Strontium Citrate
A bioavailable organic strontium salt that uniquely supports bone health through a dual mechanism of action — stimulating osteoblast-mediated bone formation while simultaneously inhibiting osteoclast-driven bone resorption.
Overview
Strontium citrate is an organic salt combining strontium with citric acid, providing a bioavailable form of elemental strontium for nutritional supplementation. Strontium is an alkaline earth metal that shares remarkable chemical similarity with calcium — it has nearly identical ionic radius and charge — allowing it to substitute for calcium in hydroxyapatite crystals within the bone matrix. This incorporation is not merely passive storage: strontium exerts a unique dual mechanism of action on bone metabolism that distinguishes it from virtually all other bone-active agents. It simultaneously stimulates osteoblast (bone-forming cell) proliferation and differentiation via activation of the calcium-sensing receptor (CaSR) and Wnt/β-catenin signaling, while also inhibiting osteoclast (bone-resorbing cell) differentiation and activity through RANKL suppression and OPG upregulation.
The pharmacological version of this principle, strontium ranelate (Protelos/Osseor), was approved in Europe for osteoporosis treatment based on large-scale clinical trials (SOTI and TROPOS) demonstrating 41% reduction in vertebral fracture risk and 16% reduction in non-vertebral fracture risk over three years. However, strontium ranelate was later restricted and eventually withdrawn from most markets due to cardiovascular safety concerns (increased risk of myocardial infarction and venous thromboembolism) attributed to the ranelic acid moiety, not the strontium itself. Strontium citrate has emerged as the preferred supplemental alternative, providing the bone-active strontium cation without the ranelic acid carrier. While strontium citrate lacks the same level of large-scale RCT evidence as strontium ranelate, the mechanistic basis for strontium's bone effects is independent of the anion partner.
Strontium citrate is typically dosed at 680 mg/day (providing approximately 227 mg elemental strontium), taken at bedtime on an empty stomach and separated from calcium supplementation by at least 2-4 hours, as calcium and strontium compete for intestinal absorption. An important consideration in monitoring is that strontium incorporation into bone artificially elevates dual-energy X-ray absorptiometry (DEXA) bone mineral density readings by 8-12% due to strontium's higher atomic number, which must be accounted for in clinical interpretation. Strontium citrate is often integrated into comprehensive bone health protocols alongside vitamin D3, vitamin K2, calcium, magnesium, and boron, where it addresses bone turnover dynamics that calcium and vitamin D alone cannot fully optimize.
Mechanism of Action
Strontium citrate provides strontium ions (Sr2+) that incorporate into bone mineral by partially substituting for calcium in the hydroxyapatite crystal lattice. Strontium's unique pharmacological profile derives from its dual mechanism: simultaneously promoting bone formation and reducing bone resorption. On the anabolic side, Sr2+ activates the calcium-sensing receptor (CaSR) on osteoblasts, triggering downstream signaling through ERK1/2 and p38 MAPK pathways that increase osteoblast proliferation, differentiation, and survival. CaSR activation also stimulates the Wnt/beta-catenin signaling pathway, a master regulator of osteoblastogenesis, by increasing Wnt3a and Wnt5a ligand expression and reducing the Wnt antagonist sclerostin.
On the antiresorptive side, strontium reduces osteoclast formation and activity by modulating the RANKL/OPG axis. It decreases RANKL expression by osteoblasts and stromal cells while increasing osteoprotegerin (OPG) production, shifting the balance away from osteoclastogenesis. In osteoclast precursors, strontium suppresses NFATc1 nuclear translocation, a transcription factor essential for osteoclast differentiation. It also activates the Akt/PI3K pathway in osteoblasts, promoting cell survival and reducing apoptosis.
Strontium citrate is the supplement form of strontium, distinct from strontium ranelate (the pharmaceutical form studied in clinical trials). While both deliver strontium ions, citrate is the organic acid salt available over the counter. It is important to note that strontium artificially increases bone mineral density measurements on DXA scans due to its higher atomic number compared to calcium, which must be accounted for in clinical interpretation.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Bone Health Research:: Multiple animal and human studies support bone mineral density benefits, particularly in osteoporotic populations, though most research focuses on prescription strontium ranelate rather than the citrate form. Bioavailability:: In vivo studies confirm that oral strontium citrate is absorbed and incorporated into bone tissue, with measurable accumulation over time. Limited Human Data:: While animal models show promise, large-scale human clinical trials specifically for strontium citrate supplementation are lacking compared to the prescription form. Dose-Dependent Effects:: Higher doses show greater bone incorporation but also increased risk of adverse effects, with safety thresholds established in animal studies
- Multiple animal and human studies support bone mineral density benefits, particularly in osteoporotic populations, though most research focuses on prescription strontium ranelate rather than the citrate form
- In vivo studies confirm that oral strontium citrate is absorbed and incorporated into bone tissue, with measurable accumulation over time
- While animal models show promise, large-scale human clinical trials specifically for strontium citrate supplementation are lacking compared to the prescription form
- Higher doses show greater bone incorporation but also increased risk of adverse effects, with safety thresholds established in animal studies
Safety Profile
Safety Profile: Strontium Citrate
Common Side Effects
- Gastrointestinal symptoms: nausea, diarrhea, and stomach discomfort
- Headache
- Mild skin irritation or dermatitis
- Interference with calcium metabolism markers on blood tests
Serious Adverse Effects
- Cardiovascular risk: Strontium ranelate (prescription form) was restricted in Europe due to increased risk of myocardial infarction and venous thromboembolism; while strontium citrate (supplement form) has less data, the cardiovascular concern may be class-wide
- DRESS syndrome (Drug Reaction with Eosinophilia and Systemic Symptoms): Reported with strontium ranelate; potentially relevant to strontium citrate
- DXA scan interference: Strontium accumulates in bone and artificially inflates bone mineral density readings by 8–12%, leading to overestimation of bone density improvements
- Renal impairment may lead to strontium accumulation and toxicity
Contraindications
- History of cardiovascular disease, venous thromboembolism, or stroke
- Severe renal impairment (CrCl < 30 mL/min)
- Current or recent immobilization (increased thrombosis risk)
- Known hypersensitivity to strontium compounds
- Concurrent use with calcium supplements at the same time (competitive absorption)
Drug Interactions
- Calcium supplements: Compete for absorption; separate dosing by at least 2 hours (strontium best taken at bedtime, away from calcium)
- Tetracycline and quinolone antibiotics: Strontium chelates these antibiotics, reducing absorption
- Bisphosphonates: May compete for bone binding sites; clinical significance uncertain
- Antacids (aluminum, magnesium): May reduce strontium absorption
Population-Specific Considerations
- Osteoporosis patients: Used as an alternative to strontium ranelate; efficacy data for citrate form specifically is very limited
- DXA monitoring: Clinicians must be aware that strontium elevates DXA readings; molecular weight correction factors should be applied
- Postmenopausal women: Primary target population; cardiovascular screening essential before starting
- Pregnancy/lactation: No data; not recommended
- Men with osteoporosis: Limited specific data; same cardiovascular concerns apply
Pharmacokinetic Profile
Molecular Structure
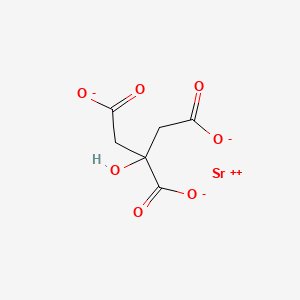
- Formula
- C6H5O7Sr-
- Weight
- 276.72 Da
- PubChem CID
- 181658
- Exact Mass
- 276.9091 Da
- TPSA
- 141 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 7
- Rotatable Bonds
- 2
- Complexity
- 211
Identifiers (SMILES, InChI)
InChI=1S/C6H8O7.Sr/c7-3(8)1-6(13,5(11)12)2-4(9)10;/h13H,1-2H2,(H,7,8)(H,9,10)(H,11,12);/q;+2/p-3
AOOMUVYEFFNMQG-UHFFFAOYSA-KSafety Profile
Common Side Effects
- Developmental Concerns:: High doses in animal studies caused bone and eye anomalies in developing fetuses, raising questions about safety during pregnancy
- Limited Safety Data:: Unlike prescription strontium ranelate which has extensive safety monitoring, over-the-counter strontium citrate lacks comprehensive long-term human safety studies
- Bone Quality Questions:: While increasing density, there are theoretical concerns about whether strontium incorporation affects bone quality versus just increasing measured density
- Interaction Potential:: May affect calcium metabolism and absorption, requiring consideration when combined with calcium supplementation
References (9)
- [1]Prenatal developmental toxicity study of strontium citrate in Sprague Dawley rats
→ High doses of strontium citrate (2267 mg/kg) showed adverse effects on fetal bone and eye development in rats, while doses up to 1360 mg/kg/day were considered safe with no observable adverse effects on prenatal development.
- [2]Accumulation of bone strontium measured by in vivo XRF in rats supplemented with strontium citrate and strontium ranelate
→ Both strontium citrate and strontium ranelate effectively accumulated in bone tissue of rats over 8 weeks, with in vivo X-ray fluorescence successfully measuring bone strontium levels in a live animal model.
- [3]Influence of Various Strontium Formulations on Bone Mineral Density in Ovariectomized Mice
→ Strontium citrate, along with ranelate and chloride forms, showed positive effects on bone mineral density and microarchitecture in an osteoporotic mouse model, supporting its potential role in bone health.
- [4]Monitoring bone strontium intake in osteoporotic females self-supplementing with strontium citrate
→ Long-term monitoring of osteoporotic women taking strontium citrate supplements showed measurable bone strontium accumulation using non-invasive X-ray fluorescence, demonstrating bioavailability and bone incorporation.
- [5]Monitoring bone strontium levels of an osteoporotic subject due to self-administration of strontium citrate: a case study
→ A single osteoporotic patient showed measurable increases in bone strontium levels within 24 hours of supplementation (680 mg Sr/day), with continued accumulation over time in both cortical and trabecular bone.
- [6]Modeling elemental strontium in human bone based on in vivo measurements in osteoporotic females
→ Kinetic modeling in osteoporotic women taking strontium citrate revealed half-lives of 508 days in cortical bone and 232 days in trabecular bone, with measurable accumulation patterns over four years.
- [7]Effect of strontium citrate on bone consolidation during mandibular distraction osteogenesis
→ Strontium citrate supplementation enhanced new bone formation and healing during mandibular distraction osteogenesis in a rabbit model, suggesting potential benefits for bone healing applications.
- [8]Dietary strontium increases bone mineral density in intact zebrafish
→ Dietary strontium citrate supplementation significantly increased whole-body and spinal column bone mineral density in zebrafish, with strontium incorporating into bone tissue in a dose-dependent manner.
- [9]Strontium increasing calcium accessibility from calcium citrate
→ Strontium chloride added to calcium citrate suspensions increased calcium ion activity through thermodynamic mechanisms, potentially enhancing calcium bioavailability and bone mineralization processes.
Stinging Nettle
Stinging Nettle (Urtica dioica) is a perennial herbaceous plant traditionally used as a medicinal vegetable for treating allergies, urinary tract issues, inflam
Substance P
**Substance P** is an undecapeptide (11 amino acids) belonging to the tachykinin neuropeptide family, first identified by Ulf von Euler and John Gaddum in 1931