Prucalopride
A highly selective serotonin 5-HT4 receptor agonist approved for chronic idiopathic constipation that stimulates colonic motility and accelerates gut transit, representing a significant advancement over earlier prokinetic agents with improved cardiac safety.
Overview
Prucalopride is a dihydrobenzofurancarboxamide derivative and a highly selective agonist of the serotonin 5-HT4 receptor, developed by Janssen Pharmaceutica and approved in Europe (2009), Canada (2011), and the United States (2018, marketed as Motegrity) for the treatment of chronic idiopathic constipation (CIC) in adults who have not achieved adequate relief with laxatives. Its development addressed a critical unmet need, as previous 5-HT4 agonists — notably cisapride and tegaserod — were withdrawn or restricted due to cardiovascular adverse effects (QT prolongation and cardiac ischemic events, respectively). Prucalopride's high selectivity for the 5-HT4 receptor (>150-fold selectivity over other serotonin receptor subtypes and the hERG potassium channel) confers a significantly improved cardiac safety profile.
The 5-HT4 receptor is expressed throughout the gastrointestinal tract, particularly in the myenteric plexus, where its activation initiates the peristaltic reflex by triggering acetylcholine release from excitatory motor neurons. Prucalopride stimulates this pathway, producing coordinated propulsive contractions that accelerate colonic transit. Unlike stimulant laxatives that primarily affect water secretion or direct smooth muscle contraction, prucalopride restores physiological motility patterns — enhancing high-amplitude propagating contractions, the primary propulsive force in the colon. Clinical trials have demonstrated that prucalopride (2 mg once daily) significantly increases the proportion of patients achieving three or more spontaneous complete bowel movements per week, improves stool consistency, reduces straining, and enhances patient satisfaction and quality of life.
Prucalopride is generally well tolerated, with headache, nausea, abdominal pain, and diarrhea as the most common adverse effects, typically occurring on the first day of treatment and resolving subsequently. Extensive cardiovascular safety studies, including thorough QT studies and long-term monitoring, have confirmed no clinically meaningful effects on cardiac repolarization or ischemic risk. Prucalopride represents a distinct mechanism from osmotic laxatives like polyethylene glycol, secretagogues like linaclotide and lubiprostone, and bulk-forming agents. Its prokinetic action complements the motility-supporting effects of ginger and artichoke extract, and it occupies a role similar to the gastroprokinetic effects of itopride but targeted to the lower gastrointestinal tract.
Mechanism of Action
Mechanism of Action: Prucalopride
Prucalopride is a dihydrobenzofurancarboxamide derivative that acts as a highly selective, high-affinity agonist at serotonin 5-HT4 receptors. Unlike older prokinetic agents, it has minimal affinity for other serotonin receptors or the hERG potassium channel, resulting in a favorable cardiac safety profile.
5-HT4 Receptor Pharmacology
Prucalopride binds 5-HT4 receptors with >150-fold selectivity over other serotonin receptor subtypes. Receptor activation couples to Gs proteins, stimulating adenylyl cyclase and increasing cAMP production. This enhances neuronal excitability in the enteric nervous system and facilitates neurotransmitter release at neuroeffector junctions.
Enteric Neural Circuit Activation
In the myenteric plexus, prucalopride activates both excitatory cholinergic motor neurons (releasing ACh and substance P) and inhibitory motor neurons (releasing NO and VIP). This dual activation enables coordinated peristaltic reflexes: contraction proximal to the bolus and relaxation distally, propelling intestinal contents aborally.
Colonic Motility Enhancement
Prucalopride significantly increases the frequency and amplitude of high-amplitude propagating contractions (HAPCs) in the colon, which are the primary motor pattern responsible for mass movement and defecation. It also enhances colonic transit by increasing the basal motor index and reducing colonic tone.
Neurotrophic Effects
Emerging evidence suggests 5-HT4 receptor activation has neurotrophic properties in the enteric nervous system, potentially promoting neuronal survival and network integrity. This may contribute to sustained efficacy with chronic use, as the enteric nervous system maintains improved function.
Pharmacokinetic Considerations
Prucalopride achieves >90% oral bioavailability with a half-life of approximately 24 hours, supporting once-daily dosing. It undergoes minimal CYP450 metabolism and is primarily excreted renally unchanged, resulting in few drug-drug interactions.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Primary Indication:: Most effective for chronic constipation and IBS-C in patients who have not responded adequately to traditional laxatives. Comparative Efficacy:: Studies show benefits but bisacodyl demonstrated greater effects on bowel movement frequency in comparative analyses. Clinical Applications:: Used in specialized settings including ICU patients with motility disturbances and Parkinson's disease patients with GI dysfunction. Evidence Base:: Multiple randomized controlled trials and systematic reviews support use in chronic constipation management
- Most effective for chronic constipation and IBS-C in patients who have not responded adequately to traditional laxatives
- Studies show benefits but bisacodyl demonstrated greater effects on bowel movement frequency in comparative analyses
- Used in specialized settings including ICU patients with motility disturbances and Parkinson's disease patients with GI dysfunction
- Multiple randomized controlled trials and systematic reviews support use in chronic constipation management
Safety Profile
Safety Profile: Prucalopride
Common Side Effects
- Headache (most frequent, reported in ~25% of patients), typically resolving within the first few days of treatment
- Gastrointestinal symptoms including nausea, abdominal pain, diarrhea, and flatulence
- Dizziness and fatigue, particularly during initial dosing
- Abdominal cramping and bloating
Serious Adverse Effects
- Suicidal ideation, depression, and psychiatric disturbances reported in post-marketing surveillance; causal relationship not fully established
- Severe diarrhea leading to dehydration and electrolyte imbalances, particularly in elderly patients
- Palpitations and tachycardia; rare cases of QT prolongation reported
- Ischemic colitis in rare post-marketing reports
- Severe allergic reactions including angioedema and anaphylaxis (very rare)
Contraindications
- Known hypersensitivity to prucalopride or any excipients
- Intestinal perforation or obstruction, obstructive ileus, or severe inflammatory conditions of the intestinal tract (Crohn's disease, ulcerative colitis, toxic megacolon)
- Dialysis-dependent renal impairment (CrCl < 30 mL/min requires dose reduction to 1 mg daily)
- Pregnancy and breastfeeding (insufficient safety data; animal studies showed no teratogenicity but caution advised)
Drug Interactions
- Erythromycin and ketoconazole: P-glycoprotein inhibitors may increase prucalopride plasma concentrations; clinical significance is limited but monitor
- Anticoagulants (warfarin): No clinically significant interaction demonstrated, but GI motility changes may theoretically alter absorption of co-administered drugs
- Anticholinergics: May counteract the prokinetic effects of prucalopride
- Other serotonergic agents: Theoretical risk of additive serotonergic effects; monitor for symptoms of serotonin syndrome
Population-Specific Considerations
- Elderly (≥65 years): Start with 1 mg daily; increased incidence of headache and GI side effects; monitor for dehydration
- Pediatric: Approved in some regions for children ≥6 months; dose based on weight; safety profile similar to adults
- Renal impairment: Reduce dose to 1 mg daily for severe impairment (CrCl < 30 mL/min)
- Hepatic impairment: No dose adjustment needed for mild-to-moderate; caution in severe hepatic impairment (limited data)
- Cardiac history: Use cautiously in patients with arrhythmias or conduction abnormalities
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 2mg is the commonly cited therapeutic dose in research studies
Molecular Structure
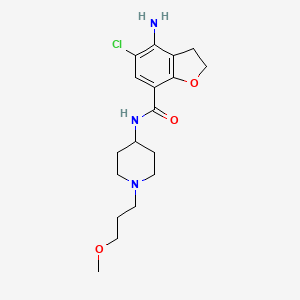
- Formula
- C18H26ClN3O3
- Weight
- 367.9 Da
- PubChem CID
- 3052762
- Exact Mass
- 367.1663 Da
- LogP
- 2
- TPSA
- 76.8 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 6
- Complexity
- 445
Identifiers (SMILES, InChI)
InChI=1S/C18H26ClN3O3/c1-24-9-2-6-22-7-3-12(4-8-22)21-18(23)14-11-15(19)16(20)13-5-10-25-17(13)14/h11-12H,2-10,20H2,1H3,(H,21,23)
ZPMNHBXQOOVQJL-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Digestive Issues:: May be associated with gastrointestinal symptoms in some patients as part of prokinetic effects
- Individual Variation:: Limited user data suggests tolerability varies between individuals
- Monitoring Required:: Patients with pre-existing conditions like SIBO should be closely monitored
- Research Safety:: Generally well-tolerated in clinical trials but long-term safety data continues to accumulate
References (8)
- [2]New Options in Constipation Management
→ Review of novel anti-constipation medications including prucalopride, lubiprostone, linaclotide, and others for patients with chronic constipation who have inadequate responses to traditional laxatives and lifestyle modifications.
- [6]Comparative Efficacy of Drugs for the Treatment of Chronic Constipation: Quantitative Information for Medication Guidelines
→ Model-based meta-analysis of 20 studies with 9,998 participants comparing prucalopride and 7 other drugs found bisacodyl had the greatest effect on bowel movement frequency, providing quantitative data for treatment guidelines.
- [3]Novel pharmacological therapies for irritable bowel syndrome
→ Comprehensive review of emerging IBS treatments including prucalopride as a potential therapy for IBS-C, alongside other novel pharmacological agents like lubiprostone and linaclotide.
- [4]Cost Effectiveness of Treatments for Chronic Constipation: A Systematic Review
→ Model-based meta-analysis evaluating the economic impact and cost-effectiveness of various chronic constipation treatments including prucalopride compared to traditional therapies.
- [5]Effect of ICU interventions on gastrointestinal motility
→ Review discussing how the 5-HT4 agonist prucalopride may help restore intestinal function in critically ill patients experiencing gastroparesis and gastrointestinal motility disturbances.
- [7]The treatment of gastroparesis, constipation and small intestinal bacterial overgrowth syndrome in patients with Parkinson's disease
→ Review of treatment options for GI dysfunction in Parkinson's patients, including prucalopride as a 5-HT4 receptor agonist for managing constipation alongside other prokinetic agents.
- [8]Clinical Practice Guideline: Irritable bowel syndrome with constipation and functional constipation in the adult
→ Comprehensive clinical practice guidelines for managing IBS-C and functional constipation in adults, including evidence-based recommendations for prucalopride and other pharmacological therapies.
- [1]The Effect of Prucalopride on the Completion Rate and Polyp Detection Rate of Colon Capsule Endoscopies
→ Study investigating prucalopride's ability to increase completion rates of colon capsule endoscopy found that 2mg supplementation improved transit times and bowel preparation quality in a cohort of 406 individuals undergoing screening.
Protegrin-1 (PG-1)
**Protegrin-1 (PG-1)** is a potent 18-amino acid cationic antimicrobial peptide originally isolated from porcine leukocytes by Kokryakov et al. in 1993. It belo
PT-141
**PT-141**, also known as bremelanotide, is a synthetic cyclic heptapeptide analog of alpha-melanocyte stimulating hormone (alpha-MSH). Unlike phosphodiesterase