoagulants
- Seizures have been reported in isolated cases, potentially related to bilobalide and ginkgotoxin content in non-standardized preparations
- Severe allergic reactions including anaphylaxis (very rare)
- Stevens-Johnson syndrome has been reported in extremely rare cases with Ginkgo biloba products
- Liver toxicity has not been directly attributed to bilobalide but cannot be excluded with concentrated isolates
Contraindications
- Known allergy to Ginkgo biloba or any Ginkgoaceae family plants
- Active bleeding disorders or hemorrhagic stroke
- Pre-surgical patients should discontinue use at least 2 weeks before surgery due to antiplatelet effects
- Seizure disorders (epilepsy), as ginkgo-derived compounds may lower seizure threshold
- Should not be used concurrently with other known seizure-threshold-lowering agents
Drug Interactions
- Anticoagulants and antiplatelets (warfarin, aspirin, clopidogrel, heparin): significantly increased bleeding risk
- Anticonvulsants: bilobalide may alter seizure threshold and reduce efficacy of antiepileptic medications
- CYP enzyme interactions: Ginkgo components may modulate CYP3A4, CYP2C9, and CYP1A2 activity, potentially affecting metabolism of numerous drugs
- SSRIs and MAOIs: potential for serotonin syndrome-like effects when combined with ginkgo extracts
- NSAIDs: additive antiplatelet effects increasing bleeding risk
- Insulin and oral hypoglycemics: ginkgo may alter blood glucose levels
Special Populations
- Pregnant and breastfeeding women should avoid bilobalide/Ginkgo biloba due to insufficient safety data and potential antiplatelet effects
- Children: safety not established; use is not recommended
- Elderly patients are at greater risk of bleeding complications and should use with medical supervision
Pharmacokinetic Profile
Bilobalide — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- 240mg daily of standardized EGb 761 extract is the most researched and effective dose for dementia treatment
Molecular Structure
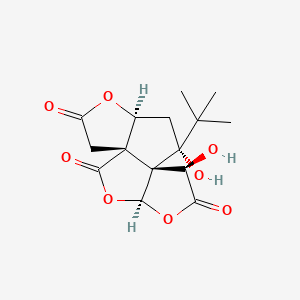
- Formula
- C15H18O8
- Weight
- 326.30 Da
- PubChem CID
- 73581
- Exact Mass
- 326.1002 Da
- LogP
- -0.3
- TPSA
- 119 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 8
- Rotatable Bonds
- 1
- Complexity
- 650
Identifiers (SMILES, InChI)
InChI=1S/C15H18O8/c1-12(2,3)14(20)4-6-13(5-7(16)21-6)10(19)23-11-15(13,14)8(17)9(18)22-11/h6,8,11,17,20H,4-5H2,1-3H3/t6-,8-,11-,13-,14+,15+/m0/s1
MOLPUWBMSBJXER-YDGSQGCISA-NSafety Profile
Common Side Effects
- Generally Well-Tolerated:: Clinical trials report good tolerability with minimal adverse effects at standard doses
- Quality Concerns:: No specific bilobalide side effects reported in Reddit data; user concerns focus on product quality and standardization
- Individual Variation:: Limited User feedback of personal experiences with bilobalide specifically, as most users discuss whole Ginkgo biloba extract
- Safety Profile:: Research demonstrates acceptable safety profile in elderly populations with dementia over extended treatment periods
References (4)
- [3]Ginkgo biloba for Preventing Cognitive Decline in Older Adults: A Randomized Trial
→ Large randomized trial examining Ginkgo biloba for preventing cognitive decline in older adults, providing evidence on its efficacy in dementia prevention and cognitive maintenance.
- [4]Ginkgo biloba extract EGb 761® in the symptomatic treatment of mild-to-moderate dementia: a profile of its use
→ Comprehensive profile review concluded that EGb 761 displays positive effects on cognitive outcomes, neuropsychiatric symptoms, and functional abilities in adults with mild-to-moderate dementia, with good tolerability and safety profile.
- [1]Efficacy and tolerability of Ginkgo biloba extract EGb 761® in dementia: a systematic review and meta-analysis of randomized placebo-controlled trials
→ Systematic review and meta-analysis found that EGb 761 treatment resulted in significant improvements in cognition, psychopathology, functional measures and quality of life in patients with mild-to-moderate dementia compared to placebo.
- [2]Ginkgo biloba extract EGb 761® in dementia with neuropsychiatric features: A randomised, placebo-controlled trial to confirm the efficacy and safety of a daily dose of 240 mg
→ 24-week trial with 410 outpatients demonstrated that 240mg daily dose of EGb 761 significantly improved cognition (SKT scores) and neuropsychiatric symptoms (NPI scores) in patients with mild-to-moderate dementia, with improvements in quality of life and functional measures.
Beta-Sitosterol
Beta-sitosterol is a plant-derived phytosterol (plant sterol) structurally similar to cholesterol, commonly found in saw palmetto extract and various plants. It
Bioglutide
Bioglutide (NA-931) is a first-in-class oral quadruple receptor agonist targeting IGF-1, GLP-1, GIP, and glucagon receptors simultaneously. Developed by Biom...