Phloretin
A dihydrochalcone flavonoid found predominantly in apple tree tissues that exhibits potent antioxidant, anti-inflammatory, and glucose transport-inhibiting properties with emerging applications in skin health and metabolic research.
Overview
Phloretin is a dihydrochalcone — a subclass of flavonoids — found primarily in the leaves, bark, and fruit skin of apple trees (Malus domestica). Its glycosylated form, phlorizin (phloretin-2'-O-glucoside), was one of the first compounds historically used to induce experimental glycosuria in research, as it inhibits sodium-glucose linked transporters (SGLTs) in the kidneys. Phloretin itself is a potent inhibitor of GLUT2, a facilitative glucose transporter expressed in the intestinal epithelium, hepatocytes, pancreatic beta cells, and renal tubules, giving it the ability to modulate glucose absorption and hepatic glucose handling. This mechanism has drawn attention in metabolic research, though the development of synthetic SGLT2 inhibitors for diabetes treatment has largely superseded phlorizin-derived compounds in clinical practice.
Beyond glucose transport, phloretin demonstrates broad biological activity as a potent antioxidant and anti-inflammatory agent. It scavenges reactive oxygen species, inhibits NF-kB signaling, and suppresses the production of pro-inflammatory cytokines including TNF-alpha, IL-1beta, and IL-6. These properties have been studied in models of UV-induced skin damage, where phloretin protects against photoaging, DNA damage, and melanogenesis. In dermatology, phloretin is incorporated into topical antioxidant formulations, often combined with vitamin C, ferulic acid, and vitamin E to create synergistic photoprotective serums. Its lipophilic nature allows good skin penetration compared to more polar flavonoids.
Emerging research has explored phloretin's anticancer potential, with in vitro studies showing growth-inhibitory effects across multiple cancer cell lines through mechanisms involving apoptosis induction, cell cycle arrest, and inhibition of the PI3K/Akt/mTOR pathway. Phloretin also appears to enhance the efficacy of certain chemotherapeutic agents by inhibiting multidrug resistance proteins. While these findings are primarily preclinical, they underscore the compound's pharmacological versatility. Dietary sources include apples (particularly the skin), apple juice, and apple cider, though concentrations are modest compared to supplemental or topical formulations. Phloretin shares structural and functional similarities with other polyphenolic antioxidants such as quercetin, resveratrol, and fisetin.
Mechanism of Action
Mechanism of Action
Phloretin (2',4',6'-trihydroxy-3-(4-hydroxyphenyl)-propiophenone) is a dihydrochalcone polyphenol primarily found in Malus domestica (apple). Unlike most flavonoids, phloretin has an open C-ring structure that contributes to its unique pharmacological profile, particularly its potent glucose transporter inhibition.
Glucose Transport Inhibition
Phloretin's best-characterized biochemical activity is competitive inhibition of facilitative glucose transporters, particularly GLUT2. The dihydrochalcone scaffold occupies the glucose-binding pocket of GLUT2, preventing glucose translocation across the cell membrane. This is pharmacologically significant in the intestinal epithelium (reducing dietary glucose absorption), hepatocytes (reducing hepatic glucose uptake), and pancreatic beta cells (modulating glucose sensing). Phloretin also inhibits GLUT1, SGLT1, and SGLT2 with lower potency.
Antioxidant Mechanisms
Phloretin's four hydroxyl groups provide robust free radical scavenging capacity through hydrogen atom transfer and single electron transfer mechanisms. The 2',4',6'-trihydroxy pattern on the A-ring is particularly effective at stabilizing phenoxyl radicals. Beyond direct scavenging, phloretin chelates pro-oxidant transition metals (Fe2+ and Cu2+), preventing Fenton chemistry. It also activates the Nrf2/ARE pathway, upregulating phase II detoxification enzymes including heme oxygenase-1 (HO-1), NAD(P)H quinone oxidoreductase 1 (NQO1), and glutathione S-transferases.
Anti-Inflammatory Pathways
Phloretin suppresses inflammation through NF-κB pathway inhibition. By blocking IKKβ activation, it prevents IκBα phosphorylation and degradation, retaining NF-κB in the cytoplasm. This reduces expression of pro-inflammatory mediators and adhesion molecules. Phloretin also inhibits MAPK pathways (p38, JNK, ERK1/2), reducing AP-1-dependent inflammatory gene expression.
Dermatological Applications
In skin biology, phloretin inhibits melanogenesis by downregulating tyrosinase expression, and it reduces UV-induced oxidative damage through its combined antioxidant and anti-inflammatory activities. Its ability to penetrate the stratum corneum makes it effective in topical formulations for photoprotection and hyperpigmentation.
Research
Reported Effects
Research Support:: Strong preclinical evidence for anti-inflammatory and antioxidant effects across multiple organ systems. Bioavailability:: As a metabolite of phlorizin (found in apples), bioavailability may vary depending on source and formulation. Mechanism Diversity:: Works through multiple complementary pathways rather than single-target action. Clinical Data:: Limited human clinical trials available; most evidence from in vitro and animal studies
- Strong preclinical evidence for anti-inflammatory and antioxidant effects across multiple organ systems
- As a metabolite of phlorizin (found in apples), bioavailability may vary depending on source and formulation
- Works through multiple complementary pathways rather than single-target action
- Limited human clinical trials available; most evidence from in vitro and animal studies
Safety Profile
Safety Profile: Phloretin
Common Side Effects
- Generally well-tolerated in both oral and topical applications at studied doses
- Mild gastrointestinal discomfort including nausea and bloating with oral use
- Skin irritation, redness, or mild burning sensation with topical application (particularly at higher concentrations)
- Contact dermatitis in sensitive individuals
- Mild headache (uncommon)
Serious Adverse Effects
- Allergic reactions in individuals with apple or Rosaceae fruit allergies
- Potential disruption of glucose transport (phloretin is a potent SGLT1/SGLT2 and GLUT2 inhibitor); may cause hypoglycemia in susceptible individuals
- Limited human safety data for oral supplementation at therapeutic doses
- Potential endocrine disruption at very high doses (estrogenic activity reported in vitro)
- Phototoxicity potential when combined with certain topical formulations
Contraindications
- Known allergy to apples, pears, or other Rosaceae family fruits
- Diabetes (particularly those on SGLT2 inhibitors or insulin) without medical supervision due to additive glucose-lowering effects
- Pregnancy and breastfeeding (insufficient safety data; estrogenic activity concern)
- Concurrent use of SGLT2 inhibitor medications (empagliflozin, dapagliflozin, canagliflozin)
Drug Interactions
- SGLT2 inhibitors (empagliflozin, dapagliflozin): Additive glucose-lowering and glycosuric effects; increased risk of hypoglycemia and diabetic ketoacidosis
- Insulin and sulfonylureas: Increased hypoglycemia risk due to glucose transporter inhibition
- Certain antibiotics (beta-lactams): Phloretin may affect renal tubular transport of some drugs
- Topical retinoids: Potential for additive skin irritation when used in skincare combinations
- Estrogen-sensitive medications (tamoxifen): Theoretical concern due to estrogenic activity
Population-Specific Considerations
- Skincare use: Most common application; typically used at 1-2% concentration in serums; well-tolerated topically; often combined with vitamin C and ferulic acid
- Diabetics: Exercise caution; phloretin's glucose transporter inhibition may synergize with diabetes medications
- Oral supplementation: Limited standardized dosing data; apple polyphenol extracts containing phloretin generally dosed at 200-500 mg/day
- Research context: Most safety data from in vitro and animal studies; human clinical data is limited primarily to dermatological applications
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Limited commercial supplement standardization data available
Molecular Structure
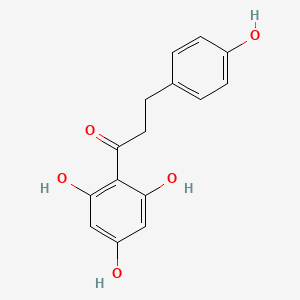
- Formula
- C15H14O5
- Weight
- 274.27 Da
- PubChem CID
- 4788
- Exact Mass
- 274.0841 Da
- LogP
- 2.6
- TPSA
- 98 Ų
- H-Bond Donors
- 4
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 4
- Complexity
- 312
Identifiers (SMILES, InChI)
InChI=1S/C15H14O5/c16-10-4-1-9(2-5-10)3-6-12(18)15-13(19)7-11(17)8-14(15)20/h1-2,4-5,7-8,16-17,19-20H,3,6H2
VGEREEWJJVICBM-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Cardiac Effects:: May affect cardiac ion channels and action potential duration based on cellular studies
- Limited Safety Data:: Insufficient long-term human safety data from clinical trials
- Drug Interactions:: Potential interactions with medications affecting cardiac function or glucose transport
- Individual Variation:: Effects may vary significantly based on individual physiology and concurrent medications
References (5)
- [2]Nootropic, neuroprotective and neurotrophic effects of phloretin in scopolamine induced amnesia in mice
→ In vitro and animal studies demonstrated that phloretin significantly influences neuronal membrane fluidity and potential, decreased excitotoxicity, and showed nootropic and neuroprotective effects in scopolamine-induced amnesia models.
- [1]The Molecular Pharmacology of Phloretin: Anti-Inflammatory Mechanisms of Action
→ Comprehensive review detailing phloretin's anti-inflammatory mechanisms through inhibition of cytokine signaling pathways, suppression of COX and iNOS, and protective effects in multiple organ systems including cardiovascular, hepatic, renal, and nervous tissue.
- [3]Apple Polyphenols, Phloretin and Phloridzin: New Trapping Agents of Reactive Dicarbonyl Species
→ Research identified phloretin as an effective trapping agent for reactive dicarbonyl species, providing antioxidant protection that may contribute to preventing oxidative stress-related diseases and cellular damage.
- [4]Effects of Phloretin and Phloridzin on Ca2+ Handling, the Action Potential, and Ion Currents in Rat Ventricular Myocytes
→ Study revealed that phloretin affects cardiac cellular function by modulating calcium handling, inhibiting transient outward potassium currents, and increasing action potential duration in ventricular myocytes.
- [5]Flavonoids and their role in oxidative stress, inflammation, and human diseases
→ Comprehensive review discussing how flavonoids including phloretin modulate oxidative stress and chronic inflammation, which are key drivers in pathogenesis of cancers, cardiovascular diseases, neurodegenerative disorders, and autoimmune conditions.
Phenylpiracetam
Phenylpiracetam is a potent racetam with a phenyl group that increases its stimulant properties and ability to cross the blood-brain barrier. It's banned by WAD
Phosphatidylcholine
Phosphatidylcholine (PC) is a phospholipid and essential component of cell membranes, particularly abundant in brain tissue and the liver. It serves as a precur