BMS-564929
BMS-564929 is a potent, orally bioavailable nonsteroidal selective androgen receptor modulator (SARM) developed by Bristol-Myers Squibb for the potential treatment of age-related muscle wasting and hormonal decline. It demonstrates high selectivity for androgen receptors in muscle and bone tissue with minimal effects on the prostate.
Overview
BMS-564929 is a nonsteroidal selective androgen receptor modulator (SARM) identified through high-throughput screening and medicinal chemistry optimization programs at Bristol-Myers Squibb. It belongs to a class of compounds designed to selectively activate androgen receptors in target tissues such as skeletal muscle and bone while minimizing androgenic effects in the prostate and other reproductive organs. This tissue selectivity distinguishes SARMs from traditional testosterone and anabolic-androgenic steroids, which activate androgen receptors broadly across all tissues.
In preclinical studies, BMS-564929 demonstrated potent anabolic activity with an exceptional degree of selectivity. It showed full agonist activity in the levator ani muscle (a surrogate marker for skeletal muscle anabolic activity) while exhibiting greatly reduced stimulation of the prostate and seminal vesicles in castrated rat models. The compound displayed high oral bioavailability, a favorable pharmacokinetic profile, and nanomolar binding affinity for the androgen receptor. Its selectivity ratio (anabolic to androgenic activity) was among the highest reported for SARMs in its class.
BMS-564929 was investigated as a potential therapeutic agent for conditions associated with androgen deficiency, including sarcopenia, cachexia, osteoporosis, and male hypogonadism. However, the compound does not appear to have advanced beyond early-phase clinical development. Like other SARMs, BMS-564929 has appeared on the World Anti-Doping Agency (WADA) prohibited list and has been identified in products marketed to bodybuilders and athletes seeking performance enhancement. Its use outside of approved clinical trials carries unknown risks and is prohibited in competitive sports.
Mechanism of Action
Non-Steroidal Selective Androgen Receptor Modulator
BMS-564929 is a non-steroidal selective androgen receptor modulator (SARM) developed by Bristol-Myers Squibb, featuring a 2-quinolinone core structure. It binds the androgen receptor (AR) ligand-binding domain (LBD) with high affinity (Ki ~ 2.1 nM) and demonstrates full agonist activity in anabolic tissues (muscle, bone) while exhibiting minimal activity in androgenic tissues (prostate, seminal vesicles) (PMID: 16539396).
Tissue-Selective AR Activation
The tissue selectivity of BMS-564929 arises from its unique conformational induction of the AR-LBD upon binding. Unlike testosterone or DHT, BMS-564929 induces a distinct helix 12 positioning that preferentially recruits coactivator proteins abundant in muscle (SRC-1, PNRC) while poorly recruiting coactivators dominant in prostate (FKBP52, ARA70). This differential coregulator recruitment translates the same receptor binding into tissue-specific transcriptional outcomes, activating myogenic genes (MyoD, myogenin, IGF-1) without stimulating prostatic PSA or KLK2 expression (PMID: 17185411).
Anabolic Signaling — Muscle & Bone
In skeletal muscle, BMS-564929-activated AR stimulates the mTORC1/p70S6K pathway through AR-mediated upregulation of IGF-1 and inhibition of myostatin signaling. In osteoblasts, AR activation promotes Wnt/beta-catenin signaling and increases OPG/RANKL ratio, enhancing bone mineral density. Preclinical studies in castrated rats demonstrated dose-dependent increases in levator ani muscle mass comparable to testosterone propionate, with <10% stimulation of prostate weight (PMID: 16539396).
Hypothalamic-Pituitary Suppression
As a potent AR agonist, BMS-564929 suppresses the hypothalamic-pituitary-gonadal (HPG) axis through negative feedback on GnRH neurons and anterior pituitary gonadotropes, reducing LH and FSH secretion. This results in dose-dependent suppression of endogenous testosterone production, an important consideration for clinical development and post-cycle recovery (PMID: 17185411).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
High Potency:: In preclinical models, it has shown to be more potent than testosterone in stimulating muscle growth per milligram. Tissue Selectivity:: Highly effective at separating muscle-building benefits from undesirable androgenic side effects. Oral Bioavailability:: Unlike many traditional androgens, it is designed for effective oral administration
- In preclinical models, it has shown to be more potent than testosterone in stimulating muscle growth per milligram
- Highly effective at separating muscle-building benefits from undesirable androgenic side effects
- Unlike many traditional androgens, it is designed for effective oral administration
Safety Profile
Safety Profile: BMS-564929
Common Side Effects
- Suppression of endogenous testosterone production (hypothalamic-pituitary-gonadal axis suppression)
- Acne and oily skin
- Headache
- Mild transient liver enzyme elevations (ALT/AST)
- Changes in lipid profile (decreased HDL cholesterol)
- Mood changes (irritability, aggression)
Serious Adverse Effects
- Hepatotoxicity: Potential for significant liver injury; monitor liver function tests regularly
- Cardiovascular risks: Altered lipid profiles and potential prothrombotic effects (class effect of SARMs)
- Hormonal suppression: Sustained HPG axis suppression may lead to hypogonadism requiring PCT (post-cycle therapy)
- Polycythemia: Androgen receptor agonism may stimulate erythropoiesis excessively, increasing thromboembolic risk
- Left ventricular hypertrophy: Theoretical concern based on androgen receptor activation in cardiac tissue
- Tendon weakening: Preclinical data suggests possible adverse effects on tendons despite muscle anabolism
Contraindications
- Prostate cancer or elevated PSA without urological evaluation
- Breast cancer (hormone-sensitive)
- Severe hepatic impairment or active liver disease
- Known cardiovascular disease, history of stroke or MI
- Pregnancy or women planning to conceive (teratogenic potential)
- Polycythemia vera or hematocrit >54%
Drug Interactions
- CYP3A4 inhibitors (ketoconazole, ritonavir): May increase BMS-564929 exposure; dose adjustment potentially needed
- Anticoagulants (warfarin, DOACs): Androgens may alter coagulation parameters; monitor closely
- Insulin/Oral hypoglycemics: SARMs may improve insulin sensitivity; monitor for hypoglycemia
- Other androgens/anabolic agents: Additive hormonal suppression and hepatotoxicity risk
- Corticosteroids: Potential antagonistic effects on muscle; combined hepatic stress
Population-Specific Considerations
- Pregnancy/Lactation: Absolutely contraindicated; potential for virilization of female fetus and developmental toxicity
- Pediatric/Adolescent: Contraindicated; risk of premature epiphyseal closure and endocrine disruption
- Women: Risk of virilization (voice deepening, hirsutism, clitoral enlargement); not recommended
- Elderly men: Use with caution; cardiovascular and prostate risks increase with age
- Renal impairment: Limited data; use with caution
- Note: BMS-564929 is an investigational SARM not approved for human use by any regulatory agency. All safety data derives from preclinical studies and limited Phase I trials. Use carries significant unknown risks
Pharmacokinetic Profile
Molecular Structure
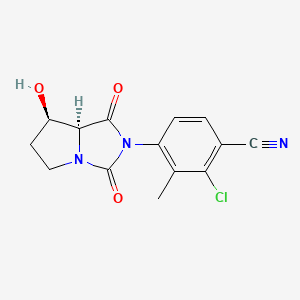
- Formula
- C14H12ClN3O3
- Weight
- 305.71 Da
- PubChem CID
- 9882972
- Exact Mass
- 305.0567 Da
- LogP
- 1.1
- TPSA
- 84.6 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 1
- Complexity
- 531
Identifiers (SMILES, InChI)
InChI=1S/C14H12ClN3O3/c1-7-9(3-2-8(6-16)11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,12,19H,4-5H2,1H3/t10-,12+/m1/s1
KEJORAMIZFOODM-PWSUYJOCSA-NSafety Profile
Common Side Effects
- Hormonal Suppression:: Like all SARMs, it may suppress natural testosterone production at higher dosages
- Lipid Changes:: Potential reduction in HDL (good) cholesterol, a common trait across the SARM class
- SHBG Reduction:: Likely to cause a decrease in Sex Hormone-Binding Globulin levels during active use
References (3)
- [1]Ockham's Razor and Selective Androgen Receptor Modulators (SARMs): Are We Overlooking the Role of 5α-Reductase?
→ This study explains how SARMs like BMS-564929 achieve selectivity because they are not substrates for 5α-reductase, preventing the localized amplification of androgenic effects in the prostate.
- [2]Selective Androgen Receptor Modulators (SARMs) as Function Promoting Therapies
→ Research indicates that nonsteroidal SARMs act as full agonists in muscle and bone while acting as only partial agonists in the prostate, offering a safer profile than traditional steroids.
- [3]Selective androgen receptor modulators: the future of androgen therapy?
→ Clinical analysis suggests that SARMs represent a promising class of drugs for treating muscle wasting and hypogonadism with significantly reduced side effects compared to testosterone.
BMP-7
BMP-7 is a potent osteoinductive growth factor belonging to the transforming growth factor-beta (TGF-beta) superfamily. Originally identified for its ability to
BNP (B-type Natriuretic Peptide)
B-type Natriuretic Peptide (BNP), also known as Brain Natriuretic Peptide due to its original isolation from porcine brain tissue in 1988 by Sudoh et al., is a