Idebenone
A synthetic analog of CoQ10 with superior blood-brain barrier penetration, used for mitochondrial support, neuroprotection, and treatment of Leber's hereditary optic neuropathy.
Overview
Idebenone (2-(10-hydroxydecyl)-5,6-dimethoxy-3-methyl-2,5-cyclohexadiene-1,4-dione) is a short-chain synthetic benzoquinone structurally related to coq10 but engineered for superior bioavailability and blood-brain barrier (BBB) penetration. Developed in the 1980s by Takeda Pharmaceutical Company, idebenone was designed to overcome CoQ10's primary limitation — its large molecular weight and high lipophilicity that restrict CNS access. Idebenone functions as an electron carrier in the mitochondrial electron transport chain, shuttling electrons from complexes I and II to complex III, thereby restoring ATP production in cells with compromised mitochondrial function. It also acts as a potent antioxidant, scavenging reactive oxygen species and preventing lipid peroxidation.
Clinically, idebenone is approved in Europe (marketed as Raxone) for the treatment of Leber's hereditary optic neuropathy (LHON), a mitochondrial disease causing acute vision loss. In the RHODOS trial, idebenone at 900 mg/day demonstrated significant protection of visual acuity in LHON patients with recent onset. It has also been investigated in Friedreich's ataxia, where it reduced cardiac hypertrophy markers, and in Duchenne muscular dystrophy, though results in these conditions have been mixed. Its ability to bypass a dysfunctional complex I makes it particularly valuable in diseases characterized by complex I deficiency, distinguishing it from coq10, which requires a functional complex I for optimal electron transfer.
In the nootropic and longevity communities, idebenone is valued for its cognitive-enhancing and anti-aging potential. Preclinical studies show improvements in memory, learning, and neuronal survival in models of oxidative stress and neurodegeneration. Typical supplemental doses range from 150–300 mg/day, though clinical doses for LHON reach 900 mg/day. Idebenone pairs well with other mitochondrial support compounds such as pqq, nad-plus, and alpha-lipoic-acid in protocols targeting mitochondrial biogenesis and cellular energy production. Its dermatological applications have also expanded, with topical idebenone formulations demonstrating superior antioxidant protection against photoaging compared to other antioxidants including vitamin C and vitamin E.
Mechanism of Action
Idebenone is a synthetic short-chain analog of coenzyme Q10 (CoQ10) designed to have improved bioavailability and ability to cross the blood-brain barrier. Its primary mechanism involves functioning as an electron carrier within the mitochondrial electron transport chain (ETC). Idebenone can shuttle electrons between Complex I, II, and III, and critically, it can bypass a dysfunctional Complex I to deliver electrons directly to Complex III. This bypass capability is particularly valuable in mitochondrial diseases such as Leber's hereditary optic neuropathy (LHON), where Complex I dysfunction impairs ATP production.
As an antioxidant, idebenone directly scavenges reactive oxygen species (ROS), protecting cellular components from oxidative damage. Its antioxidant capacity is enhanced through interaction with the NAD(P)H:quinone oxidoreductase 1 (NQO1) enzyme, which reduces idebenone to its hydroquinone form—a more potent antioxidant. This NQO1-mediated activation is linked to the Nrf2 signaling pathway, a master regulator of cellular antioxidant responses that upregulates a battery of protective genes.
The dual mechanism of idebenone—maintaining mitochondrial ATP production while providing antioxidant protection—makes it particularly suited for conditions involving mitochondrial dysfunction and oxidative stress. It has demonstrated efficacy in mitochondrial diseases, neurodegenerative conditions, and dermatological applications where oxidative damage plays a central role in pathology.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Clinical Evidence:: Strongest evidence exists for Leber's Hereditary Optic Neuropathy (LHON) and mitochondrial disorders. Individual Variation:: Effectiveness varies significantly between individuals, with some experiencing clear benefits while others notice little effect. Stacking Context:: Often used as part of complex nootropic stacks rather than standalone, making isolated effects difficult to assess. Comparison to CoQ10:: Users debate whether idebenone offers advantages over standard CoQ10 supplementation, with mixed conclusions
- Strongest evidence exists for Leber's Hereditary Optic Neuropathy (LHON) and mitochondrial disorders
- Effectiveness varies significantly between individuals, with some experiencing clear benefits while others notice little effect
- Often used as part of complex nootropic stacks rather than standalone, making isolated effects difficult to assess
- Users debate whether idebenone offers advantages over standard CoQ10 supplementation, with mixed conclusions
Safety Profile
Safety Profile: Idebenone
Common Side Effects
- Gastrointestinal symptoms (most common): nausea, diarrhea, abdominal pain, and dyspepsia affecting up to 20% of users
- Headache and dizziness
- Mild insomnia or sleep disturbance when taken late in the day
- Fatigue (paradoxical) during initial dosing period
- Skin flushing or warmth sensation
Serious Adverse Effects
- Hepatotoxicity: rare but documented cases of elevated liver enzymes; dose-dependent risk increases above 900 mg/day
- Hypotension: CoQ10-like activity may lower blood pressure; symptomatic drops reported in predisposed patients
- Thrombocytopenia: very rare reports in post-marketing surveillance
- Rhabdomyolysis: extremely rare; theoretical concern due to mitochondrial effects, primarily when combined with statins
- Allergic reactions: rare anaphylactoid reactions reported
Contraindications
- Known hypersensitivity to idebenone or benzoquinone derivatives
- Severe hepatic impairment (Child-Pugh C)
- Concurrent use of drugs that impair mitochondrial function (e.g., certain antiretrovirals) without monitoring
- Caution in patients with bleeding disorders (mild antiplatelet activity reported)
Drug Interactions
- Anticoagulants / antiplatelets (warfarin, aspirin): structurally related to CoQ10; may enhance or reduce anticoagulant effects — INR monitoring recommended
- Statins: potential additive risk of myopathy; both affect mitochondrial energetics
- Antihypertensives: additive blood pressure–lowering effects; monitor BP
- CYP450 metabolism: idebenone undergoes extensive hepatic metabolism; potential interactions with strong CYP3A4 inhibitors (ketoconazole, ritonavir) may increase idebenone levels
- Chemotherapy agents: antioxidant properties may theoretically interfere with oxidative-stress-based cancer treatments
Population-Specific Considerations
- Pregnancy / lactation: no adequate human safety data; animal studies show no teratogenicity, but avoid unless benefits clearly outweigh risks
- Children: approved in the EU for Leber's hereditary optic neuropathy (LHON) in adolescents ≥12 years at 900 mg/day; off-label pediatric use requires specialist supervision
- Elderly: start at lower doses; increased risk of hypotension and GI side effects; monitor hepatic and renal function
- Friedreich's ataxia patients: extensively studied but did not meet primary endpoints in Phase III trials; cardiac biomarkers may still improve
- Renal impairment: limited data; metabolites are renally cleared — use caution with severe renal insufficiency
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Commonly used at 200-600mg daily, divided into 1-2 doses
Molecular Structure
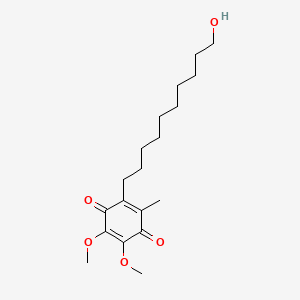
- Formula
- C19H30O5
- Weight
- 338.4 Da
- PubChem CID
- 3686
- Exact Mass
- 338.2093 Da
- LogP
- 4.3
- TPSA
- 72.8 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 12
- Complexity
- 502
Identifiers (SMILES, InChI)
InChI=1S/C19H30O5/c1-14-15(12-10-8-6-4-5-7-9-11-13-20)17(22)19(24-3)18(23-2)16(14)21/h20H,4-13H2,1-3H3
JGPMMRGNQUBGND-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Generally Well-Tolerated:: Most users report minimal to no side effects at standard doses
- Pro-oxidant Risk:: At certain doses or contexts, may act as a pro-oxidant rather than antioxidant
- Interaction Concerns:: Potential interactions with methylene blue and other electron transport chain modulators
- Limited Long-term Data:: Less human data available compared to CoQ10 for extended use periods
References (7)
- [2]Idebenone and Neuroprotection: Antioxidant, Pro-oxidant, or Electron Carrier?
→ While idebenone is marketed as an antioxidant, it can also act as a pro-oxidant by forming unstable semiquinone at complex I, and its antioxidant function critically depends on two-electron reduction to idebenol without creating unstable intermediates.
- [3]Current and Emerging Treatment Modalities for Leber's Hereditary Optic Neuropathy: A Review of the Literature
→ Idebenone shows the most promising clinical outcomes among treatment options for LHON, with recent real-life studies demonstrating effectiveness in treating visual impairment in this mitochondrial disorder.
- [4]Current treatment options for treating OPA1-mutant dominant optic atrophy
→ Idebenone has shown the most promising clinical outcomes in real-life studies for treating dominant optic atrophy caused by OPA1 gene mutations, though more evidence from clinical trials is needed for gene therapy approaches.
- [5]Therapeutic Advances and Future Prospects in Progressive Forms of Multiple Sclerosis
→ Idebenone is being studied in phase II/III trials as an antioxidant therapy for progressive forms of multiple sclerosis, alongside other novel therapeutic approaches targeting neurodegeneration.
- [6]Nutritional and exercise-based therapies in the treatment of mitochondrial disease
→ Idebenone has been shown to bypass defective respiratory enzymes or scavenge free radicals in mitochondrial diseases, though responses vary and many reports show no benefit, requiring careful individual assessment.
- [7]A case of reversible toxic optic neuropathy from tacrolimus (FK506)
→ Idebenone supplementation demonstrated marked improvement in visual acuity and color vision in a transplant patient with tacrolimus-induced toxic optic neuropathy within two months of treatment.
- [1]Border between natural product and drug: Comparison of the related benzoquinones idebenone and coenzyme Q10
→ Idebenone shares CoQ10's quinone moiety but has a shorter, less lipophilic tail, making it a synthetic pharmacologically active compound that can bypass complex I defects in the mitochondrial respiratory chain while maintaining antioxidant properties.
I3C
Indole-3-carbinol (I3C) is a naturally occurring compound found in cruciferous vegetables like broccoli and Brussels sprouts that is converted to DIM (diindolyl
IDRA-21
IDRA-21 is a benzothiadiazine derivative and a positive allosteric modulator of the glutamatergic AMPA receptor. It works by attenuating the rapid desensitizati