R-Alpha Lipoic Acid
The naturally occurring R-enantiomer of alpha-lipoic acid with superior biological activity and bioavailability compared to the racemic mixture, functioning as a mitochondrial cofactor and universal antioxidant that regenerates other antioxidants and supports glucose metabolism.
Overview
R-alpha-lipoic acid (R-ALA, R-LA) is the naturally occurring enantiomer of alpha-lipoic acid, a dithiol compound that functions as an essential cofactor for mitochondrial alpha-keto acid dehydrogenase complexes including pyruvate dehydrogenase and alpha-ketoglutarate dehydrogenase — enzymes central to aerobic energy metabolism. While most commercial alpha-lipoic acid supplements contain a 50:50 racemic mixture of R and S enantiomers, only the R-form is synthesized endogenously and recognized by mitochondrial enzyme systems. The S-enantiomer, produced during chemical synthesis, is not only biologically inactive at the cofactor binding site but may actually compete with R-ALA for absorption and enzyme binding, potentially attenuating the therapeutic effects of the racemic mixture.
R-ALA's distinction as a "universal antioxidant" derives from several unique properties. It is both water- and fat-soluble, allowing it to function in all cellular compartments — cytoplasm, membranes, and extracellular fluid. When reduced to dihydrolipoic acid (DHLA), it forms a redox couple capable of regenerating oxidized forms of vitamin C, vitamin E, glutathione, and CoQ10, effectively recycling the body's antioxidant network. R-ALA also chelates pro-oxidant metals (iron, copper, cadmium) and activates Nrf2-dependent phase II detoxification enzymes. Its insulin-mimetic properties — enhancing glucose uptake by stimulating GLUT4 translocation to the cell membrane via AMPK activation and insulin receptor signaling — have made it a widely studied agent for diabetic neuropathy and insulin resistance, with R-ALA showing approximately twice the potency of racemic ALA in some glucose metabolism assays.
Clinical applications of R-ALA include diabetic peripheral neuropathy (where intravenous ALA is approved in Germany under the trade name Thioctacid), metabolic syndrome, non-alcoholic fatty liver disease, and neuroprotection. The bioavailability advantage of R-ALA over racemic ALA has been documented in pharmacokinetic studies showing higher peak plasma concentrations and greater AUC (area under the curve) for the R-enantiomer. Stabilized sodium R-lipoate (Na-RALA) formulations further enhance absorption by preventing the polymerization that can occur with unstabilized R-ALA. Typical supplemental dosing ranges from 100-600 mg/day. R-ALA works synergistically with acetyl-L-carnitine for mitochondrial function (the Ames cocktail for age-related mitochondrial decay), biotin (which shares a similar sulfur-containing cofactor role), and B vitamins that support energy metabolism.
Mechanism of Action
Mechanism of Action: R-Alpha Lipoic Acid
R-alpha lipoic acid (R-LA, R-ALA) is the naturally occurring R-enantiomer of thioctic acid, a sulfur-containing fatty acid that functions as an essential mitochondrial cofactor. The R-form has 40-60% greater bioavailability and substantially higher biological potency compared to the S-enantiomer or racemic mixture.
Mitochondrial Cofactor Function
R-LA is covalently bound (via lipoamide linkage) to the E2 subunit of mitochondrial multienzyme complexes: pyruvate dehydrogenase complex (PDC), α-ketoglutarate dehydrogenase (α-KGDH), branched-chain α-keto acid dehydrogenase, and the glycine cleavage system. In these complexes, the dithiolane ring undergoes reversible oxidation-reduction, shuttling acyl groups and electrons during oxidative decarboxylation. This function is essential for the citric acid cycle and aerobic ATP production.
Redox Biology and Antioxidant Network
R-LA and its reduced form dihydrolipoic acid (DHLA) form the most versatile biological redox couple (E° = -0.32V). Unique properties include: (1) Both oxidized (LA) and reduced (DHLA) forms are antioxidant-active; (2) Activity in both aqueous and lipid compartments; (3) Direct scavenging of hydroxyl radicals, hypochlorous acid, peroxynitrite, and singlet oxygen; (4) Regeneration of glutathione (by reducing GSSG and upregulating γ-glutamylcysteine ligase/GCL expression), ascorbate (by reducing dehydroascorbic acid), and α-tocopherol (by reducing tocopheroxyl radical). DHLA also chelates Fe²⁺ and Cu²⁺, preventing Fenton reaction-mediated oxidative damage.
Glucose Metabolism and Insulin Sensitization
R-LA enhances glucose disposal through insulin-dependent and insulin-independent mechanisms: (1) Activates PI3K, leading to Akt phosphorylation and GLUT4 translocation to the cell surface; (2) Reduces inhibitory IRS-1 serine phosphorylation (Ser307, Ser636/639), relieving insulin resistance; (3) Activates AMPK, promoting glucose uptake in skeletal muscle; (4) Inhibits protein tyrosine phosphatase 1B (PTP1B), prolonging insulin receptor signaling. Clinical studies in type 2 diabetes demonstrate significant improvements in insulin sensitivity and fasting glucose.
Neuroprotective Effects
R-LA crosses the blood-brain barrier and accumulates in neural tissue. It protects neurons by: maintaining mitochondrial function, reducing advanced glycation end-products (AGEs) and their receptor (RAGE) signaling, increasing nerve blood flow, and enhancing nerve conduction velocity. These mechanisms underlie its efficacy in diabetic polyneuropathy.
Anti-inflammatory Actions
R-LA inhibits NF-κB activation by maintaining IκBα levels and reducing ICAM-1 and VCAM-1 expression. It downregulates MMP-9, reduces CRP, and attenuates TNF-α signaling. These effects contribute to its benefits in metabolic syndrome, vascular inflammation, and neuroinflammation.
Research
Reported Effects
Diabetic Neuropathy:: Research and user reports consistently show strong efficacy for diabetic nerve pain, considered one of the primary evidence-based applications. Dose-Response:: Effectiveness is dose and duration-dependent, with 600mg being a common therapeutic dose and higher doses (1200mg) used initially for severe cases. Form Matters:: R-ALA (stabilized as Na-R-ALA) is preferred over racemic ALA for better bioavailability and reduced side effects, though standard ALA still shows benefits. Combination Synergy:: Works best as part of mitochondrial support stacks with CoQ10, ALCAR, and B-vitamins rather than as a standalone supplement
- Research and user reports consistently show strong efficacy for diabetic nerve pain, considered one of the primary evidence-based applications
- Effectiveness is dose and duration-dependent, with 600mg being a common therapeutic dose and higher doses (1200mg) used initially for severe cases
- R-ALA (stabilized as Na-R-ALA) is preferred over racemic ALA for better bioavailability and reduced side effects, though standard ALA still shows benefits
- Works best as part of mitochondrial support stacks with CoQ10, ALCAR, and B-vitamins rather than as a standalone supplement
Safety Profile
Safety Profile: R-Alpha Lipoic Acid
Common Side Effects
- Nausea, vomiting, and stomach discomfort, especially when taken on an empty stomach
- Skin rash, itching, or hives (more common with higher doses)
- Hypoglycemia symptoms (sweating, shakiness, dizziness) in diabetic patients due to potent glucose-lowering effects
- Unpleasant body odor (described as sulfurous) due to thiol metabolism
- Headache and mild insomnia
Serious Adverse Effects
- Hypoglycemia: Clinically significant blood glucose reduction, especially in type 1 and type 2 diabetics on insulin or oral hypoglycemics; can be severe
- Insulin autoimmune syndrome (IAS): Rare but serious condition reported primarily in Japanese patients with specific HLA genotypes; presents as severe hypoglycemia with high insulin antibody titers
- Rare severe allergic reactions including anaphylaxis
- Potential for thiamine (vitamin B1) depletion with chronic use, which can cause Wernicke-like encephalopathy in deficient individuals
- Seizures reported in children with accidental overdose; R-form may be more potent than racemic mixture
Contraindications
- Known hypersensitivity to alpha-lipoic acid
- Thiamine deficiency or risk factors for thiamine deficiency (alcoholism, malnutrition) without concurrent B1 supplementation
- Poorly controlled diabetes with frequent hypoglycemic episodes
- Pregnancy and lactation (insufficient human safety data)
- Children (risk of severe hypoglycemia and seizures with overdose)
Drug Interactions
- Insulin and oral hypoglycemics (metformin, sulfonylureas, SGLT2 inhibitors): Significant additive blood glucose-lowering effect; dose adjustments often required
- Thyroid hormones (levothyroxine): ALA may reduce conversion of T4 to T3; monitor thyroid function
- Cisplatin and other platinum-based chemotherapy: ALA may reduce chemotherapy efficacy as an antioxidant; avoid during active treatment
- Iron supplements: ALA chelates metal ions; separate dosing by at least 2 hours
- Alcohol: Increased risk of thiamine depletion and lactic acidosis; avoid concurrent heavy use
Population-Specific Considerations
- Elderly: Start with lower doses (100–200 mg/day); monitor blood glucose closely; supplement with thiamine (B1)
- Pediatric: Not recommended; potentially dangerous in overdose situations
- Diabetic patients: Close glucose monitoring essential; R-ALA is more potent than racemic ALA for glucose lowering
- Alcoholic patients: Must co-supplement with thiamine to prevent deficiency; monitor for neuropathy
- Post-surgical patients: Discontinue 1–2 weeks before surgery due to effects on blood glucose and potential for bleeding
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 600mg once daily is the most commonly reported effective dose, typically taken in the morning on an empty stomach
Molecular Structure
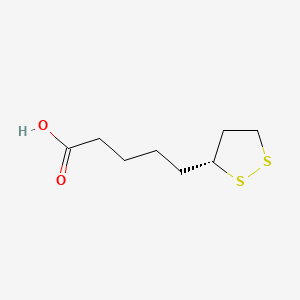
- Formula
- C8H14O2S2
- Weight
- 206.3 Da
- PubChem CID
- 6112
- Exact Mass
- 206.0435 Da
- LogP
- 1.7
- TPSA
- 87.9 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 5
- Complexity
- 150
Identifiers (SMILES, InChI)
InChI=1S/C8H14O2S2/c9-8(10)4-2-1-3-7-5-6-11-12-7/h7H,1-6H2,(H,9,10)/t7-/m1/s1
AGBQKNBQESQNJD-SSDOTTSWSA-NSafety Profile
Common Side Effects
- Heavy Metal Concerns:: Users and practitioners warn about potential redistribution of heavy metals, particularly mercury, requiring proper chelation protocol following Dr. Cutler's method
- Gastrointestinal Effects:: Heartburn and stomach discomfort reported occasionally, usually short-lived and managed by taking on empty stomach
- Mineral Depletion:: Can chelate copper and zinc, potentially causing deficiencies with long-term use; monitoring and supplementation may be needed
- Timing Challenges:: Short half-life makes consistent dosing difficult, with some protocols recommending every 3-hour dosing for chelation purposes
References (8)
- [1]Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential
→ ALA improves glucose handling, increases eNOS activity, activates Phase II detoxification via Nrf2, and lowers inflammatory markers through NF-kappa B repression. Beneficial effects are achieved with low micromolar levels, suggesting therapeutic potential beyond strict antioxidant action.
- [2]Alpha-Lipoic Acid: Biological Mechanisms and Health Benefits
→ ALA exhibits antioxidant, anti-inflammatory, and metal-chelating properties with applications in neuroprotection, metabolic syndrome, and age-related conditions. The ALA/DHLA redox couple provides ideal antioxidant characteristics by functioning in both aqueous and lipid environments.
- [3]Mitochondrial Dysfunction and Neurodegenerative Disorders: Role of Nutritional Supplementation
→ ALA is recommended as part of a combination protocol with CoQ10, B-vitamins, L-carnitine, and vitamin D for treating mitochondrial dysfunction in neurodegenerative disorders including Parkinson's, Alzheimer's, and ALS.
- [4]Alpha-lipoic acid supplementation and diabetes
→ ALA improves glycemic control and reduces polyneuropathies associated with diabetes mellitus. It directly terminates free radicals, chelates metals, and increases cytosolic glutathione and vitamin C levels while protecting against cardiovascular disease risk factors.
- [5]Alpha-lipoic acid supplement in obesity treatment: A systematic review and meta-analysis of clinical trials
→ Meta-analysis demonstrates ALA supplementation has positive effects on anthropometric indices in obesity treatment. Effects are dose and duration-dependent with optimal results seen in cases of elevated triglycerides.
- [6]α-Lipoic acid as a triglyceride-lowering nutraceutical
→ ALA shows efficacy in lowering hypertriglyceridemia, particularly when blood triglycerides are markedly elevated. It is clinically proven safe and effective against diabetic polyneuropathies with a strong safety record.
- [7]Therapeutic Benefits of Alpha-Lipoic Acid Supplementation in Diabetes Mellitus: A Narrative Review
→ Clinical trials show ALA improves glucose metabolism, reduces oxidative stress, improves endothelial dysfunction, and decreases platelet reactivity in diabetes. It inhibits NF-kB, chelates metal ions, and induces AMPK expression.
- [8]The role of diet and non-pharmacologic supplements in the treatment of chronic neuropathic pain: A systematic review
→ Systematic review identifies ALA as an important dietary intervention for managing chronic neuropathic pain. Evidence supports its efficacy as an alternative to poorly tolerated pharmacological treatments.
Quercetin Phytosome
Quercetin Phytosome is a specialized delivery system that combines the flavonoid quercetin with food-grade lecithin (phospholipids) to dramatically enhance its
RANTES / CCL5-Derived Peptides
RANTES (Regulated upon Activation, Normal T cell Expressed and Secreted), systematically renamed CCL5 (C-C motif chemokine ligand 5), is a 68-amino acid chemoki