SS-31
SS-31 (elamipretide) is a mitochondria-targeted tetrapeptide that stabilizes cardiolipin to improve ATP production and reduce reactive oxygen species, with research applications in mitochondrial myopathy, heart failure, diabetes, and inflammatory diseases.
Overview
Cardiolipin is a phospholipid component of the inner mitochondrial membrane that serves as a fundamental scaffold for the electron transport chain. Dysfunction of cardiolipin has been implicated in the pathology of numerous diseases including Alzheimer's disease, Parkinson's disease, nonalcoholic fatty liver disease, diabetes, heart failure, and cancer. Mitochondrial myopathy, a group of neuromuscular disorders caused by mitochondrial damage, is characterized by muscle weakness, exercise intolerance, heart failure, seizures, and dementia. SS-31 was developed to directly target this mitochondrial dysfunction and has progressed through multiple phases of clinical trials.
Mechanism of Action
SS-31 concentrates in the inner mitochondrial membrane approximately 1000-5000 fold relative to extracellular concentration, driven by its interaction with cardiolipin. By binding cardiolipin, SS-31 stabilizes the cristae structure and optimizes the organization of electron transport chain supercomplexes, improving electron transfer efficiency and reducing electron leak that generates reactive oxygen species. Research by Birk et al. (2013) demonstrated that SS-31 re-energizes ischemic mitochondria specifically through cardiolipin interaction. Notably, Chatfield et al. (2019) found that SS-31 improves mitochondrial oxygen flux through a mechanism independent of cardiolipin restructuring, suggesting a second mechanism of action that remains under investigation.
Reconstitution Calculator
SS-31
SS-31 (elamipretide) is a small, aromatic tetrapeptide that readily penetrates c

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Heart Failure
SS-31 has shown compelling results in cardiac research. Chatfield et al. (2019) demonstrated significant improvements in mitochondrial oxygen flux and ATP production in failing human heart tissue treated with SS-31. Sabbah et al. (2016) showed that chronic treatment with SS-31 improved left ventricular function and mitochondrial respiration in dogs with advanced heart failure, suggesting potential as a long-term therapeutic for cardiac remodeling.
In the context of acute myocardial infarction, Hortmann et al. (2019) found that SS-31 reduced circulating HtrA2 levels (a marker of cardiomyocyte apoptosis) in patients with ST-elevation myocardial infarction, suggesting cardioprotective effects during acute ischemic events.
Diabetes and Metabolic Dysfunction
Escribano-Lopez et al. (2018) demonstrated that SS-31 increases SIRT1 levels and ameliorates inflammation, oxidative stress, and leukocyte-endothelium interactions in type 2 diabetes. SIRT1 elevation is associated with improved insulin sensitivity and reduced inflammation, suggesting SS-31 may address the mitochondrial component of diabetic microvascular disease.
Hou et al. (2018) showed that SS-31 downregulates CD36, reduces activated MnSOD, suppresses NADPH oxidase function, and inhibits NF-kappaB p65 in diabetic nephropathy models, improving renal function through antioxidant mechanisms.
Anti-Inflammatory Effects
SS-31 demonstrates broad anti-inflammatory activity through mitochondrial stabilization. Mo et al. (2019) showed that SS-31 reduces inflammation and oxidative stress by inhibiting FIS1 expression in lipopolysaccharide-stimulated microglia. FIS1 is a mitochondrial fission protein elevated in neurodegenerative diseases and cancers, indicating dysfunctional mitochondrial division. By preventing inflammasome activation, SS-31 maintains normal mitochondrial function rather than allowing the shift from ATP production to primarily ROS generation.
Mitochondrial Disease
The first evidence that SS-31 could restore energy production in primary mitochondrial diseases came from animal studies. Szeto et al. (2011) demonstrated in rats with ischemia-reperfusion kidney injury that SS-31 protected kidney structure, accelerated ATP recovery, and reduced cell necrosis. Subsequent studies showed the peptide could reduce symptoms of mitochondrial disease regardless of etiology, including age-related mitochondrial dysfunction. Siegel et al. (2013) showed that SS-31 rapidly improves mitochondrial energetics and skeletal muscle performance in aged mice, supporting its potential in age-related mitochondrial decline.
In phase II human trials, Karaa et al. (2018) demonstrated that SS-31 increased exercise performance after just 5 days of treatment in patients with primary mitochondrial myopathy, with no safety concerns or prominent side effects. Phase III trials did not meet their primary endpoints, though researchers have suggested this may reflect inappropriate endpoint selection rather than lack of efficacy. Subsequent trials with refined endpoints and SS-31 derivatives are in progress.
Safety Profile
SS-31 has been evaluated in multiple phase I, II, and III clinical trials and is considered safe for human use. In phase II trials, no significant safety concerns or prominent side effects were observed at therapeutic doses. The most commonly reported adverse effect is injection site reaction. The peptide has been granted orphan drug status by the FDA for primary mitochondrial myopathy and Barth syndrome. SS-31 can be prescribed under compassionate use exceptions for patients with no other treatment options. Its targeted accumulation in mitochondrial membranes limits off-target effects, and its mechanism of stabilizing (rather than disrupting) existing mitochondrial structures provides an inherent safety advantage.
Phase I Dose-Escalation Studies
Initial human pharmacokinetic studies evaluated single intravenous doses of SS-31 (elamipretide) at 0.005, 0.01, 0.025, 0.05, and 0.25 mg/kg in healthy volunteers. The peptide demonstrated linear pharmacokinetics across this dose range with no dose-limiting toxicities. Subcutaneous administration was subsequently evaluated at 4 mg and 40 mg single doses (Szeto, 2014).
Phase II - Primary Mitochondrial Myopathy (MMPOWER)
Karaa et al. (2018) conducted a randomized, double-blind, dose-escalation trial (MMPOWER) in 36 adults with genetically confirmed primary mitochondrial myopathy. Patients received subcutaneous elamipretide at 0.01, 0.1, or 0.25 mg/kg once daily for 5 consecutive days. The 0.25 mg/kg dose produced statistically significant improvements in 6-minute walk test (6MWT) distance (+64.5 meters, p<0.05). No serious adverse events were reported; injection site reactions were the most common side effect.
Phase II - Heart Failure (PROGRESS-HF)
Patients with stable heart failure with reduced ejection fraction (HFrEF, LVEF <=40%) received elamipretide 4 mg subcutaneous daily for 4 weeks in the PROGRESS-HF trial. While the primary endpoint (change in LVESV by cardiac MRI) did not reach statistical significance, secondary analyses showed trends toward improved diastolic function and reduced NT-proBNP levels.
Phase III - Barth Syndrome (TAZPOWER)
Steele et al. (2022) reported results from TAZPOWER, a pivotal trial in Barth syndrome (a genetic cardiomyopathy caused by tafazzin mutations affecting cardiolipin remodeling). Patients received elamipretide 40 mg subcutaneous daily for 12 weeks. While the primary endpoint (6MWT) did not reach statistical significance (p=0.15), significant improvements were observed in the Barth Syndrome Symptom Assessment scale. An open-label extension showed sustained benefits over 168 weeks.
Phase III - Primary Mitochondrial Myopathy (MMPOWER-3)
The phase III MMPOWER-3 trial enrolled 218 patients with primary mitochondrial myopathy randomized to elamipretide 40 mg subcutaneous daily or placebo for 24 weeks. The trial did not meet its co-primary endpoints of change in 6MWT distance and Total Fatigue Score on the Primary Mitochondrial Myopathy Symptom Assessment (PMMSA). Post-hoc analyses suggested that endpoint selection may have been suboptimal for capturing SS-31's effects, which may be more reflected in quality-of-life measures than peak exercise performance.
Pharmacokinetic Profile
SS-31 — Pharmacokinetic Curve
Subcutaneous injection, IntravenousQuick Start
- Typical Dose
- 5-10mg general support, 10-40mg clinical/performance protocols
- Frequency
- Once daily (morning or pre-workout for athletic use)
- Route
- Subcutaneous injection, Intravenous
- Cycle Length
- 4-12 weeks for therapeutic protocols
- Storage
- Reconstituted: 2-8°C in amber/dark vials, use within 30 days. Light sensitive
Molecular Structure
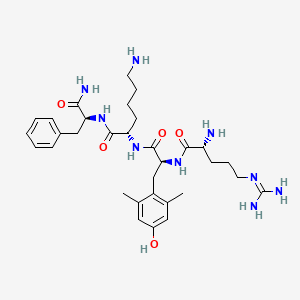
- Formula
- C32H49N9O5
- Weight
- 639.8 Da
- Length
- 4 amino acids
- CAS
- 736992-21-5
- PubChem CID
- 11764719
- Exact Mass
- 639.3857 Da
- LogP
- 0
- TPSA
- 267 Ų
- H-Bond Donors
- 9
- H-Bond Acceptors
- 8
- Rotatable Bonds
- 19
- Complexity
- 990
Identifiers (SMILES, InChI)
InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-27(41-29(44)24(34)11-8-14-38-32(36)37)31(46)39-25(12-6-7-13-33)30(45)40-26(28(35)43)17-21-9-4-3-5-10-21/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,45)(H,41,44)(H4,36,37,38)/t24-,25+,26+,27+/m1/s1
SFVLTCAESLKEHH-WKAQUBQDSA-NResearch Indications
Mitochondrial
Optimizes electron transport chain function for improved cellular energy.
Prevents cardiolipin peroxidation and reduces mitochondrial oxidative damage.
Significant improvements in 6-minute walk test and fatigue scores in PMM patients.
Cardiovascular
Reduces cardiac injury markers and improves mitochondrial function in surgical patients.
Reduces ischemia-reperfusion injury and supports cardiac function.
Enhanced exercise capacity and reduced fatigue.
Anti-Aging
Reversed age-related decline in muscle function in animal studies.
Neuroprotective effects demonstrated in Alzheimer's models.
Supports overall cellular health through mitochondrial optimization.
Research Protocols
topical
Topical cream/gel formulation available with limited research data. Used primarily for anti-aging skin applications.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Skin anti-aging | 0.5-1% cream/gel | Twice daily | —(Route: Face, neck, hands) |
subcutaneous Injection
Mitochondria-targeted peptide. Standard and advanced protocols available.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Standard — Loading | 5 mg | Once daily | Weeks 1-2 |
| Standard — Full dose | 10 mg | Once daily | Weeks 3-8 |
| Advanced — Loading | 5 mg | Once daily | Weeks 1-2 |
| Advanced — Escalation 1 | 10 mg | Once daily | Weeks 3-4 |
| Advanced — Escalation 2 | 15 mg | Split into 2 injections daily | Weeks 5-8 |
| Advanced — Full dose | 20 mg | Split into 2 injections daily | Weeks 9-12(Cycle 8-12 weeks) |
Reconstitution Guide (10mg vial + 1mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 1.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 10 mg/mL
- For 5 mg dose: draw 50 units (0.50 mL)
- For 10 mg dose: draw 100 units (1.00 mL)
- For 15 mg dose (split): draw 75 units (0.75 mL) per injection
- Store reconstituted vial refrigerated at 2-8°C
Interactions
Peptide Interactions
SS-31 and humanin protect mitochondria through complementary mechanisms at different membrane locations. SS-31 stabilizes cardiolipin in the inner mitochondrial membrane to optimize ETC function, while humanin prevents Bax/Bid-mediated pore formation in the outer mitochondrial membrane. Combined,...
CoQ10 serves as the mobile electron carrier between Complex I/II and Complex III. SS-31 optimizes the supercomplex scaffold through which CoQ10 shuttles electrons. Supplementing both could improve the "hardware" (membrane organization via SS-31) and the "software" (electron carrier availability v...
Both peptides address mitochondrial dysfunction but from opposite directions: SS-31 directly stabilizes mitochondrial membrane structure, while MOTS-c activates nuclear gene expression programs (via AMPK/PGC-1alpha) that promote mitochondrial biogenesis and quality control. SS-31 "fixes" existing...
What to Expect
What to Expect
Subtle energy improvements, reduced fatigue
Better exercise endurance, faster recovery
Improved stamina, clearer thinking, better sleep
Significant exercise capacity improvements
Sustained energy, potential biomarker changes
Safety Profile
Common Side Effects
- Excellent safety profile in clinical trials
- No significant side effects reported at therapeutic doses
- Mild injection site reactions possible
- Safe for long-term use based on current data
Contraindications
- Known hypersensitivity to peptides
- Pregnancy or breastfeeding (limited data)
Discontinue If
- Severe injection site reactions
- Allergic reaction signs (rash, breathing difficulty)
- Unusual fatigue or weakness (paradoxical reaction)
- Cardiovascular symptoms
- Severe headaches or vision changes
Quality Indicators
What to look for
- Pharmaceutical grade (>98% purity required for clinical applications)
- Clear, colorless solution after reconstitution
- Certificate of Analysis available
- Proper cold chain maintenance
Caution
- Light sensitive - protect from exposure using amber vials
- Solution pH should be 6.5-7.5 (physiological range)
Red flags
- Yellow or brown discoloration indicates degradation
- Cloudy solution or visible particles
- Improper storage conditions
Frequently Asked Questions
References (21)
- [7]Kidney Protection in Acute Injury
- [2]Cardioprotection in Heart Failure Study (2019)
- [3]Age-Related Muscle Dysfunction Study (2020)
- [4]Barth Syndrome Clinical Trial (2021)
- [5]COVID-19 Cardiac Injury Protection
- [6]Neuroprotective Effects in Alzheimer's
- [19]Liu et al — SS-31 efficacy in cardiovascular disease: a systematic review of preclinical and clinical evidence Heart Fail Rev (2023)
- [21]Reid Thompson et al — Elamipretide in patients with Barth syndrome: a randomized, double-blind, placebo-controlled clinical trial Cardiology (2023)
- [1]
- [11]Chatfield KC et al Elamipretide improves mitochondrial function in the failing human heart JACC Basic Transl Sci (2019)
- [12]Karaa A et al Randomized dose-escalation trial of elamipretide in adults with primary mitochondrial myopathy Neurology (2018)
- [13]Sabbah HN et al Chronic therapy with elamipretide improves left ventricular and mitochondrial function in dogs with advanced heart failure Circ Heart Fail (2016)
- [18]Zhao et al — Elamipretide (SS-31) improves mitochondrial dysfunction, synaptic and memory impairment induced by lipopolysaccharide in mice J Neuroinflammation (2023)
- [16]Hou Y et al The antioxidant peptide SS-31 prevents oxidative stress and improves renal function in diabetic nephropathy Nephrol Dial Transplant (2018)
- [20]Steele et al — Elamipretide in Barth syndrome: updated results from TAZPOWER Orphanet J Rare Dis (2022)
- [8]Szeto HH et al Mitochondria-targeted peptide accelerates ATP recovery and reduces ischemic kidney injury J Am Soc Nephrol (2011)
- [9]Siegel MP et al Mitochondrial-targeted peptide rapidly improves mitochondrial energetics and skeletal muscle performance in aged mice Aging Cell (2013)
- [10]Birk AV et al The mitochondrial-targeted compound SS-31 re-energizes ischemic mitochondria by interacting with cardiolipin J Am Soc Nephrol (2013)
- [14]Hortmann M et al Elamipretide diminishes circulating HtrA2 in ST-segment elevation myocardial infarction Eur Heart J Acute Cardiovasc Care (2019)
- [15]Escribano-Lopez I et al SS-31 increases SIRT1 levels and ameliorates inflammation in type 2 diabetes Sci Rep (2018)
- [17]Mo Y et al SS-31 reduces inflammation and oxidative stress through inhibition of Fis1 expression Biochem Biophys Res Commun (2019)
SR-9011
SR-9011 is a synthetic compound marketed as a selective androgen receptor modulator (SARM), though there is limited scientific evidence supporting its classific
Stamakort
Stamakort is a complex peptide preparation classified as a cytomedine — a class of tissue-specific bioregulators developed at the St. Petersburg Institute of Bi