Evodiamine
Evodiamine is a quinazolinocarboline alkaloid derived from the fruit of Evodia rutaecarpa (Wu Zhu Yu) that activates TRPV1 receptors and modulates multiple signaling pathways, exhibiting thermogenic, anti-obesity, anti-inflammatory, and anticancer properties in preclinical research.
Overview
Evodiamine is an indoloquinazoline alkaloid isolated from the unripe fruit of Evodia rutaecarpa (now reclassified as Tetradium ruticarpum), a plant used extensively in traditional Chinese medicine under the name Wu Zhu Yu for conditions involving cold, pain, nausea, and digestive disorders. Structurally, evodiamine features a pentacyclic ring system incorporating both indole and quinazoline moieties, and it is typically co-extracted with the related alkaloid rutaecarpine. The compound was first isolated in 1914 but has received increasing pharmacological attention since the 2000s.
The thermogenic and anti-obesity effects of evodiamine are mediated primarily through activation of transient receptor potential vanilloid 1 (TRPV1) channels, the same receptor activated by capsaicin. Unlike capsaicin, evodiamine produces minimal pungency, making it more tolerable for oral administration. TRPV1 activation stimulates catecholamine secretion from adrenal medulla, increases uncoupling protein 1 (UCP1) expression in brown adipose tissue, and enhances energy expenditure. Animal studies have demonstrated reduced visceral fat accumulation, lower serum lipids, and prevention of diet-induced obesity without changes in food intake.
Beyond metabolic effects, evodiamine has shown broad anticancer activity in preclinical models across multiple tumor types including lung, breast, colon, liver, and cervical cancers. Proposed antitumor mechanisms include topoisomerase I inhibition, induction of apoptosis through both intrinsic (mitochondrial) and extrinsic (death receptor) pathways, cell cycle arrest at G2/M phase, inhibition of angiogenesis via VEGF suppression, and reduction of NF-κB-mediated survival signaling. Anti-inflammatory effects involve suppression of prostaglandin E2 synthesis through COX-2 inhibition and reduction of pro-inflammatory cytokine production. While evodiamine shows compelling preclinical profiles, its relatively low oral bioavailability (due to extensive first-pass metabolism by CYP enzymes) remains a challenge for clinical translation.
Mechanism of Action
TRPV1 Agonism & Thermogenesis
Evodiamine is a quinazolinocarboline alkaloid isolated from the fruit of Evodia rutaecarpa (Wu Zhu Yu). It acts as a potent agonist of transient receptor potential vanilloid 1 (TRPV1) channels, the same receptor activated by capsaicin. TRPV1 activation in sensory neurons and adipose tissue triggers calcium influx and stimulates catecholamine release from the adrenal medulla, activating beta-adrenergic signaling in brown and beige adipocytes to upregulate uncoupling protein 1 (UCP1), enhancing non-shivering thermogenesis (PMID: 11577981).
Anti-Inflammatory Pathways
Evodiamine suppresses NF-kB activation by inhibiting IkB-alpha phosphorylation and degradation, thereby reducing transcription of pro-inflammatory mediators including TNF-alpha, IL-1beta, IL-6, iNOS, and COX-2. It also inhibits the NLRP3 inflammasome assembly by preventing ASC oligomerization and caspase-1 activation, reducing mature IL-1beta and IL-18 secretion. Additionally, evodiamine activates the Nrf2/HO-1 axis, bolstering cellular antioxidant defenses (PMID: 27048780).
Anti-Proliferative & Apoptotic Mechanisms
In cancer cells, evodiamine arrests the cell cycle at G2/M phase by inhibiting topoisomerase I and II catalytic activity, causing DNA damage and activating the ATM/ATR-Chk1/Chk2 checkpoint pathway. It induces mitochondrial apoptosis through Bax upregulation, Bcl-2 downregulation, cytochrome c release, and caspase-9/3 activation. Evodiamine also suppresses PI3K/Akt/mTOR signaling, reducing cell survival and proliferation (PMID: 23652588).
Metabolic & Lipid Effects
Evodiamine inhibits adipogenesis by suppressing PPAR-gamma and C/EBP-alpha transcription factor expression in preadipocytes. It activates AMPK, promoting fatty acid oxidation and inhibiting lipogenesis. These combined thermogenic and anti-adipogenic effects support its investigation as an anti-obesity compound (PMID: 18554506).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Animal vs Human Disparity:: Strong weight loss effects in rodent studies at 0.02-0.06% of diet do not translate to measurable thermogenic effects in human trials. Bioavailability Issues:: Poor absorption and extensive first-pass metabolism limit effectiveness, with research ongoing into improved delivery systems. Insufficient Human Data:: Lack of robust clinical trials and standalone user experiences make effectiveness for weight loss or other uses uncertain. Part of Blends:: Most commonly found in proprietary fat burner formulations where individual contribution cannot be isolated
- Strong weight loss effects in rodent studies at 0.02-0.06% of diet do not translate to measurable thermogenic effects in human trials
- Poor absorption and extensive first-pass metabolism limit effectiveness, with research ongoing into improved delivery systems
- Lack of robust clinical trials and standalone user experiences make effectiveness for weight loss or other uses uncertain
- Most commonly found in proprietary fat burner formulations where individual contribution cannot be isolated
Safety Profile
Safety Profile: Evodiamine
Common Side Effects
- Gastrointestinal irritation: stomach pain, nausea, heartburn, and diarrhea (capsaicinoid-like activity)
- Sensation of warmth or flushing due to thermogenic and vanilloid receptor activation
- Mild headache and dizziness
- Increased sweating (diaphoresis)
- Transient heart rate elevation (mild tachycardia)
- Dry mouth and appetite suppression
Serious Adverse Effects
- Hepatotoxicity at high doses: elevated liver enzymes (ALT, AST) observed in animal studies; human data limited
- Cardiovascular effects: significant tachycardia and blood pressure fluctuations at supratherapeutic doses
- Gastrointestinal ulceration or erosion with chronic high-dose use (TRPV1-mediated mucosal damage)
- Potential genotoxicity reported in some in vitro studies (clinical significance uncertain)
- Uterine stimulant effects: may induce contractions (abortifacient potential documented in traditional use)
- Rare severe allergic reactions
Contraindications
- Pregnancy (uterine stimulant and potential teratogenic effects documented in animal models)
- Breastfeeding (insufficient safety data; bioactive alkaloids may pass into breast milk)
- Active peptic ulcer disease or erosive gastritis
- Severe hepatic disease or active liver inflammation
- Known hypersensitivity to Evodia rutaecarpa or Rutaceae family plants
- Children under 18 years (no pediatric safety data)
Drug Interactions
- CYP enzyme substrates: Evodiamine inhibits CYP1A2, CYP2D6, and CYP3A4 in vitro; may increase plasma levels of caffeine, theophylline, SSRIs, statins, and many other drugs
- Anticoagulants (warfarin): CYP interaction may alter warfarin metabolism; increased bleeding risk
- Antihypertensives: Thermogenic effects may counteract blood pressure-lowering medications
- NSAIDs: Additive gastric mucosal damage; avoid concurrent chronic use
- Cardiac glycosides (digoxin): Potential tachycardic effects may destabilize cardiac rhythm in digitalized patients
- Thermogenic supplements (caffeine, synephrine): Additive stimulatory and cardiovascular effects; increased arrhythmia risk
Population-Specific Considerations
- Elderly: Start with minimal effective dose; increased risk of GI and cardiovascular side effects; monitor liver function
- Pediatric: Not recommended; no safety or efficacy data in children
- Pregnant/Lactating: Strictly contraindicated; documented uterine stimulant activity and potential embryotoxicity
- Hepatic impairment: Avoid or use at reduced doses with frequent LFT monitoring; hepatotoxicity reported in preclinical models
- Cardiovascular disease: Use with extreme caution; thermogenic properties may exacerbate angina, arrhythmias, or hypertension
- GI disorders: Avoid in patients with GERD, ulcers, or inflammatory bowel disease due to capsaicinoid-like irritant effects
Pharmacokinetic Profile
Molecular Structure
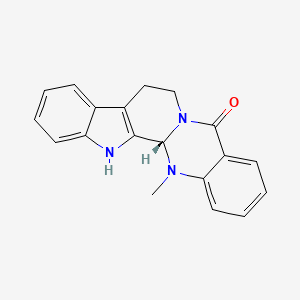
- Formula
- C19H17N3O
- Weight
- 303.4 Da
- PubChem CID
- 442088
- Exact Mass
- 303.1372 Da
- LogP
- 3.1
- TPSA
- 39.3 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 0
- Complexity
- 495
Identifiers (SMILES, InChI)
InChI=1S/C19H17N3O/c1-21-16-9-5-3-7-14(16)19(23)22-11-10-13-12-6-2-4-8-15(12)20-17(13)18(21)22/h2-9,18,20H,10-11H2,1H3/t18-/m0/s1
TXDUTHBFYKGSAH-SFHVURJKSA-NSafety Profile
Common Side Effects
- Metabolic Toxicity:: Forms reactive metabolites that can bind to cellular proteins and inhibit CYP3A4 enzyme, raising long-term safety concerns
- Drug Interactions:: May interact with medications metabolized by CYP3A4 due to enzyme inhibition
- Unknown Long-term Effects:: Limited human safety data makes chronic supplementation risks unclear
- Generally Well-Tolerated:: Short-term human studies report no significant adverse effects at tested doses
References (7)
- [5]Metabolic activation of the indoloquinazoline alkaloids evodiamine and rutaecarpine by human liver microsomes: dehydrogenation and inactivation of cytochrome P450 3A4
→ Study revealed evodiamine undergoes metabolic activation in human liver forming reactive metabolites that bind to proteins and inactivate CYP3A4, raising concerns about potential toxicity and drug interactions with long-term use.
- [2]Long-term effects of evodiamine on expressions of lipogenesis and lipolysis genes in mouse adipose and liver tissues
→ Evodiamine supplementation (0.02-0.06% of diet) significantly reduced body weight gain and rectal temperature in mice in a dose-dependent manner, while modulating expression of genes involved in lipid metabolism including PPAR-gamma.
- [3]Capsaicin and evodiamine ingestion does not augment energy expenditure and fat oxidation at rest or after moderately-intense exercise
→ Human study found that 500mg evodiamine supplementation did not significantly increase energy expenditure, thermogenesis, or fat oxidation at rest or following moderate exercise in 11 male subjects, contradicting animal study findings.
- [6]Antiproliferative Effects of Alkaloid Evodiamine and Its Derivatives
→ Comprehensive review of evodiamine's anti-cancer properties, demonstrating multitargeting antiproliferative activity through inhibition of topoisomerases, tubulin, and other proteins, though clinical application is limited by poor bioavailability.
- [7]Research progress on evodiamine, a bioactive alkaloid of Evodiae fructus: Focus on its anti-cancer activity and bioavailability
→ Review highlighting evodiamine's anticancer effects through inhibition of cell proliferation, invasion and metastasis, while noting challenges with poor bioavailability and potential toxicity from metabolism that limit clinical application.
- [1]Capsaicin-like anti-obese activities of evodiamine from fruits of Evodia rutaecarpa, a vanilloid receptor agonist
→ Evodiamine demonstrated weight loss effects comparable to capsaicin in mice, significantly reducing perirenal and epididymal fat mass, serum free fatty acids, and liver lipid levels when supplemented at 0.02-0.03% of diet, without the peppery taste of capsaicin.
- [4]Novel interactomics approach identifies ABCA1 as direct target of evodiamine, which increases macrophage cholesterol efflux
→ Research identified ABCA1 as a direct molecular target of evodiamine, demonstrating its ability to increase cholesterol efflux from macrophages, providing mechanistic insight into its lipid metabolism effects.
Everolimus
Everolimus is a semi-synthetic derivative of rapamycin that acts as a potent inhibitor of the mechanistic target of rapamycin (mTOR) complex 1. It is primarily
Exenatide (Byetta/Bydureon)
Exenatide is a glucagon-like peptide-1 (GLP-1) receptor agonist used primarily as an antidiabetic medication for type 2 diabetes mellitus. It works by stimulati