Oxaloacetate
A key TCA cycle intermediate and gluconeogenic substrate investigated as a caloric restriction mimetic, with emerging research in longevity, neuroprotection, and metabolic modulation through its effects on the NAD+/NADH ratio.
Overview
Oxaloacetate (OAA) is a four-carbon dicarboxylic acid that occupies a uniquely central position in cellular metabolism, serving as both a tricarboxylic acid (TCA) cycle intermediate and the entry point for gluconeogenesis. In the TCA cycle, OAA condenses with acetyl-CoA (via citrate synthase) to form citrate, effectively gating the rate of oxidative metabolism. OAA is also the product of pyruvate carboxylation, connecting glycolysis to both the TCA cycle and gluconeogenic pathways. As a supplement, stabilized oxaloacetate (typically as the thermally stable anhydrous or enol-oxaloacetate form) has attracted significant interest as a caloric restriction (CR) mimetic — a compound that replicates the metabolic and longevity-associated signaling of caloric restriction without actual caloric reduction.
The caloric restriction mimetic properties of oxaloacetate are mediated primarily through its effects on the cytoplasmic NAD+/NADH ratio. OAA is reduced to malate by cytoplasmic malate dehydrogenase, consuming NADH and increasing the NAD+/NADH ratio — the same metabolic shift produced by caloric restriction. Elevated NAD+ activates sirtuins (particularly SIRT1 and SIRT3), AMPK, and PGC-1alpha, triggering downstream effects including enhanced mitochondrial biogenesis, improved autophagy, reduced mTOR signaling, and anti-inflammatory gene expression programs. In C. elegans studies, OAA supplementation extended lifespan by up to 25%, and in mouse models, it demonstrated neuroprotective effects in Alzheimer's disease by reducing neuroinflammation and glutamate excitotoxicity — OAA can transaminate with glutamate (via aspartate aminotransferase) to form aspartate, effectively functioning as a glutamate scavenger that may protect against excitotoxic neuronal damage.
Supplemental oxaloacetate is typically dosed at 100–200 mg daily in its thermally stabilized form. It is often combined with NMN or NR for complementary NAD+ elevation through both salvage pathway supplementation and NADH consumption, with resveratrol for sirtuin activation, and with berberine or metformin for AMPK-mediated metabolic optimization. Clinical data remains limited, with a small human trial showing reduced glutamate levels in glioblastoma patients and ongoing investigation in neurodegenerative conditions. Side effects are generally mild and may include transient gastrointestinal discomfort. Oxaloacetate represents a mechanistically compelling approach to CR mimicry, though human clinical evidence is still in early stages.
Mechanism of Action
Oxaloacetate (OAA) is a four-carbon dicarboxylic acid that serves as a critical intermediate in the tricarboxylic acid (TCA) cycle, where it condenses with acetyl-CoA via citrate synthase to form citrate, initiating each turn of the cycle. OAA is also the product of malate oxidation by malate dehydrogenase using NAD+ as a cofactor. This reaction links OAA directly to the cellular NAD+/NADH ratio, and supplemental OAA has been shown to increase the NAD+/NADH ratio, mimicking the metabolic state induced by caloric restriction and activating AMPK and sirtuin pathways.
A particularly notable mechanism is OAA's ability to lower brain glutamate levels through peripheral glutamate scavenging. In the blood, OAA undergoes transamination with glutamate via glutamate-oxaloacetate transaminase (GOT/aspartate aminotransferase), converting glutamate to aspartate and OAA to alpha-ketoglutarate. This creates a concentration gradient that draws excess glutamate out of the brain across the blood-brain barrier, providing neuroprotection against excitotoxicity without directly crossing into the CNS.
OAA also serves as a substrate for phosphoenolpyruvate carboxykinase (PEPCK) in gluconeogenesis, converting to phosphoenolpyruvate for glucose production. It participates in the malate-aspartate shuttle, transferring reducing equivalents from the cytosol into mitochondria. Through its roles in amino acid transamination, OAA connects to the urea cycle and amino acid synthesis pathways, making it a metabolic hub linking energy production, nitrogen metabolism, and neuroprotection.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
ME/CFS Response:: Over 25% average fatigue reduction in clinical trials, with 40.5% of patients showing enhanced response (63% reduction). Dose-Dependent:: Clinical studies show efficacy at 2,000mg daily (1,000mg twice daily), with safety established up to this level. Tolerability:: Well-tolerated in clinical trials with Alzheimer's and ME/CFS patients at therapeutic doses. Individual Variation:: Clinical trials identified 'enhanced responders' suggesting individual variability in effectiveness
- Over 25% average fatigue reduction in clinical trials, with 40.5% of patients showing enhanced response (63% reduction)
- Clinical studies show efficacy at 2,000mg daily (1,000mg twice daily), with safety established up to this level
- Well-tolerated in clinical trials with Alzheimer's and ME/CFS patients at therapeutic doses
- Clinical trials identified 'enhanced responders' suggesting individual variability in effectiveness
Safety Profile
Oxaloacetate is generally considered safe at recommended doses, with mild gastrointestinal discomfort being the most commonly reported side effect. It may affect blood glucose levels and should be used with caution by individuals with diabetes or hypoglycemia. Long-term human safety studies are limited, and it should be avoided during pregnancy and breastfeeding.
Pharmacokinetic Profile
Molecular Structure
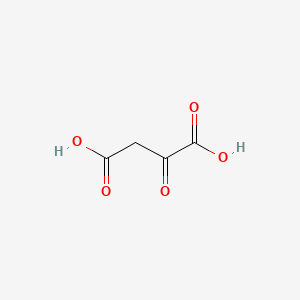
- Formula
- C4H4O5
- Weight
- 132.07 Da
- PubChem CID
- 970
- Exact Mass
- 132.0059 Da
- LogP
- -0.6
- TPSA
- 91.7 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 3
- Complexity
- 158
Identifiers (SMILES, InChI)
InChI=1S/C4H4O5/c5-2(4(8)9)1-3(6)7/h1H2,(H,6,7)(H,8,9)
KHPXUQMNIQBQEV-UHFFFAOYSA-NSafety Profile
Common Side Effects
- General Tolerability:: Clinical trials report good tolerability with no significant adverse events at tested doses
- Safety Profile:: Well-tolerated in vulnerable populations including ME/CFS and Alzheimer's patients
- No Major Concerns:: Research literature does not report serious side effects at therapeutic doses
- Limited Long-term Data:: Most studies are 1-3 months; longer-term safety data is limited
References (4)
- [2]Oxaloacetate activates brain mitochondrial biogenesis, enhances the insulin pathway, reduces inflammation and stimulates neurogenesis
→ Oxaloacetate administration in animal models enhanced brain mitochondrial biogenesis, improved insulin signaling, reduced inflammatory markers, and stimulated neurogenesis, suggesting potential neuroprotective effects.
- [3]Safety and target engagement profile of two oxaloacetate doses in Alzheimer's patients
→ A safety study in Alzheimer's patients testing 500mg and 1,000mg twice daily for one month demonstrated good tolerability and safety profile, establishing OAA as safe for human consumption in these doses.
- [4]Oxaloacetate supplementation increases lifespan in Caenorhabditis elegans through an AMPK/FOXO-dependent pathway
→ In C. elegans model organisms, oxaloacetate supplementation extended lifespan through activation of AMPK and FOXO pathways, key regulators of metabolism and longevity.
- [1]RESTORE ME: a RCT of oxaloacetate for improving fatigue in patients with myalgic encephalomyelitis/chronic fatigue syndrome
→ In a randomized, double-blind trial of 82 ME/CFS patients, 2,000mg daily oxaloacetate significantly reduced fatigue by over 25% compared to baseline, with 40.5% classified as 'enhanced responders' showing 63% fatigue reduction. Both physical and mental fatigue improved with good tolerability.
Ovagen
Ovagen is a synthetic tetrapeptide (Thr-Glu-Asp-Phe, or TEDF) classified as a bioregulatory peptide with primary effects on liver and gastrointestinal tract tis
Oxiracetam
Oxiracetam is a racetam nootropic known for its stimulating cognitive enhancement. It improves memory, focus, and logical thinking while providing mild stimulat