Gonadorelin
Gonadorelin (GnRH) is a decapeptide gonadotropin-releasing hormone that stimulates LH and FSH release, with clinical applications in fertility assessment and research into breast cancer, prostate cancer, and Alzheimer's disease.
Overview
Gonadorelin is the synthetic form of the endogenous gonadotropin-releasing hormone produced by the hypothalamus. It acts on GnRH receptors in the anterior pituitary to trigger pulsatile release of LH and FSH, which in turn regulate gonadal steroidogenesis and gametogenesis. Continuous (non-pulsatile) administration leads to receptor downregulation and suppression of the hypothalamic-pituitary-gonadal (HPG) axis, a property exploited therapeutically to reduce sex hormone levels in hormone-sensitive conditions.
Mechanism of Action
Gonadorelin binds to GnRH type I receptors (GnRHR) on gonadotroph cells of the anterior pituitary. When administered in a pulsatile fashion, it stimulates the release of both LH and FSH. In contrast, continuous administration causes GnRHR desensitization and internalization, leading to profound suppression of LH and FSH secretion and downstream sex steroid production. This dual nature -- stimulatory when pulsatile, suppressive when continuous -- underlies its diverse clinical and research applications.
Reconstitution Calculator
Gonadorelin
Gonadorelin (GnRH) is a naturally occurring decapeptide gonadotropin-releasing h

Set up a clean workspace with all supplies ready.
2x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Prostate Cancer
Prostate cancer is among the most hormone-sensitive malignancies. Gonadorelin administration achieves medical castration by suppressing testicular testosterone production, a breakthrough first applied in 1979 that replaced surgical orchiectomy. GnRH-based androgen deprivation therapy (ADT) proved equally effective and potentially reversible (Labrie, 2010).
This research revealed that some androgens are produced locally in the prostate through intracrinology (Labrie, 2014), meaning systemic GnRH blockade is only partially effective. This discovery led to the development of combined androgen blockade (CAB), which pairs GnRH agonists with drugs targeting local androgen production. When combined with early detection, CAB offers a near-curative approach, with research suggesting up to 99% of prostate cancers can be cured when caught early (Labrie, 2006).
Breast Cancer Prevention
Higher lifetime exposure to estrogen is associated with increased breast cancer risk. Research demonstrates that gonadorelin can suppress ovarian estrogen production, offering a potential chemoprevention strategy for high-risk women. Studies suggest that 10 years of GnRH agonist use could reduce breast cancer risk by approximately 50%, with 15 years potentially reducing risk by 70% (Spicer & Pike, 1994).
In postmenopausal women with hyperandrogenism, long-term GnRH treatment reduces overall estrogen levels and breast cancer risk without serious adverse effects, offering an alternative to surgical oophorectomy (Vollaard et al., 2011). Additionally, adjuvant therapy with GnRH agonists may enhance the efficacy of estrogen receptor-blocking medications by reducing estrogen expression at the source, potentially extending the useful lifespan of anti-estrogen therapies (Secreto et al., 2017).
Alzheimer's Disease and Dementia
Luteinizing hormone (LH) acts on the hippocampus and has been correlated with increased Alzheimer's disease risk. Research in rats shows that LH administration decreases memory performance and hippocampal function (Burnham et al., 2017). Higher LH levels have been associated with increased neuropathology and amyloid plaque development (Rao, 2017).
Genetic ablation of the LH receptor in mouse models of Alzheimer's disease improves amyloid pathology and preserves astrocyte health (Lin et al., 2010). Leuprolide, a GnRH receptor agonist, has been associated with decreased dementia risk compared to other GnRH analogues, likely due to its ability to down-regulate serum gonadotropins while offsetting effects of reduced testosterone (Bowen et al., 2016; Smith et al., 2018).
Research into the interaction of APOE and MS4A6A genetic loci with GnRH signaling in late-onset Alzheimer's disease suggests that interfering with either locus may be sufficient to disrupt the pathological pathway (Caceres et al., 2017).
Safety Profile
Gonadorelin has a well-established safety profile in clinical use. Common side effects include hot flashes, headache, nausea, and injection site reactions. Prolonged use of GnRH agonists can lead to bone mineral density loss, which is typically managed with calcium and vitamin D supplementation or bisphosphonates. In men undergoing androgen deprivation therapy, metabolic effects including weight gain, insulin resistance, and cardiovascular considerations have been reported. The initial "flare" effect (transient increase in sex hormones before suppression) is a known phenomenon with agonist formulations and may require concurrent anti-androgen therapy in prostate cancer treatment.
Pituitary Function Diagnostic Testing
The standard GnRH stimulation test uses a single intravenous bolus of 100 microg gonadorelin acetate. Blood samples are drawn at baseline, 15, 30, 45, and 60 minutes post-injection for LH and FSH measurement. A normal response is a 3-6 fold rise in LH peaking at 15-30 minutes, with a more modest FSH rise peaking at 30-60 minutes. Failure to mount an LH response suggests pituitary gonadotroph insufficiency, while an exaggerated response suggests hypothalamic (rather than pituitary) dysfunction Roth et al. (1972).
Pulsatile GnRH for Hypothalamic Amenorrhea
Pulsatile gonadorelin is administered subcutaneously or intravenously via a portable infusion pump delivering 5-20 microg per pulse every 60-120 minutes for ovulation induction in women with hypothalamic amenorrhea. Studies report ovulation rates of 70-95% per cycle, with a cumulative pregnancy rate of approximately 80-90% over 6 cycles. The subcutaneous route uses 15-20 microg/pulse at 90-minute intervals, while intravenous delivery uses 5-10 microg/pulse at 60-90 minute intervals Martin et al. (1990).
Prostate Cancer ADT Protocols
In prostate cancer androgen deprivation therapy, continuous GnRH agonist administration achieves medical castration (testosterone < 50 ng/dL) within 2-4 weeks. Standard protocols include depot formulations of GnRH agonists (leuprolide 7.5 mg monthly or 22.5 mg every 3 months; goserelin 3.6 mg monthly or 10.8 mg every 3 months). Combined androgen blockade adds an anti-androgen (bicalutamide 50 mg/day or flutamide 250 mg TID) starting 1-2 weeks before GnRH agonist to prevent testosterone flare Labrie (2010).
Breast Cancer Chemoprevention Research
Modeling studies by Spicer & Pike estimated that GnRH agonist administration beginning at age 25 and continued for 10 years could reduce breast cancer risk by ~50%, while 15 years of use could achieve ~70% risk reduction. These projections are based on the relationship between cumulative ovarian estrogen exposure and breast cell division rates Spicer & Pike (1994). Clinical protocols in postmenopausal hyperandrogenic women used goserelin 3.6 mg subcutaneous depot every 28 days for 6-12 months, achieving significant reductions in estradiol, androstenedione, and testosterone Vollaard et al. (2011).
Pharmacokinetic Profile
Gonadorelin — Pharmacokinetic Curve
Subcutaneous injection, Intravenous injectionQuick Start
- Typical Dose
- 100-200 mcg for single injections; 5-20 mcg per pulse for pulsatile therapy
- Frequency
- 2-3 times weekly for fertility/TRT support; every 90-120 minutes for pulsatile therapy; single dose for diagnostic
- Route
- Subcutaneous injection, Intravenous injection
- Cycle Length
- Varies - diagnostic single use, fertility therapy ongoing, TRT support continuous
- Storage
- Lyophilized: 2-8°C refrigerated; Reconstituted: 2-8°C refrigerated, use within 14 days due to short stability
Molecular Structure
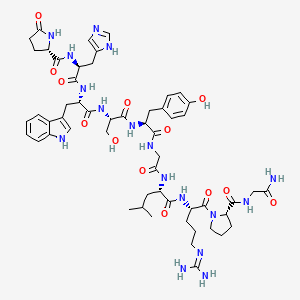
- Formula
- C55H75N17O13
- Weight
- 1 Da
- Length
- 10 amino acids
- CAS
- 9034-40-6
- PubChem CID
- 638793
- Exact Mass
- 1181.5730 Da
- LogP
- -2.4
- TPSA
- 475 Ų
- H-Bond Donors
- 16
- H-Bond Acceptors
- 15
- Rotatable Bonds
- 31
- Complexity
- 2390
Identifiers (SMILES, InChI)
InChI=1S/C55H75N17O13/c1-29(2)19-38(49(80)67-37(9-5-17-60-55(57)58)54(85)72-18-6-10-43(72)53(84)62-25-44(56)75)66-46(77)26-63-47(78)39(20-30-11-13-33(74)14-12-30)68-52(83)42(27-73)71-50(81)40(21-31-23-61-35-8-4-3-7-34(31)35)69-51(82)41(22-32-24-59-28-64-32)70-48(79)36-15-16-45(76)65-36/h3-4,7-8,11-14,23-24,28-29,36-43,61,73-74H,5-6,9-10,15-22,25-27H2,1-2H3,(H2,56,75)(H,59,64)(H,62,84)(H,63,78)(H,65,76)(H,66,77)(H,67,80)(H,68,83)(H,69,82)(H,70,79)(H,71,81)(H4,57,58,60)/t36-,37-,38-,39-,40-,41-,42-,43-/m0/s1
XLXSAKCOAKORKW-AQJXLSMYSA-NResearch Indications
Fertility & Reproductive
Treatment of conditions like Kallmann syndrome where GnRH deficiency causes low testosterone and infertility.
Induces ovulation in women with hypothalamic amenorrhea who don't ovulate regularly.
Stimulates testosterone and sperm production by restoring LH/FSH pulsatility.
Treatment of delayed puberty due to hypothalamic-pituitary dysfunction.
Diagnostic
Used to assess hypothalamic-pituitary function and diagnose GnRH deficiency.
Evaluates pituitary reserve and responsiveness to GnRH stimulation.
Hormone Optimization
Maintains testicular function and fertility during testosterone replacement therapy.
Helps restore natural testosterone production after anabolic steroid use.
Treatment of undescended testes in pediatric patients.
Research Protocols
intranasal Injection
Nasal spray formulation available in some regions for convenience of administration.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Hormone support | As prescribed | Per protocol | —(Route: Intranasal) |
subcutaneous Injection
GnRH analog for LH/FSH stimulation. Standard protocol: 2-3x weekly on non-consecutive days.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Initiation | 50 mcg | 2-3x weekly | Weeks 1-2(Non-consecutive days) |
| Escalation | 100 mcg | 2-3x weekly | Weeks 3-4 |
| Standard dose | 100-150 mcg | 2-3x weekly | Weeks 5-8(Cycle 4-8 weeks) |
| Advanced/post-cycle | 200 mcg | Once daily | 7 days, then 100 mcg daily(Daily dosing max 14 days to avoid pituitary desensitization) |
Reconstitution Guide (2mg vial + 2mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 2.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 1.0 mg/mL
- For 50 mcg dose: draw 5 units (0.05 mL)
- For 100 mcg dose: draw 10 units (0.10 mL)
- For 150 mcg dose: draw 15 units (0.15 mL)
- For 200 mcg dose: draw 20 units (0.20 mL)
- Store reconstituted vial refrigerated at 2-8°C
Interactions
Peptide Interactions
The most extensively studied combination pairs GnRH agonist-induced suppression of testicular testosterone with an androgen receptor blocker (bicalutamide, flutamide, or enzalutamide) to inhibit adrenal and intracrine androgen signaling. In a meta-analysis of 27 randomized trials involving 8,275 ...
What to Expect
What to Expect
LH and FSH release begins
Testosterone increase in response to LH
Improved hormonal markers on blood tests
Restoration of natural hormone pulsatility
Maintained testicular function and fertility
Safety Profile
Common Side Effects
- Injection site reactions
- Headache
- Flushing
- Nausea
- Abdominal discomfort
Contraindications
- Hormone-sensitive tumors
- Pregnancy
- Hypersensitivity to GnRH or analogs
- Conditions worsened by sex hormone release
Discontinue If
- Signs of allergic reaction (rash, difficulty breathing)
- Severe headaches
- Unusual abdominal pain
Quality Indicators
What to look for
- White lyophilized powder
- Clear solution after reconstitution
- Intact vacuum seal
Caution
- Slight clumping that dissolves easily
Red flags
- Discolored powder
- Cloudy or particulate solution
- Broken seal
Frequently Asked Questions
References (20)
- [21]Del Re et al -- GnRH agonists vs antagonists in prostate cancer: a systematic review and meta-analysis Crit Rev Oncol Hematol (2022)
- [1]Gonadorelin in Hypogonadotropic Hypogonadism (2018)
- [2]GnRH Pulsatility and Reproductive Function (2020)
- [3]Gonadorelin for Ovulation Induction (2015)
- [7]Vollaard et al *J Clin Endocrinol Metab* J Clin Endocrinol Metab (2011)
- [8]Labrie *Prog Brain Res* Prog Brain Res (2010)
- [18]Maggi et al -- GnRH analogues in prostate cancer: current roles and emerging strategies Endocr Relat Cancer (2023)
- [19]Rossi et al -- GnRH agonists and antagonists in prostate cancer: a re-evaluation of cardiovascular safety J Endocrinol Invest (2023)
- [20]Bowen et al -- Luteinizing hormone as a therapeutic target for Alzheimer's disease Front Endocrinol (2023)
- [14]Lin et al *J Neuropathol Exp Neurol* J Neuropathol Exp Neurol (2010)
- [15]Bowen et al *J Clin Oncol* J Clin Oncol (2016)
- [16]Smith et al *J Alzheimers Dis* J Alzheimers Dis (2018)
- [17]
- [4]
- [10]
- [5]Spicer & Pike *J Natl Cancer Inst Monogr* J Natl Cancer Inst Monogr (1994)
- [6]Secreto et al *Endocr Relat Cancer* Endocr Relat Cancer (2017)
- [11]
- [12]
- [13]Rao *Reprod Sci* Reprod Sci (2017)
GnRH (Gonadotropin-Releasing Hormone)
**GnRH (Gonadotropin-Releasing Hormone)** is a hypothalamic decapeptide that functions as the master regulator of the reproductive endocrine axis. First isolate
Gotu Kola
Centella asiatica, commonly known as Gotu Kola, is a traditional Ayurvedic herb used as a neuroprotective brain tonic that enhances cognitive function through t