Motilin
Motilin is a 22-amino-acid peptide hormone produced by Mo cells of the duodenal and jejunal mucosa that regulates interdigestive gastrointestinal motility through initiation of the migrating motor complex (MMC). It acts through the motilin receptor (GPR38) and is the physiological basis for erythromycin's prokinetic effects, making it a key target in gastroparesis and motility disorder research.
Overview
Motilin was first isolated from porcine duodenal mucosa in 1971 by Brown et al. and subsequently characterized as a key regulator of interdigestive gastrointestinal motility. During fasting, motilin is released in a cyclical pattern with a periodicity of approximately 90-120 minutes, corresponding to Phase III of the MMC — the intense burst of propagating contractions that sweeps aborally from the stomach through the small intestine. This cyclical release is regulated by a complex interplay between enteric nervous system activity, duodenal pH, bile acid exposure, and neural inputs.
The MMC serves critical housekeeping functions: it clears undigested food residues, prevents bacterial overgrowth in the upper small intestine, and propels bile and pancreatic secretions toward the colon. Disruption of the MMC is associated with small intestinal bacterial overgrowth (SIBO), gastroparesis, and functional dyspepsia. Motilin's central role in MMC regulation has made the motilin receptor a primary target for prokinetic drug development.
The discovery that erythromycin, a widely used macrolide antibiotic, stimulates gastrointestinal motility through direct agonism of the motilin receptor was a landmark finding. This explained the well-known "side effect" of GI disturbance with erythromycin therapy and opened a new therapeutic avenue. Erythromycin at sub-antimicrobial doses (40-250 mg) remains the most effective prokinetic available for accelerating gastric emptying, though its use is limited by tachyphylaxis (receptor desensitization within days), QT prolongation risk, antibiotic resistance concerns, and drug interactions.
Motilin biology is notably species-specific. Rodents lack a functional motilin gene and do not have motilin-driven MMC activity, which has hampered preclinical research and necessitated the use of dogs, rabbits, or primates for motilin studies. This species limitation has slowed development of motilin-based therapeutics compared to other GI peptide targets.
Mechanism of Action
Motilin exerts its prokinetic effects through binding to the motilin receptor (MLNR, also designated GPR38), a Gq/11-coupled GPCR:
Phase III MMC Initiation: Motilin is released from Mo cells in cyclical peaks that correspond to the initiation of Phase III contractions of the MMC. The release is driven by a combination of enteric neural reflexes, luminal stimuli (alkaline pH, bile acids), and intrinsic oscillatory activity of Mo cells. Plasma motilin peaks precede the onset of Phase III gastric contractions by several minutes, consistent with a hormonal trigger role. Vantrappen G et al. (1979) — J. Clin. Invest. 63, 1268-1276.
Gastric Smooth Muscle Contraction: Motilin receptor activation on gastric antral smooth muscle cells triggers Gq-mediated phospholipase C activation, IP3-dependent calcium release from the sarcoplasmic reticulum, and calcium/calmodulin-dependent smooth muscle contraction. This pathway directly stimulates antral contractions that initiate gastric emptying of residual contents during fasting.
Enteric Nervous System: Motilin receptors are expressed on cholinergic neurons of the myenteric plexus. Motilin activates these neurons to release acetylcholine, which amplifies the contractile response through muscarinic receptors on smooth muscle. This neural component is important for the coordinated propagation of Phase III contractions — blocking cholinergic transmission with atropine attenuates but does not abolish motilin's motor effects.
Erythromycin as Motilin Agonist: Erythromycin and related 14-membered-ring macrolides directly bind and activate the motilin receptor. The binding site on the motilin receptor for erythromycin overlaps with but is not identical to the motilin binding site, allowing for structural optimization of motilide compounds. Erythromycin's prokinetic potency is comparable to motilin itself at sub-antimicrobial doses, but the therapeutic window is limited by rapid tachyphylaxis (downregulation of motilin receptor expression within 3-5 days of continuous exposure). Peeters TL (1993) — Gastroenterology 105, 1886-1899.
Receptor Desensitization: A major challenge for motilin-based therapeutics is rapid receptor desensitization. Continuous or repeated agonist exposure leads to motilin receptor internalization and downregulation, resulting in loss of prokinetic efficacy (tachyphylaxis). This is observed clinically with erythromycin, where prokinetic effects diminish after 3-4 weeks of continuous use. Strategies to overcome tachyphylaxis include intermittent dosing, partial agonists, and allosteric modulators.
Enterochromaffin Cell Effects: Motilin receptors on enterochromaffin cells stimulate serotonin (5-HT) release, which contributes to the prokinetic response through 5-HT3 and 5-HT4 receptor activation on enteric neurons and smooth muscle. This serotonergic component links motilin signaling to the broader enteric serotonin system.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
The Migrating Motor Complex
The MMC is a cyclical pattern of gastrointestinal motor activity that occurs during fasting and is interrupted by feeding. It consists of four phases: Phase I (quiescence, ~40-60% of cycle), Phase II (irregular contractions, ~20-30%), Phase III (intense rhythmic contractions propagating from stomach to terminal ileum, ~5-10 minutes), and Phase IV (brief transition back to Phase I). Motilin is the hormonal trigger for gastric Phase III, while the intestinal component of Phase III may be more dependent on intrinsic enteric neural pacemaker activity. The MMC cycle length (~90-120 minutes) corresponds to the periodicity of motilin release from Mo cells. Disruption of the MMC is clinically associated with SIBO, gastroparesis, and functional GI symptoms. Deloose E et al. (2012) — Neurogastroenterol. Motil. 24, 420-432.
Gastroparesis and Prokinetic Development
Gastroparesis — delayed gastric emptying without mechanical obstruction — affects approximately 2% of the population and has limited treatment options. Motilin receptor agonism represents one of the most effective pharmacological approaches to accelerate gastric emptying. Erythromycin at prokinetic doses (40-250 mg IV or oral) produces greater acceleration of gastric emptying than metoclopramide or domperidone, but its use is limited by tachyphylaxis, antibiotic properties, and cardiac risk (QT prolongation). This has driven extensive development of non-antibiotic motilin receptor agonists (motilides). Camilleri M et al. (2013) — Gastroenterology 144, 218-238.
Motilide Drug Development
The term "motilide" describes macrolide compounds that retain motilin receptor agonist activity while lacking antibiotic properties. Several motilides have entered clinical development:
Mitemcinal (GM-611): An erythromycin-derived motilide that reached Phase 3 trials for diabetic gastroparesis. While it demonstrated acceleration of gastric emptying, Phase 3 results were disappointing, with modest symptom improvements that did not consistently reach statistical significance. Development was discontinued.
Camicinal (GSK962040): A small-molecule motilin receptor agonist (not a macrolide) developed by GlaxoSmithKline. It demonstrated dose-dependent acceleration of gastric emptying in Phase 1/2 studies and was evaluated for gastroparesis, critical illness gastroparesis, and Parkinson's disease-associated gastroparesis. Development was paused.
ABT-229: An erythromycin derivative that paradoxically worsened symptoms in functional dyspepsia trials, highlighting the dissociation between gastric emptying acceleration and symptom improvement — a recurring challenge in gastroparesis drug development.
Motilin and Small Intestinal Bacterial Overgrowth (SIBO)
The MMC's housekeeping function is critical for preventing bacterial colonization of the upper small intestine. Impaired MMC activity (from conditions such as diabetes, scleroderma, or post-vagotomy states) is a major risk factor for SIBO. Motilin receptor agonists, including erythromycin at sub-antimicrobial doses, can restore MMC-like activity and reduce bacterial counts in the small intestine. This "motility as antimicrobial defense" concept has been explored as a non-antibiotic approach to managing recurrent SIBO.
Species Specificity in Motilin Research
The absence of functional motilin and motilin receptor genes in rodents presents a significant translational challenge. Mice and rats lack both the motilin peptide and the motilin receptor, and their interdigestive motility patterns differ from those of humans and dogs. Preclinical motilin research has relied on dogs (which have robust motilin-driven MMC activity), rabbits (which express functional motilin receptors), and non-human primates. This species limitation has slowed drug development and increased costs compared to GI peptide targets that can be studied in rodent models.
Motilin in Critical Illness
Critically ill patients frequently develop gastroparesis and intestinal dysmotility, leading to feeding intolerance and nutritional compromise. Erythromycin is widely used as a prokinetic in ICU settings to improve tolerance of enteral feeding. Studies have demonstrated that erythromycin (70-250 mg IV every 6-8 hours) significantly improves gastric emptying and feeding volumes in critically ill patients, though tachyphylaxis develops within 3-7 days. Combination with metoclopramide may extend the duration of prokinetic efficacy.
Safety Profile
Motilin itself has not been developed as a therapeutic agent due to its very short half-life (~5 minutes) and peptide nature. Safety considerations primarily apply to motilin receptor agonists, particularly erythromycin used at prokinetic doses. Erythromycin's prokinetic adverse effects include abdominal cramping, nausea (paradoxically), and diarrhea at higher doses. The most significant safety concerns are QT prolongation (dose-dependent, increased risk with co-administration of CYP3A4 inhibitors), promotion of antibiotic resistance when used at antimicrobial doses, and drug interactions through CYP3A4 inhibition. Tachyphylaxis limits long-term efficacy and necessitates drug holidays or intermittent dosing strategies. Non-antibiotic motilides in clinical development have generally been well tolerated but have not overcome the tachyphylaxis challenge. Motilin receptor agonists should be used with caution in patients receiving QT-prolonging medications, those with hepatic impairment (reduced erythromycin clearance), and in settings where antibiotic stewardship is a concern.
Pharmacokinetic Profile
Motilin — Pharmacokinetic Curve
Intravenous (research), oral motilides (clinical)Quick Start
- Route
- Intravenous (research), oral motilides (clinical)
Molecular Structure
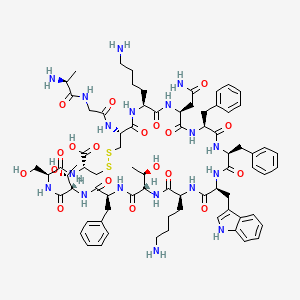
- Formula
- C121H187N33O33S1
- Weight
- 2698.1 Da
- CAS
- 9072-41-7
- PubChem CID
- 16129706
- Exact Mass
- 1636.7167 Da
- LogP
- -3.1
- TPSA
- 664 Ų
- H-Bond Donors
- 22
- H-Bond Acceptors
- 24
- Rotatable Bonds
- 26
- Complexity
- 3240
Identifiers (SMILES, InChI)
InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42+,43+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,62-,63-/m0/s1
NHXLMOGPVYXJNR-ATOGVRKGSA-NResearch Indications
Gastrointestinal Motility
Motilin receptor agonists (erythromycin, azithromycin) are used off-label for gastroparesis, stimulating gastric contractions and reducing nausea/vomiting. Novel non-motilide agonists like GSK962040 are in clinical trials to avoid tachyphylaxis.
Motilin is the primary hormone driving the migrating motor complex (MMC), occurring every 1.5-2 hours during fasting. It facilitates transport of undigested food and prevents bacterial migration from colon to small intestine.
Despite promising gastric emptying acceleration in healthy volunteers, motilide ABT-229 failed to improve symptoms in functional dyspepsia patients. Novel selective motilin receptor agonists continue in development.
Clinical Procedures
Motilin receptor agonists are used in acute care to accelerate gastric emptying before rapid sequence intubation or endoscopy, reducing aspiration risk.
Research Protocols
oral
Erythromycin at prokinetic doses (40-250 mg IV or oral) produces greater acceleration of gastric emptying than metoclopramide or domperidone, but its use is limited by tachyphylaxis, antibiotic properties, and cardiac risk (QT prolongation).
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| At sub-antimicrobial doses | 40-250 mg | Per protocol | — |
| Critically ill patients | 70-250 mg | Per protocol | 3-7 days |
intravenous Injection
Administered via intravenous injection.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| At sub-antimicrobial doses | 40-250 mg | Per protocol | — |
| Critically ill patients | 70-250 mg | Per protocol | 3-7 days |
Interactions
Peptide Interactions
Motilin and ghrelin activate structurally related receptors (GPR38 and GHSR1a respectively) with complementary effects on gastric motility. Motilin primarily drives phase III migrating motor complex activity while ghrelin enhances gastric emptying and appetite. Combined action may provide comprehensive GI motility support (Sanger, 2008).
Erythromycin is a motilin receptor agonist that mimics motilin action on GI smooth muscle. Co-administration with exogenous motilin could produce additive prokinetic effects, increasing risk of abdominal cramping, diarrhea, and excessive GI motility. Dose adjustment of either agent may be required (Peeters et al., 1992).
What to Expect
What to Expect
Rapid onset expected; half-life of ~5 minutes indicates fast-acting pharmacokinetics
Studies have demonstrated that erythromycin (70-250 mg IV every 6-8 hours) significantly improves gastric emptying and feeding volumes in critically...
Erythromycin's prokinetic potency is comparable to motilin itself at sub-antimicrobial doses, but the therapeutic window is limited by rapid...
This is observed clinically with erythromycin, where prokinetic effects diminish after 3-4 weeks of continuous use.
Continued use as directed
Quality Indicators
What to look for
- Phase 3 clinical trial data available
- Well-established safety profile
Caution
- Short half-life may require frequent dosing
Frequently Asked Questions
References (9)
- [2]Vantrappen, G. et al Motilin and the interdigestive migrating motor complex in man J. Clin. Invest. (1979)
- [1]Brown, J. C. et al A gastric inhibitory polypeptide. I. The amino acid composition and the tryptic peptides Can. J. Biochem. (1971)
- [3]
- [5]Deloose, E. et al The migrating motor complex: control mechanisms and its role in health and disease Neurogastroenterol. Motil. (2012)
- [6]
- [4]Camilleri, M. et al Gastroparesis Gastroenterology (2013)
- [8]Broad J et al — The motilin receptor: challenges and opportunities for drug development Br. J. Pharmacol. (2023)
- [9]Tack J et al — Gastroparesis: new therapeutic options and future directions Gut (2023)
- [7]Camilleri M et al — Gastrointestinal motility disorders: management that optimizes nutritional and pharmacologic treatment JPEN J. Parenter. Enteral Nutr. (2023)
Modified GRF 1-29
Modified GRF 1-29 (also known as Mod GRF or CJC-1295 without DAC) is a synthetic analog of growth hormone-releasing hormone (GHRH) consisting of the first 29 am
MOTS-c
MOTS-c is a mitochondrial-derived peptide (MDP) composed of 16 amino acids, encoded by the 12S rRNA gene of the mitochondrial genome. It plays a critical role i