Octreotide
Octreotide (Sandostatin) is a synthetic cyclic octapeptide analog of somatostatin with a significantly longer half-life (~90 minutes vs ~1-3 minutes). It is a clinically established treatment for acromegaly, carcinoid syndrome, neuroendocrine tumors, and variceal bleeding, available in immediate-release and long-acting depot (LAR) formulations.
Overview
Octreotide was designed through systematic structure-activity studies of somatostatin-14, identifying the minimal pharmacophore (Phe⁷-Trp⁸-Lys⁹-Thr¹⁰) required for receptor binding and incorporating D-amino acid substitutions and C-terminal reduction to resist enzymatic degradation (Bauer et al., 1982). The resulting octapeptide binds preferentially to somatostatin receptor subtypes 2 and 5 (SSTR2 >> SSTR5 > SSTR3), with minimal activity at SSTR1 and SSTR4.
First approved by the FDA in 1988 for acromegaly and carcinoid syndrome, octreotide has since become one of the most commercially successful peptide therapeutics. The development of the long-acting release (LAR) microsphere formulation in 1998 enabled once-monthly intramuscular injection, transforming patient compliance. In 2020, an oral octreotide formulation (Mycapssa) using transient permeability enhancer (TPE) technology received FDA approval, marking a milestone in oral peptide delivery.
Mechanism of Action
Octreotide replicates the inhibitory actions of native somatostatin through preferential SSTR2/SSTR5 activation:
- SSTR2-mediated GH suppression: Octreotide's primary clinical effect in acromegaly derives from high-affinity SSTR2 binding on pituitary somatotroph cells, inhibiting adenylyl cyclase, reducing cAMP, and suppressing GH secretion. Tumor shrinkage occurs in approximately 30-50% of patients through SSTR2-mediated antiproliferative signaling (Melmed et al., 2005).
- Hormone secretion inhibition: Broadly suppresses secretion of serotonin (carcinoid), VIP (VIPomas), gastrin (gastrinomas), insulin (insulinomas), and glucagon (glucagonomas) from neuroendocrine tumor cells.
- Antiproliferative effects: SSTR2 activation engages phosphotyrosine phosphatases (SHP-1, SHP-2), inducing G1 cell cycle arrest and apoptosis in neuroendocrine tumor cells. The PROMID trial demonstrated that octreotide LAR significantly prolongs time to tumor progression in midgut NETs (Rinke et al., 2009).
- Splanchnic blood flow reduction: Inhibits release of vasodilatory peptides (glucagon, VIP) and directly reduces portal blood flow, providing efficacy in variceal bleeding.
- GI motility and secretion: Reduces gastric acid, pancreatic enzyme secretion, and intestinal fluid secretion, underlying both therapeutic effects (diarrhea control) and side effects (steatorrhea, gallstones).
Reconstitution Calculator
Octreotide
**Octreotide** (Sandostatin) is a synthetic cyclic octapeptide that mimics the p

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Acromegaly
Octreotide is a first-line medical therapy for acromegaly when surgery is not curative or not feasible. Octreotide LAR (10-40 mg IM monthly) normalizes GH and IGF-1 levels in approximately 50-70% of patients and produces clinically significant tumor shrinkage (>20% volume reduction) in 30-75% of patients depending on SSTR2 expression levels. Primary medical therapy (octreotide before surgery) has gained acceptance for debulking large macroadenomas. Predictors of response include high tumor SSTR2 expression and T2-hypointense MRI signal (Colao et al., 2011).
Carcinoid Syndrome
Octreotide effectively controls the flushing, diarrhea, and bronchoconstriction of carcinoid syndrome by suppressing serotonin and other vasoactive peptide secretion from midgut neuroendocrine tumors. Symptom control is achieved in 60-70% of patients. The LAR formulation provides consistent 28-day symptom control. Breakthrough symptoms are managed with supplemental subcutaneous octreotide (Rubin et al., 1999).
Neuroendocrine Tumor Growth Control
The landmark PROMID trial (2009) randomized patients with well-differentiated metastatic midgut NETs to octreotide LAR 30 mg monthly vs placebo, demonstrating significant prolongation of time to tumor progression (14.3 vs 6.0 months, HR 0.34) (Rinke et al., 2009). This established somatostatin analogs as antiproliferative agents, not merely symptom-control drugs, fundamentally changing NET management paradigms.
VIPomas and Other Secretory Tumors
Octreotide is the treatment of choice for VIP-secreting tumors causing watery diarrhea-hypokalemia-achlorhydria (WDHA/Verner-Morrison) syndrome. It controls diarrhea in the majority of patients and can be used as a bridge to surgery or as long-term palliation for unresectable disease. Similar efficacy is seen in glucagonomas, gastrinomas, and GRFomas (Nikou et al., 2005).
GI Bleeding
Octreotide (50 mcg IV bolus followed by 25-50 mcg/hr infusion) is standard adjunctive therapy for acute variceal bleeding in portal hypertension. It reduces portal pressure by decreasing splanchnic blood flow, and meta-analyses confirm improved bleeding control when combined with endoscopic therapy. It has largely replaced native somatostatin and vasopressin for this indication due to practical advantages and fewer side effects (Corley et al., 2001).
Safety Profile
Octreotide has a well-characterized safety profile from decades of clinical use. The most significant long-term adverse effect is gallstone formation, occurring in 15-30% of patients on chronic therapy due to reduced gallbladder contractility and altered bile composition. Other common adverse effects include:
- Gastrointestinal: Diarrhea, steatorrhea, abdominal pain, nausea, flatulence (often transient, improving over weeks)
- Metabolic: Hyperglycemia or hypoglycemia (altered insulin/glucagon balance), vitamin B12 deficiency
- Hepatobiliary: Gallstones, biliary sludge, rarely cholecystitis
- Injection site: Pain, nodules (SC); injection site reactions (LAR)
- Cardiac: Bradycardia, QT prolongation (rare)
- Endocrine: Hypothyroidism (TSH suppression, rare)
Gallstone screening by ultrasound is recommended before and during long-term therapy. Most GI side effects attenuate with continued use. The oral formulation (Mycapssa) has a similar adverse effect profile with additional transient GI symptoms related to the permeability enhancer.
Pharmacokinetic Profile
Octreotide — Pharmacokinetic Curve
Subcutaneous injection, Intramuscular (LAR depot), Oral (Mycapssa)Quick Start
- Route
- Subcutaneous injection, Intramuscular (LAR depot), Oral (Mycapssa)
Molecular Structure
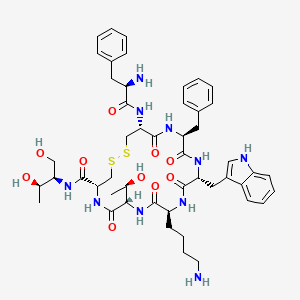
- Formula
- C₄₉H₆₆N₁₀O₁₀S₂
- Weight
- 1019.3 Da
- CAS
- 83150-76-9
- PubChem CID
- 448601
- Exact Mass
- 1018.4405 Da
- LogP
- 1
- TPSA
- 383 Ų
- H-Bond Donors
- 13
- H-Bond Acceptors
- 14
- Rotatable Bonds
- 17
- Complexity
- 1740
Identifiers (SMILES, InChI)
InChI=1S/C49H66N10O10S2/c1-28(61)39(25-60)56-48(68)41-27-71-70-26-40(57-43(63)34(51)21-30-13-5-3-6-14-30)47(67)54-37(22-31-15-7-4-8-16-31)45(65)55-38(23-32-24-52-35-18-10-9-17-33(32)35)46(66)53-36(19-11-12-20-50)44(64)59-42(29(2)62)49(69)58-41/h3-10,13-18,24,28-29,34,36-42,52,60-62H,11-12,19-23,25-27,50-51H2,1-2H3,(H,53,66)(H,54,67)(H,55,65)(H,56,68)(H,57,63)(H,58,69)(H,59,64)/t28-,29-,34-,36+,37+,38-,39-,40+,41+,42+/m1/s1
DEQANNDTNATYII-OULOTJBUSA-NResearch Protocols
subcutaneous Injection
Breakthrough symptoms are managed with supplemental subcutaneous octreotide (Rubin et al., 1999).
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Approximately | 10-40 mg | Monthly | — |
| General Research Protocol | 30 mg | Monthly | 0 months |
| Acute variceal bleeding in portal hyperte | 50 mcg, 25-50 mcg | Per protocol | — |
intramuscular Injection
The development of the long-acting release (LAR) microsphere formulation in 1998 enabled once-monthly intramuscular injection, transforming patient compliance.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Approximately | 10-40 mg | Monthly | — |
| General Research Protocol | 30 mg | Monthly | 0 months |
| Acute variceal bleeding in portal hyperte | 50 mcg, 25-50 mcg | Per protocol | — |
oral
In 2020, an oral octreotide formulation (Mycapssa) using transient permeability enhancer (TPE) technology received FDA approval, marking a milestone in oral peptide delivery. The oral formulation (Mycapssa) has a similar adverse effect profile with additional transient GI symptoms related to the per
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Approximately | 10-40 mg | Monthly | — |
| General Research Protocol | 30 mg | Monthly | 0 months |
| Acute variceal bleeding in portal hyperte | 50 mcg, 25-50 mcg | Per protocol | — |
Interactions
Peptide Interactions
It has largely replaced native somatostatin and vasopressin for this indication due to practical advantages and fewer side effects ([Corley et al.
What to Expect
What to Expect
Effects begin within hours of administration based on half-life of ~90 minutes (SC); ~28 days effective duration (LAR depot)
The LAR formulation provides consistent 28-day symptom control.
The landmark PROMID trial (2009) randomized patients with well-differentiated metastatic midgut NETs to octreotide LAR 30 mg monthly vs placebo,...
Continued use as directed
Quality Indicators
What to look for
- Well-established safety profile
- Multiple peer-reviewed studies available
- Oral administration available
Caution
- Injection site reactions reported
Frequently Asked Questions
References (9)
- [7]Corley DA et al Octreotide for acute esophageal variceal bleeding: a meta-analysis Gastroenterology (2001)
- [8]Samson SL et al Oral octreotide capsules for the treatment of acromegaly: the MPOWERED open-label study Pituitary (2020)
- [6]
- [1]Bauer W et al SMS 201-995: a very potent and selective octapeptide analogue of somatostatin with prolonged action Life Sci (1982)
- [3]
- [4]Colao A et al Octreotide LAR vs. surgery in newly diagnosed patients with acromegaly Clin Endocrinol (2011)
- [5]Rubin J et al Octreotide acetate long-acting formulation versus open-label subcutaneous octreotide acetate in malignant carcinoid syndrome J Clin Oncol (1999)
- [9]Strosberg J et al Phase 3 Trial of ¹⁷⁷Lu-Dotatate for Midgut Neuroendocrine Tumors (NETTER-1) N Engl J Med (2017)
- [2]
Obestatin
Obestatin is a 23-amino-acid amidated peptide encoded by the preproghrelin gene (GHRL) and produced primarily in the stomach, small intestine, and colon. It was
Oleic Acid
Oleic acid is a monounsaturated omega-9 fatty acid commonly found in olive oil, nuts, and various seeds. It functions as an anti-inflammatory compound that modu