Benfotiamine
Benfotiamine is a synthetic S-acyl derivative of thiamine with enhanced oral bioavailability, studied for its role in preventing complications associated with advanced glycation end-products.
Overview
Benfotiamine is a lipid-soluble synthetic derivative of thiamine (vitamin B1) that belongs to the class of allithiamines, originally identified in roasted garlic. Unlike water-soluble thiamine, benfotiamine achieves significantly higher intracellular thiamine diphosphate levels following oral administration, owing to its improved intestinal absorption and bioavailability. Once absorbed, it is dephosphorylated to S-benzoylthiamine, which enters cells passively before being converted to thiamine and subsequently to the active coenzyme thiamine pyrophosphate (TPP).
The primary therapeutic interest in benfotiamine centers on its ability to activate transketolase, a key enzyme in the pentose phosphate pathway. By diverting excess glycolytic intermediates such as fructose-6-phosphate and glyceraldehyde-3-phosphate into the pentose phosphate pathway, benfotiamine reduces the formation of advanced glycation end-products (AGEs) and mitigates activation of several pathways implicated in diabetic complications, including the hexosamine, protein kinase C, and polyol pathways. Preclinical studies have demonstrated neuroprotective, retinal-protective, and vascular-protective effects in models of diabetes.
Clinical trials have explored benfotiamine for diabetic neuropathy, with some studies reporting improvements in neuropathy symptom scores and nerve conduction parameters. It has also been investigated for its potential to reduce AGE-mediated damage following high-AGE meals. Benfotiamine is approved as a pharmaceutical product for neuropathy in several countries and is available as a dietary supplement in others. It is generally well-tolerated, with adverse effects being rare and typically mild. Ongoing research continues to investigate its applications in neurodegenerative diseases and age-related conditions where AGE accumulation plays a pathological role.
Mechanism of Action
Lipid-Soluble Thiamine Prodrug
Benfotiamine (S-benzoylthiamine O-monophosphate) is a synthetic, lipid-soluble prodrug of thiamine (vitamin B1) with 5-fold greater bioavailability than thiamine hydrochloride. Unlike allithiamine, benfotiamine is absorbed via the intestinal alkaline phosphatase-mediated dephosphorylation pathway, generating S-benzoylthiamine, which passively diffuses across intestinal enterocytes. Intracellular esterases then cleave the benzoyl group to release free thiamine for phosphorylation to thiamine pyrophosphate (TPP) (PMID: 16389547).
Transketolase Activation & AGE Prevention
Benfotiamine's primary therapeutic mechanism involves potent activation of transketolase, the rate-limiting enzyme of the pentose phosphate pathway. Enhanced transketolase activity diverts the glycolytic metabolites fructose-6-phosphate and glyceraldehyde-3-phosphate into the non-oxidative pentose phosphate pathway, preventing their accumulation and subsequent flux into four damaging pathways of hyperglycemic injury: (1) the polyol pathway, (2) the hexosamine pathway, (3) the protein kinase C activation pathway, and (4) the advanced glycation end-product (AGE) formation pathway (PMID: 12714638).
NF-kB & Inflammatory Pathway Inhibition
Benfotiamine directly inhibits NF-kB activation in endothelial cells and monocytes exposed to AGEs. By reducing intracellular AGE accumulation and blocking RAGE (receptor for AGEs) signaling, it attenuates downstream MAPK/ERK and JAK-STAT inflammatory cascades. This reduces expression of VCAM-1, ICAM-1, and MCP-1 in vascular endothelium, decreasing monocyte adhesion and transmigration (PMID: 16873689).
Neuroprotective Mechanisms
In diabetic neuropathy, benfotiamine preserves nerve function by: (1) maintaining Na⁺/K⁺-ATPase activity in peripheral nerves, (2) preventing glycation of myelin and neurofilament proteins, (3) reducing oxidative stress via enhanced NADPH generation through the pentose phosphate pathway, and (4) supporting mitochondrial PDH complex activity for neuronal energy metabolism (PMID: 18003764).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Superior Bioavailability:: Highly effective compared to thiamine HCl because its fat-soluble nature allows it to cross cell membranes easily.. Neuropathy Pain:: Consistently rated as an effective intervention for diabetic and alcoholic nerve damage in both clinical trials and user reports.. Alcohol Recovery:: Used effectively to replenish thiamine levels in those recovering from severe alcohol dependence.. Dose-Dependent:: Effectiveness often increases with higher dosages, particularly for chronic nerve pain.
- Highly effective compared to thiamine HCl because its fat-soluble nature allows it to cross cell membranes easily.
- Consistently rated as an effective intervention for diabetic and alcoholic nerve damage in both clinical trials and user reports.
- Used effectively to replenish thiamine levels in those recovering from severe alcohol dependence.
- Effectiveness often increases with higher dosages, particularly for chronic nerve pain.
Safety Profile
Safety Profile: Benfotiamine
Common Side Effects
- Mild gastrointestinal discomfort (nausea, bloating, stomach upset) in ~5-10% of users
- Headache (infrequent)
- Skin rash or itching (rare)
- Mild dizziness
Serious Adverse Effects
- Anaphylactic reactions (extremely rare; discontinue immediately if facial/throat swelling occurs)
- Severe dermatologic reactions have been reported in isolated case reports
- Potential for hypotension at very high doses
Contraindications
- Known hypersensitivity to thiamine or any benfotiamine excipients
- Patients with decompensated heart failure should use with caution (thiamine can affect cardiac output)
- Not recommended in patients with known thiamine allergy
Drug Interactions
- 5-Fluorouracil (5-FU): Thiamine derivatives may reduce efficacy of 5-FU; avoid concurrent use during chemotherapy
- Loop diuretics (furosemide): May increase thiamine excretion, potentially reducing benfotiamine effectiveness
- Alcohol: Chronic alcohol use depletes thiamine; benfotiamine may be beneficial but monitor closely
- Metformin: Long-term metformin use may reduce thiamine levels; benfotiamine may be complementary
Population-Specific Considerations
- Pregnancy/Lactation: Limited data; use only if clearly needed. Thiamine requirements increase during pregnancy
- Pediatric: Safety not established in children; dosing data insufficient
- Renal impairment: No dose adjustment typically needed, but monitor in severe CKD
- Hepatic impairment: Use with caution in severe liver disease; thiamine metabolism may be altered
- Diabetic patients: Generally well-tolerated; monitor blood glucose as benfotiamine may affect AGE formation pathways
- Elderly: No specific dose adjustments; well-tolerated in clinical trials involving elderly diabetic patients
Pharmacokinetic Profile
Benfotiamine — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Often taken at 300mg to 600mg daily for neuropathy management.
Molecular Structure
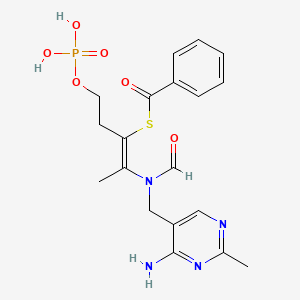
- Formula
- C19H23N4O6PS
- Weight
- 466.4 Da
- PubChem CID
- 3032771
- Exact Mass
- 466.1076 Da
- LogP
- 0.5
- TPSA
- 181 Ų
- H-Bond Donors
- 3
- H-Bond Acceptors
- 10
- Rotatable Bonds
- 10
- Complexity
- 697
Identifiers (SMILES, InChI)
InChI=1S/C19H23N4O6PS/c1-13(23(12-24)11-16-10-21-14(2)22-18(16)20)17(8-9-29-30(26,27)28)31-19(25)15-6-4-3-5-7-15/h3-7,10,12H,8-9,11H2,1-2H3,(H2,20,21,22)(H2,26,27,28)/b17-13-
BTNNPSLJPBRMLZ-LGMDPLHJSA-NSafety Profile
Common Side Effects
- Low Toxicity:: Generally reported to have a very high safety profile with minimal side effects even at high doses.
- Mild GI Distress:: Occasional reports of upset stomach if taken on an empty stomach.
- Skin Rash:: Rare reports of allergic skin reactions or 'vitamin smells' in sweat at extremely high doses.
References (5)
- [1]Benfotiamine and Cognitive Decline in Alzheimer's Disease: Results of a Randomized Placebo-Controlled Phase IIa Clinical Trial
→ This clinical trial found that benfotiamine significantly slowed cognitive decline and improved brain glucose metabolism in patients with mild Alzheimer's disease over 12 months.
- [3]Advances in the management of diabetic neuropathy
→ A review highlighting benfotiamine's ability to inhibit damaging glucose-driven pathways, making it a key pathogenetically oriented treatment for neuropathic complications.
- [4]Benfotiamine in diabetic polyneuropathy (BENDIP): results of a randomised, double blind, placebo-controlled clinical study
→ Clinical results showed that higher doses of benfotiamine led to significant improvements in neuropathy pain scores compared to placebo.
- [5]A double-blind, randomized, placebo-controlled clinical trial on benfotiamine treatment in patients with diabetic nephropathy
→ Research investigating the potential for benfotiamine to protect kidney function by reducing the production of advanced glycation end products (AGEs).
- [2]BOND study: a randomised double-blind, placebo-controlled trial over 12 months to assess the effects of benfotiamine on morphometric, neurophysiological and clinical measures in patients with type 2 diabetes with symptomatic polyneuropathy
→ This study evaluated whether long-term benfotiamine supplementation could improve nerve function and clinical symptoms in diabetic patients with nerve damage.
Beetroot Extract
Beetroot extract is a concentrated source of dietary nitrate (NO3-) that the body converts to nitric oxide (NO), a crucial molecule for vascular function. It pr
Berberine
Berberine is a natural alkaloid compound extracted from various plants including Berberis species, traditionally used in Chinese and Ayurvedic medicine. It work