BPC-157
BPC-157 (Body Protection Compound-157) is a synthetic pentadecapeptide derived from human gastric juice that promotes wound healing, angiogenesis, and tissue repair through VEGFR2-mediated signaling. It demonstrates cytoprotective effects across gastrointestinal, musculoskeletal, cardiovascular, and neurological systems in preclinical models.
Overview
BPC-157 is a partial sequence of the larger Body Protection Compound found in human gastric juice, where it maintains mucosal barrier integrity and promotes tissue repair. The synthetic pentadecapeptide retains the healing properties of its parent molecule and has been extensively studied in rodent models across multiple organ systems. Unlike many peptides, BPC-157 is remarkably stable in acidic environments, which enables oral bioavailability and systemic activity from gastrointestinal absorption.
Research interest in BPC-157 spans wound healing, vascular growth, organ protection, and counteraction of drug-induced toxicity. It has shown efficacy in models of inflammatory bowel disease, ischemia-reperfusion injury, tendon transection, and medication side effects -- though clinical trials in humans remain limited.
Mechanism of Action
BPC-157 exerts its effects through multiple interconnected pathways:
- VEGFR2 activation: BPC-157 upregulates VEGFR2 expression and activation, stimulating endothelial cell proliferation and angiogenesis through the nitric oxide signaling cascade
- Fibroblast recruitment: Dose-dependent stimulation of fibroblast proliferation and migration, accelerating extracellular matrix deposition (collagen, fibrin, elastin, reticulin)
- FAK/paxillin signaling: Promotes F-actin formation and phosphorylation of FAK and paxillin, enhancing cell migration to wound sites
- Nitric oxide modulation: Interacts with the NO system, providing both cytoprotective and antioxidant effects
- Collateral vessel formation: Promotes "vascular running" -- the growth of collateral blood vessels around areas of ischemia or occlusion
Reconstitution Calculator
BPC-157
BPC-157 is a stable synthetic pentadecapeptide derived from a naturally occurrin

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Neuroprotection
BPC-157's neuroprotective effects have been demonstrated in models of traumatic brain injury, ischemic brain damage, and neurotoxin exposure. In cuprizone-induced demyelination models, BPC-157 promotes remyelination and oligodendrocyte survival. In MPTP models (Parkinson's disease), it protects dopaminergic neurons in the substantia nigra. These effects are mediated through VEGFR2-dependent angiogenesis in cerebral vasculature, NO-mediated neuroprotection, and direct cytoprotective signaling. Intranasal delivery would achieve higher peptide concentrations in these vulnerable brain regions compared to equivalent systemic doses.
Olfactory and Trigeminal Nerve Pathways
The anatomical basis for intranasal CNS delivery is well-established. The olfactory epithelium occupies approximately 10 cm² of the upper nasal cavity in humans, with olfactory sensory neurons extending dendrites into the nasal lumen and projecting axons through the cribriform plate directly into the CNS. The trigeminal nerve provides a complementary pathway from the respiratory epithelium to the brainstem. For a peptide like BPC-157 that targets both cortical (olfactory pathway) and brainstem (trigeminal pathway) regions, intranasal delivery provides dual-pathway access.
Brain-Gut Axis
The brain-gut axis represents the primary theoretical framework for intranasal BPC-157. Sikiric et al. (2022) published a comprehensive review establishing BPC-157 as a brain-gut axis modulator that bidirectionally influences central and peripheral targets. The peptide, derived from gastric juice, demonstrates CNS effects (dopamine modulation, anxiolysis, neuroprotection) when administered peripherally, suggesting endogenous brain-gut signaling. Intranasal delivery inverts this axis — delivering a gut-derived peptide directly to the brain — potentially amplifying central effects while maintaining peripheral signaling through systemic absorption of the nasally-delivered fraction.
Dopaminergic System Modulation
A substantial portion of BPC-157's CNS research involves dopamine system interactions. The peptide counteracts amphetamine-induced hyperlocomotion, haloperidol-induced catalepsy, and neuroleptic-induced QTc prolongation. It modulates D2 receptor sensitivity and dopamine transporter function. These effects position intranasal BPC-157 as a research tool for dopaminergic disorders including Parkinson's disease, schizophrenia adjunct therapy, and substance use disorders where dopamine dysregulation is central.
Comparison with Systemic Routes
No published studies have directly compared intranasal vs systemic BPC-157 for CNS endpoints with measured brain tissue concentrations. However, the intranasal peptide delivery literature provides strong inferential support:
- Intranasal insulin achieves 5-10x higher CSF concentrations than IV insulin at equivalent doses
- Intranasal Semax is clinically approved in Russia for stroke recovery, demonstrating the viability of intranasal neuroprotective peptides
- BPC-157's molecular weight (1,419 Da) and proteolytic stability are favorable for nose-to-brain transport efficiency
- Oral BPC-157 produces detectable CNS effects (PMID: 34856905), indicating BBB penetration occurs systemically, but likely at suboptimal concentrations
Gastrointestinal Protection
The arginine salt form has been used extensively in the Sikiric group's GI research. Studies on NSAID-induced gastropathy employed BPC-157-Arg at doses of 10 ng/kg to 10 µg/kg, demonstrating dose-dependent protection against mucosal lesions. The salt form's enhanced solubility is particularly advantageous for oral GI delivery, where rapid dissolution in the stomach allows direct mucosal contact before systemic absorption.
Comparison with Free-Acid and Sodium Salt Forms
Three forms of BPC-157 appear in the literature: free acid, sodium salt, and arginine salt. The Sikiric group has noted comparable efficacy across salt forms in most models, with the arginine salt occasionally showing enhanced effects in NO-dependent paradigms. The L-arginine and L-NAME interaction studies are particularly relevant, as they demonstrate that BPC-157's protective effects are modulated by NO precursor availability — a condition the arginine salt inherently optimizes.
Wound Healing and Tissue Repair
Preclinical wound healing studies using the arginine salt form show accelerated collagen deposition and angiogenesis consistent with free-acid BPC-157 data. The enhanced solubility of the salt form may improve bioavailability at wound sites when administered systemically, though head-to-head pharmacokinetic comparisons between salt forms have not been formally published.
Vascular and Ischemia Models
In models of venous occlusion and ischemia-reperfusion, BPC-157-Arg promoted collateral vessel formation and tissue preservation. The arginine counterion's role as an NO precursor is mechanistically relevant here, as NO is a primary mediator of vasodilation and angiogenesis in ischemic tissues.
Counteraction of Drug Toxicity
A substantial body of PL 14736 research addresses drug side effect counteraction. BPC 157 reverses or prevents GI lesions from NSAIDs, alcohol, and corticosteroids. It also counteracts systemic toxicity from antipsychotic medications, including cardiac QTc prolongation and extrapyramidal symptoms. These studies are clinically relevant because they suggest BPC 157 could serve as a protective adjunct in polypharmacy settings.
Tendon and Ligament Healing
Given the poor vascularity of connective tissues, BPC-157's dual action on angiogenesis and fibroblast recruitment makes it particularly relevant for tendon injuries. Research demonstrates that BPC-157 promotes tendon outgrowth, cell survival, and cell migration in both in vitro and in vivo models. It has shown superior efficacy compared to bFGF, EGF, and VGF in promoting healing in tendon, ligament, and bone tissue.
Inflammatory Bowel Disease Models
The IBD indication that drove PL 14736's clinical development is supported by extensive preclinical data. BPC 157 demonstrates efficacy in TNBS-induced colitis, acetic acid colitis, and ischemia-reperfusion colitis models, reducing mucosal damage scores, inflammatory infiltrate, and cytokine levels. In chronic colitis models, sustained oral dosing via drinking water provides continuous mucosal protection. The engineered bacterial delivery approach using Lactococcus lactis represents a next-generation strategy for sustained GI release.
Clinical Development History
BPC 157's path toward clinical use began at the University of Zagreb under Predrag Sikiric, whose group identified the pentadecapeptide sequence from the larger Body Protection Compound found in human gastric juice. Pliva, then Croatia's largest pharmaceutical company, licensed the compound and assigned it the development code PL 14736.
Phase I safety studies were conducted in healthy volunteers, establishing tolerability at oral doses. Phase II trials targeted ulcerative colitis, leveraging BPC 157's established GI cytoprotective effects in preclinical models. However, Pliva's acquisition by Barr Pharmaceuticals (2006) and subsequent acquisition by Teva Pharmaceutical Industries (2008) disrupted the development pipeline, and PL 14736 did not advance to Phase III.
The clinical data from these trials have not been fully published in peer-reviewed literature, making it difficult to assess efficacy outcomes. Some results were presented at gastroenterology conferences in the early 2000s, reporting favorable tolerability and preliminary efficacy signals in IBD patients.
Oral vs Injectable Administration
The oral-injectable comparison is central to BPC 157's pharmacological identity. The Sikiric group has systematically demonstrated that oral and parenteral routes produce comparable systemic effects across multiple models:
- GI lesion models: Oral BPC 157 provides direct mucosal contact plus systemic absorption, often showing equal or superior efficacy to injection for GI indications (PMID: 30598581)
- Musculoskeletal models: Subcutaneous injection near the injury site provides higher local concentrations, but oral dosing still demonstrates significant healing effects in tendon transection models
- Systemic protection: Both routes protect against drug-induced QTc prolongation and organ toxicity, confirming systemic bioavailability from oral dosing
- Dose equivalence: In most studies, oral and parenteral doses at 10 µg/kg produce overlapping effect sizes, though direct PK comparison studies with measured plasma levels are lacking
Gastric Stability Studies
The stability that defines PL 14736 has been characterized in several experimental paradigms. BPC 157 maintains activity after 24-hour incubation in human gastric juice at 37°C. It resists degradation by pepsin at pH 2.0 and shows minimal fragmentation in simulated intestinal fluid. This stability has been attributed to its proline-rich sequence and compact conformation. Comparative studies show that most bioactive peptides of similar size lose >90% activity under identical conditions.
Counteraction of Drug Side Effects
BPC-157 counteracts side effects across several drug classes. It prevents QTc prolongation induced by antipsychotics (haloperidol, clozapine, olanzapine, quetiapine) and prokinetics (metoclopramide). It also attenuates neuroleptic-induced catalepsy and somatosensory disturbance, potentially enabling more effective psychiatric pharmacotherapy by reducing treatment-limiting adverse effects.
Apiculture Applications
BPC-157 has been applied in honey bee colony management, where oral supplementation reduced Nosema ceranae fungal damage to bee GI tracts and improved hive survival rates under natural field conditions.
Vascular Growth and Ischemia Protection
BPC-157 is a potent angiogenic factor that promotes collateral blood vessel formation in ischemic tissues. This effect, primarily characterized in the GI tract, extends to cardiovascular, neurological, and muscle tissues. The peptide promotes vessel growth through VEGFR2-mediated pathways, with evidence from chicken embryo models confirming endothelial cell proliferation and survival. Bypassing of major venous occlusion has been demonstrated in rat models.
Safety Profile
BPC-157 has shown no reported toxic effects in preclinical studies across a wide dose range. It is stable in human gastric juice (unlike most peptides), enabling oral administration. No organ toxicity has been observed in rodent models at therapeutic or supratherapeutic doses. However, controlled clinical trial data in humans are limited. As a potent angiogenic agent, theoretical concerns exist regarding its use in the context of active malignancy, though no tumor-promoting effects have been reported in preclinical literature.
Pharmacokinetic Profile
BPC-157 — Pharmacokinetic Curve
Subcutaneous injectionOngoing & Future Research
- NCT04235660: A registered study examining BPC-157 for ulcerative colitis (status should be verified on ClinicalTrials.gov).
- Emerging hypotheses include BPC-157's role in the brain-gut axis (PMID: 34856905), potential applications in traumatic brain injury recovery, and engineered bacterial delivery systems for sustained GI release (PMID: 30276448).
- Interest growing in topical formulations for dermal wound healing and burn treatment.
Quick Start
- Typical Dose
- 250-500 mcg
- Frequency
- 1-2x daily
- Route
- Subcutaneous injection
- Timing
- Morning and/or evening, consistent daily timing
- Cycle Length
- 4-8 weeks
- Break Between Cycles
- 2-4 weeks
- Storage
- Refrigerate 2-8°C
Molecular Structure
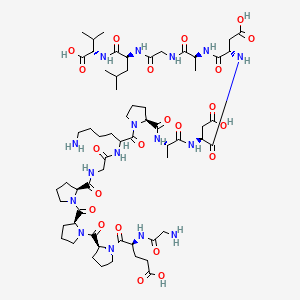
- Formula
- C62H98N16O22
- Weight
- 1419.556 Da
- Length
- 15 amino acids
- CAS
- 137525-51-0
- PubChem CID
- 108101
- Exact Mass
- 1418.7042 Da
- LogP
- -9
- TPSA
- 573 Ų
- H-Bond Donors
- 16
- H-Bond Acceptors
- 24
- Rotatable Bonds
- 39
- Complexity
- 3040
Identifiers (SMILES, InChI)
InChI=1S/C62H98N16O22/c1-31(2)25-37(55(92)74-50(32(3)4)62(99)100)71-46(81)29-65-51(88)33(5)67-53(90)38(26-48(84)85)73-54(91)39(27-49(86)87)72-52(89)34(6)68-57(94)41-15-10-21-75(41)58(95)35(13-7-8-20-63)70-45(80)30-66-56(93)40-14-9-22-76(40)60(97)43-17-12-24-78(43)61(98)42-16-11-23-77(42)59(96)36(18-19-47(82)83)69-44(79)28-64/h31-43,50H,7-30,63-64H2,1-6H3,(H,65,88)(H,66,93)(H,67,90)(H,68,94)(H,69,79)(H,70,80)(H,71,81)(H,72,89)(H,73,91)(H,74,92)(H,82,83)(H,84,85)(H,86,87)(H,99,100)/t33-,34-,35?,36-,37-,38-,39-,40-,41-,42-,43-,50-/m0/s1
HEEWEZGQMLZMFE-DGQLYNSISA-NResearch Indications
Musculoskeletal
Promotes tendon outgrowth, cell survival, and migration. Superior efficacy vs bFGF, EGF, VGF.
Accelerates ligament healing through angiogenesis and collagen deposition
Enhances osteogenic activity and bone consolidation
Accelerates skeletal muscle healing after crush or transection injury
Gastrointestinal
Counteracts NSAID, alcohol, and stress-induced gastric lesions
Significant protective effects in colitis and IBD models
Promotes healing of surgical gut anastomosis
Protects against acid reflux and esophageal lesions
Cardiovascular
Promotes collateral vessel formation in ischemic tissues
Counteracts drug-induced cardiac arrhythmia risk
Neurological
Protective effects in models of traumatic brain injury and peripheral nerve damage
Attenuates neuroleptic-induced catalepsy and somatosensory disturbance
Research Protocols
intraperitoneal Injection
Administered via intraperitoneal.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 10 µg | Per protocol | — |
intranasal Injection
Intranasal BPC-157 refers to the administration of the stable gastric pentadecapeptide BPC 157 via the nasal mucosa, utilizing the nose-to-brain pathway to achieve direct central nervous system delivery. BPC-157 at 1,419 Da is below the ~6 kDa practical threshold for intranasal nose-to-brain transpo
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 10 µg | Per protocol | —(Route: Intranasal) |
| Preclinical CNS dose extrapolation | 10 µg | Per protocol | —(Route: Intranasal) |
oral
BPC-157 is uniquely stable in gastric acid, enabling oral administration. Particularly effective for GI-targeted research.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Gut healing | 250-500 mcg | 2x daily | 4-8 weeks(Take on empty stomach) |
| Systemic effects | 500 mcg | 1-2x daily | 4-8 weeks(Lower systemic bioavailability than injection) |
subcutaneous Injection
Most common research administration route. Inject near the site of injury for localized effects, or abdominally for systemic effects.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Loading phase | 200 mcg | Once daily | Weeks 1-2(Start low to assess tolerance) |
| Standard dose | 400 mcg | Once daily | Weeks 3-4 |
| Full dose | 600 mcg | Once daily | Weeks 5-8+(Titrate up ~200 mcg every 2 weeks as tolerated) |
Reconstitution Guide (5mg vial + 3mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 3.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial — never directly onto the powder
- Gently swirl to dissolve — never shake
- Resulting concentration: 1.67 mg/mL
- For 200 mcg dose: draw 12 units on U-100 insulin syringe (0.12 mL)
- For 400 mcg dose: draw 24 units (0.24 mL)
- For 600 mcg dose: draw 36 units (0.36 mL)
- Store reconstituted vial refrigerated at 2-8°C
Interactions
Peptide Interactions
Three forms of BPC-157 appear in the literature: free acid, sodium salt, and arginine salt. The Sikiric group has noted comparable efficacy across salt forms in most models, with the arginine salt occasionally showing enhanced effects in NO-dependent paradigms. The L-arginine and L-NAME interacti...
Both promote wound healing through different pathways. GHK-Cu adds copper-dependent collagen synthesis.
Parent molecule of TB-500. Full-length TB-4 provides broader tissue repair signaling.
KPV adds anti-inflammatory effects, particularly useful for gut healing protocols.
GH-axis stimulation may complement healing effects.
GH release supports tissue repair alongside BPC-157's direct healing effects.
The most studied combination. BPC-157 promotes angiogenesis via VEGFR2 while TB-500 enhances cell migration via actin regulation. Combined, they provide complementary wound healing through parallel vascular and cellular pathways. See BPC-157/TB-500 Blend.
Complementary healing — BPC-157 via VEGFR2 angiogenesis, TB-500 via actin-mediated cell migration. See BPC-157/TB-500 Blend
What to Expect
What to Expect
Reduced inflammation and pain at injury site
Noticeable improvement in pain levels, early tissue repair begins, improved GI comfort if used for gut issues
Significant healing progress, improved mobility, continued GI improvement
Peak healing effects, substantial tissue repair, most users complete protocols in this window
Extended protocols for severe injuries or chronic conditions
Safety Profile
Common Side Effects
- Injection site redness or mild irritation
- Mild nausea (especially oral administration)
- Dizziness (rare, typically transient)
Contraindications
- Active cancer or history of cancer (angiogenic properties may theoretically promote tumor vascularization)
- Pregnancy or breastfeeding
- Children under 18
Discontinue If
- Severe allergic reaction (rash, swelling, difficulty breathing)
- Persistent nausea or GI distress
- Unexplained pain or swelling at injection site
Drug Interactions
- May interact with anticoagulants due to angiogenic effects
- Potential interaction with immunosuppressants
Uncommon
- Headache
- Fatigue
- Hot/cold flashes
Quality Indicators
What to look for
- White to off-white lyophilized powder (dry puck or cake)
- Dissolves clearly in bacteriostatic water with no particles
- Comes with third-party purity certificate (HPLC ≥98%)
- Sealed under vacuum or nitrogen atmosphere
Caution
- Slightly yellow tint (may indicate minor degradation)
- Powder appears loose/fluffy rather than solid puck
Red flags
- Gel-like or sticky consistency
- Cloudy after reconstitution or visible particles
- No COA or purity testing available
- Stored/shipped without cold chain
Frequently Asked Questions
References (20)
- [1]Seiwerth S, Rucman R, Turkovic B, et al. BPC 157 and Standard Angiogenic Growth Factors: Gastrointestinal Tract Healing, Lesson from Tendon, Ligament, and Bone Healing Current Pharmaceutical Design (2018)
→ BPC-157 showed superior efficacy compared to bFGF, EGF, and VGF in promoting healing
- [1]Huang T et al. Body protective compound-157 enhances alkali-burn wound healing in vivo and promotes proliferation, migration, and angiogenesis in vitro. Drug Des Devel Ther (2015)
- [2]Drmic D et al. Counteraction of perforated cecum lesions in rats: Effects of pentadecapeptide BPC 157, L-NAME and L-arginine. World J Gastroenterol (2018)
- [11]Strinic D, et al. BPC 157 counteracts QTc prolongation induced by haloperidol, fluphenazine, clozapine, olanzapine, quetiapine, sulpiride, and metoclopramide in rats Life Sciences (2017)
→ BPC-157 prevents drug-induced QTc prolongation
- [2]Hsieh MJ, Liu HT, Wang CN, et al. Therapeutic potential of pro-angiogenic BPC157 is associated with VEGFR2 activation and up-regulation Journal of Molecular Medicine (2017)
→ BPC-157 activates VEGFR2 to stimulate endothelial cell proliferation and angiogenesis
- [3]Chang CH, Tsai WC, Hsu YH, Pang JH. Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts Molecules (2014)
→ BPC-157 promotes FAK/paxillin signaling and F-actin formation for enhanced cell migration
- [4]Sikiric P, Rucman R, Turkovic B, et al. Novel Cytoprotective Mediator, Stable Gastric Pentadecapeptide BPC 157. Vascular Recruitment and Gastrointestinal Tract Healing Current Pharmaceutical Design (2018)
→ BPC-157 promotes collateral vessel formation around ischemic areas
- [5]Sikiric P, Seiwerth S, Rucman R, et al. Stable gastric pentadecapeptide BPC 157-NO-system relation Current Pharmaceutical Design (2014)
→ BPC-157 interacts with NO system providing cytoprotective and antioxidant effects
- [7]Sever M, Klicek R, Radic B, et al. Gastric pentadecapeptide BPC 157 and short bowel syndrome in rats Digestive Diseases and Sciences (2009)
→ Significant protective effects in colitis and ischemia-reperfusion models
- [6]Zemba M, Cilic AZ, Bayat A, et al. BPC 157 Accelerated Burn Wound Healing Annals of Burns and Fire Disasters (2016)
→ Dose-dependent stimulation of fibroblast proliferation and migration in wound models
- [8]Liang HY, et al. Engineered Lactococcus lactis for delivery of BPC 157 Journal of Biotechnology (2018)
→ L. lactis as sustained-release delivery vehicle for oral BPC-157
- [10]Sikiric P, Seiwerth S, et al. Pentadecapeptide BPC 157 interactions with the major venous occlusion Digestive Diseases and Sciences (2018)
→ Bypassing of major venous occlusion demonstrated in rat models
- [3]Amic F et al. Bypassing major venous occlusion and duodenal lesions in rats, and therapy with the stable gastric pentadecapeptide BPC 157, L-NAME and L-arginine. World J Gastroenterol (2018)
- [4]Duzel A et al. Stable gastric pentadecapeptide BPC 157 in the treatment of colitis and ischemia and reperfusion in rats: New insights. World J Gastroenterol (2017)
- [9]Skrlec K et al. Engineering recombinant Lactococcus lactis as a delivery vehicle for BPC-157 peptide with antioxidant activities. Appl Microbiol Biotechnol (2018)
- [9]Sikiric P, et al. Stable Gastric Pentadecapeptide BPC 157 in Trials for Inflammatory Bowel Disease Inflammopharmacology (2006)
→ Phase II trial initiated for IBD (PL 14736)
- [11]Tlak Gajger I et al. Stable gastric pentadecapeptide BPC 157 in honeybee (Apis mellifera) therapy, to control Nosema ceranae invasions in apiary conditions. J Vet Pharmacol Ther (2018)
- [13]Gwyer D et al. Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing. Cell Tissue Res (2019)
- [12]Sikiric P et al. Brain-gut axis and pentadecapeptide BPC 157: Theoritical and practical implications. Curr Neuropharmacol (2022)
- [8]Chang CH et al. The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration. J Appl Physiol (2010)
Botulinum Toxin
Botulinum toxin is the most potent biological toxin known, produced by the anaerobic bacterium *Clostridium botulinum*. Despite its extreme toxicity, it has bec
BPC-157 / TB-500 Blend
BPC-157 and TB-500 (Thymosin Beta-4) are peptides commonly studied for their potential in promoting tissue repair and healing. BPC-157, a stable gastric pentade