DSIP
DSIP is a naturally occurring nonapeptide neuromodulator that promotes delta-wave sleep patterns and modulates stress responses. It interacts with multiple neurotransmitter systems including opioid, serotonergic, and GABAergic pathways.
Overview
DSIP was originally discovered by Schoenenberger and Monnier in 1977 when they isolated a substance from the cerebral venous blood of rabbits in an electrically induced sleep state. The peptide was found to promote spindle and delta EEG activity when infused into recipient rabbits, leading to its designation as a "sleep-inducing" peptide. However, subsequent research revealed that DSIP's biological role extends far beyond simple sleep induction.
DSIP is found in both the central and peripheral nervous system, as well as in various peripheral tissues and blood plasma. Its widespread distribution and interaction with multiple receptor systems suggest it functions as a broad-spectrum neuromodulator rather than a dedicated sleep factor. Research has demonstrated roles in stress tolerance, pain modulation, endocrine regulation, and even antioxidant defense mechanisms.
The peptide exhibits an unusual resistance to degradation by aminopeptidases due to its N-terminal tryptophan residue, though it is still rapidly cleared from circulation. DSIP has been detected in free form and bound to carrier proteins in human blood, suggesting a reservoir mechanism for sustained biological activity.
Mechanism of Action
DSIP's mechanism of action is complex and not fully elucidated, reflecting its role as a multimodal neuromodulator rather than a conventional receptor agonist. Several key pathways have been identified:
Sleep regulation: DSIP promotes delta-wave (0.5-4 Hz) EEG activity associated with deep non-REM sleep stages 3 and 4. It appears to act on hypothalamic sleep-wake centers and modulates the balance between sleep-promoting and arousal systems. DSIP influences the firing patterns of neurons in the ventrolateral preoptic area and interacts with GABAergic signaling to facilitate sleep onset and maintenance. Studies have shown that DSIP normalizes disturbed sleep architecture rather than simply inducing sedation, making it distinct from conventional hypnotics (Schneider-Helmert & Schoenenberger, 1983).
Opioid system interaction: DSIP modulates opioid receptor activity and influences endogenous opioid peptide levels. It has been shown to alter the binding characteristics of opioid receptors and may act as an indirect modulator of enkephalin and endorphin systems. This interaction underlies both its analgesic properties and its potential utility in opioid withdrawal management (Pollard & Bhatt, 1986).
Stress modulation: DSIP exhibits stress-protective properties by modulating the hypothalamic-pituitary-adrenal (HPA) axis. It has been shown to limit ACTH and cortisol release under stress conditions while preserving basal secretion, suggesting a stabilizing rather than suppressive effect on the stress response (Sudakov et al., 1995).
Antioxidant effects: DSIP has demonstrated the ability to reduce lipid peroxidation and modulate antioxidant enzyme activity, suggesting a neuroprotective component to its biological activity.
Reconstitution Calculator
DSIP
Delta Sleep-Inducing Peptide (DSIP) is a naturally occurring nonapeptide first i

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Ongoing & Future Research
- Development of metabolically stabilized DSIP analogs with extended half-life for practical therapeutic use (Borodin et al., DOI: 10.1134/S1990519X23700529)
- Investigation of DSIP analogs in PTSD-related sleep disturbance
- Research into DSIP for substance use disorder management (opioid, alcohol, benzodiazepine withdrawal)
- Exploration of DSIP in age-related sleep architecture changes, where slow-wave sleep progressively diminishes
- Investigation of combination protocols with circadian-active peptides (Epithalon, melatonin) for comprehensive sleep optimization
- Research into DSIP's role in hibernation-like neuroprotective states (Kharlamova et al., DOI: 10.1134/S0012496615060058)
- Interest in intranasal DSIP formulations for improved practicality over IV administration
Stress and Neuroendocrine Modulation
Animal studies have demonstrated that DSIP administration prior to stress exposure significantly attenuates the physiological stress response. Sudakov and colleagues showed that DSIP reduced stress-induced ulceration, normalized catecholamine levels, and preserved immune function in rodent models of emotional stress (Sudakov et al., 1995). The peptide's stress-protective effects appear to involve stabilization of the HPA axis and modulation of monoaminergic neurotransmission.
Opioid Withdrawal and Chronic Pain
DSIP has shown promise in managing opioid and alcohol withdrawal symptoms. Clinical observations in human subjects undergoing opiate withdrawal reported that DSIP administration reduced withdrawal severity scores, improved sleep quality, and decreased craving intensity. In chronic pain research, DSIP demonstrated analgesic properties that appear to be mediated through modulation of endogenous opioid pathways rather than direct opioid receptor agonism (Dick et al., 1984).
Circadian Rhythm Effects
DSIP exhibits circadian variation in its own plasma levels and influences circadian rhythmicity of various physiological parameters. Research has shown that DSIP can modify the circadian pattern of body temperature, cortisol secretion, and sleep propensity. Graf and Kastin demonstrated that DSIP crosses the blood-brain barrier via a saturable transport mechanism, with transport efficiency varying across the circadian cycle (Graf & Kastin, 1986).
Neuroprotection
DSIP has demonstrated neuroprotective effects in experimental models of oxidative stress and ischemia. The peptide reduced markers of lipid peroxidation and preserved mitochondrial function in neuronal cell cultures exposed to oxidative insult. Khvatova and colleagues reported that DSIP's antioxidant properties involve both direct free radical scavenging and upregulation of endogenous antioxidant enzymes (Khvatova et al., 2003).
Neurological/Immunological Mechanisms
Sleep architecture modulation:
- DSIP promotes slow-wave sleep (stages 3-4, delta waves 0.5-4 Hz) through action on hypothalamic sleep-wake centers
- Modulates the ventrolateral preoptic area (VLPO) — the primary sleep-promoting nucleus
- Enhances GABAergic inhibition of wake-promoting neurons (histaminergic tuberomammillary nucleus, orexinergic lateral hypothalamus)
- Unlike benzodiazepines, DSIP does not suppress REM sleep or alter sleep spindle activity
- Normalizes disturbed sleep architecture rather than inducing sedation — a key distinction from conventional hypnotics (Schneider-Helmert, PMID: 6316133)
Opioid system modulation:
- Modulates mu-opioid receptor binding characteristics without acting as a direct agonist or antagonist
- Influences enkephalin and endorphin peptide levels in the hypothalamus and periaqueductal gray
- Proposed mechanism: DSIP stabilizes endogenous opioid tone, reducing both hyperexcitability (withdrawal) and hypofunction (chronic pain)
- This modulatory action explains its efficacy in both opioid withdrawal and chronic pain without causing dependence
HPA axis and stress response:
- Limits stress-induced ACTH and cortisol release while preserving basal HPA axis function (Sudakov et al., PMID: 8553547)
- Stabilizes catecholamine (norepinephrine, epinephrine) levels during emotional stress
- Reduces stress-induced gastric ulceration, myocardial damage, and immune suppression in rodent models
- Mechanism: dampening of CRH (corticotropin-releasing hormone) release from the paraventricular nucleus under stress conditions
Antioxidant and neuroprotective pathways:
- Reduces lipid peroxidation products (MDA/TBARS) in brain tissue (Khvatova et al., PMID: 12817700)
- Modulates superoxide dismutase (SOD) and catalase activity
- Protects mitochondrial membrane potential under oxidative stress conditions
- Mitigates ethanol-induced protein oxidation in brain tissue (Lysenko & Dovedova, PMID: 25457755)
Sleep Regulation
DSIP has been studied extensively for its effects on human sleep architecture. In clinical studies involving patients with chronic insomnia, intravenous DSIP administration improved sleep efficiency and increased the proportion of slow-wave sleep without producing morning sedation or rebound insomnia. Schneider-Helmert demonstrated that DSIP normalized disturbed sleep patterns in chronic insomniacs over multi-day treatment protocols, with effects persisting beyond the treatment period (Schneider-Helmert, 1984). Unlike benzodiazepines, DSIP did not suppress REM sleep or produce tolerance with repeated administration.
Safety Profile
DSIP has demonstrated a favorable safety profile in both animal studies and limited human clinical investigations. Key observations include:
- No sedation or cognitive impairment: Unlike benzodiazepines and other hypnotics, DSIP does not produce daytime drowsiness, psychomotor impairment, or cognitive deficits at therapeutic doses.
- No tolerance or dependence: Repeated administration in clinical studies did not produce tolerance to sleep-promoting effects or withdrawal symptoms upon cessation.
- No respiratory depression: DSIP does not suppress respiratory drive, distinguishing it from opioid-based analgesics and sedatives.
- Minimal side effects: The most commonly reported effects in clinical studies were mild and transient, including occasional headache and flushing at injection sites.
- Short half-life considerations: The rapid peripheral degradation of DSIP (half-life ~15-25 minutes) limits its practical utility as a therapeutic agent, motivating research into stabilized analogs and alternative delivery routes.
The limited scope of controlled human clinical trials means that long-term safety data remain sparse. DSIP is not currently approved for clinical use by any major regulatory authority.
Pharmacokinetic Profile
DSIP — Pharmacokinetic Curve
Intravenous / Intranasal / SubcutaneousQuick Start
- Typical Dose
- 100-300mcg
- Frequency
- 1x daily
- Route
- Intravenous / Intranasal / Subcutaneous
- Cycle Length
- 5-10 days
- Storage
- 2-8°C refrigerated
Molecular Structure
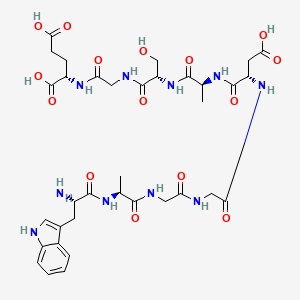
- Formula
- C₃₅H₄₈N₁₀O₁₅
- Weight
- 848.81 Da
- Length
- 9 amino acids
- CAS
- 62568-57-4
- PubChem CID
- 68816
- Exact Mass
- 848.3301 Da
- LogP
- -7.1
- TPSA
- 407 Ų
- H-Bond Donors
- 14
- H-Bond Acceptors
- 16
- Rotatable Bonds
- 25
- Complexity
- 1610
Identifiers (SMILES, InChI)
InChI=1S/C35H48N10O15/c1-16(41-32(56)20(36)9-18-11-37-21-6-4-3-5-19(18)21)30(54)39-12-25(47)38-13-26(48)44-23(10-29(52)53)34(58)42-17(2)31(55)45-24(15-46)33(57)40-14-27(49)43-22(35(59)60)7-8-28(50)51/h3-6,11,16-17,20,22-24,37,46H,7-10,12-15,36H2,1-2H3,(H,38,47)(H,39,54)(H,40,57)(H,41,56)(H,42,58)(H,43,49)(H,44,48)(H,45,55)(H,50,51)(H,52,53)(H,59,60)/t16-,17-,20-,22-,23-,24-/m0/s1
ZRZROXNBKJAOKB-GFVHOAGBSA-NResearch Indications
Sleep
Promotes delta wave sleep, the most restorative sleep phase for physical recovery.
Reduces sleep latency and nocturnal awakenings without morning grogginess.
Supports natural sleep cycles rather than forcing sedation like traditional sleep aids.
Mood
Helps regulate cortisol and stress response through HPA axis modulation.
Supports emotional balance without sedation.
May help reduce anxiety through neuromodulatory effects.
Pain & Recovery
Significantly reduced pain levels in 6 of 7 patients with chronic pain conditions.
Helps manage withdrawal symptoms in alcohol and opioid-dependent patients.
Supports post-training recovery through enhanced sleep quality.
Research Protocols
intranasal Injection
Intranasal spray offers convenient administration with limited stability data.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Sleep induction | 200-300mcg | Once at bedtime | —(Route: Nasal spray) |
| Stress relief | 150-200mcg | Twice daily | —(Route: Nasal spray) |
| Neuroprotection | 300-400mcg | Daily | —(Route: Nasal spray) |
Reconstitution Guide (mg vial + mL BAC water)
- Reconstitute DSIP to 2mg/mL concentration
- Transfer to sterile nasal spray bottle
- Prime pump to ensure consistent delivery
- Each spray should deliver ~100mcg (0.05mL)
- Test spray volume before use
- Label with concentration and date
- Use within 10 days
subcutaneous Injection
Sleep-promoting peptide administered subcutaneously in the evening before bedtime.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Week 1 | 100 mcg | Once daily | Week 1(Evening/pre-bedtime) |
| Week 2 | 150 mcg | Once daily | Week 2 |
| Week 3 | 200 mcg | Once daily | Week 3 |
| Standard dose | 250-300 mcg | Once daily | Weeks 4-8(Cycle: 4-8 weeks. Optional extension to 12 weeks with periodic breaks.) |
Reconstitution Guide (5mg vial + 3mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 3.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 1.67 mg/mL
- For 100 mcg dose: draw 6 units (0.06 mL)
- For 200 mcg dose: draw 12 units (0.12 mL)
- For 300 mcg dose: draw 18 units (0.18 mL)
- Store reconstituted vial refrigerated at 2-8°C
Interactions
Peptide Interactions
Both regulate sleep-wake cycles; complementary benefits.
Complementary anxiolytic effects.
Both support circadian regulation and sleep quality.
Different mechanisms; can optimize 24-hour function.
Both are Russian-researched peptides with sleep-modulating properties. Epithalon restores melatonin secretion patterns; DSIP promotes delta-wave sleep architecture. Complementary mechanisms for age-related sleep deterioration: Epithalon addresses circadian rhythm (melatonin), DSIP addresses sleep depth (delta waves).
What to Expect
What to Expect
Noticeable sleep pressure and easier sleep onset
Deeper sleep, fewer awakenings, vivid dreams possible
Improved morning refreshment, stable daytime energy
Cumulative stress reduction, mood improvements
Benefits may persist 1-2 weeks after stopping
Safety Profile
Common Side Effects
- Generally well-tolerated with minimal side effects
- Mild drowsiness or dizziness initially (some users)
- Occasional headaches in sensitive individuals
- No tolerance or dependence reported in studies
Contraindications
- Avoid driving until effects are known
- Pregnancy or breastfeeding
Discontinue If
- Excessive daytime sedation
- Paradoxical insomnia or agitation
- Persistent headaches
- Any allergic reactions
- Mood changes or depression
- Unusual dreams causing distress
Quality Indicators
What to look for
- White lyophilized powder before reconstitution
- Clear and colorless solution when properly mixed
- Reputable source with third-party testing
Caution
- Short stability period - use within 14 days when reconstituted
- Protect from light exposure and temperature fluctuations
- Limited nasal formulation stability (use within 10 days)
Red flags
- Yellow or cloudy solution indicates degradation or contamination
- Discolored powder
Frequently Asked Questions
References (20)
- [8]Schoenenberger GA, Monnier M Characterization of a delta-electroencephalogram sleep-inducing peptide Proc Natl Acad Sci U S A (1977)
- [3]Alcohol and Opioid Withdrawal Study (1984)
- [4]Sleep Pressure Induction Study (1983)
- [5]Stroke Recovery in Rats (2021)
- [6]40-Year Stress-Protective Effects Review
- [7]Intranasal Delivery and Bioavailability
- [17]Kharlamova AS et al The role of DSIP in regulation of adaptive brain mechanisms Dokl Biol Sci (2015)
- [20]
- [15]Khvatova EM et al Free radical status in tissues of rats during combined action of DSIP and oxidative stress Bull Exp Biol Med (2003)
- [16]Lysenko AV, Dovedova EL Effect of delta sleep-inducing peptide on oxidative modification of proteins in rat brain Bull Exp Biol Med (2014)
- [1]
- [2]Chronic Pain Pilot Study (1984)
- [9]Schneider-Helmert D, Schoenenberger GA Effects of DSIP in man. Multifunctional psychophysiological properties Neuropsychobiology (1983)
- [11]Dick P et al DSIP in the treatment of withdrawal syndromes from alcohol and opiates Eur Neurol (1984)
- [12]
- [13]
- [14]Sudakov KV et al Delta sleep-inducing peptide sequelae in the mechanisms of resistance to emotional stress Ann N Y Acad Sci (1995)
- [10]Schneider-Helmert D Clinical evaluation of DSIP Eur Neurol (1984)
- [18]Khvatova et al — Free radical status in tissues of rats during combined action of DSIP and oxidative stress — Foundational work on DSIP's antioxidant mechanisms demonstrating significant reduction in lipid peroxidation products across multiple tissue types (2003)
- [19]Lysenko & Dovedova — The effect of delta sleep-inducing peptide on oxidative modification of proteins in rat brain during acute and chronic exposure to ethanol — Demonstrated that DSIP mitigated ethanol-induced protein oxidation in brain tissue, supporting its neuroprotective potential in substance use contexts (2014)
D-Ribose
D-Ribose is a five-carbon sugar that serves as a fundamental building block for adenosine triphosphate (ATP), the primary energy molecule in cells. It works via
Dulaglutide (Trulicity)
Dulaglutide (brand name Trulicity) is a once-weekly injectable glucagon-like peptide-1 (GLP-1) receptor agonist used primarily for treating type 2 diabetes mell