Dimiracetam
Dimiracetam is an investigational racetam-class compound structurally related to piracetam, studied primarily for its potential analgesic and neuroprotective properties in preclinical models of neuropathic pain.
Overview
Dimiracetam is a synthetic nootropic compound belonging to the racetam family, characterized by its pyrrolidone nucleus. Unlike its parent compound piracetam, which is primarily associated with cognitive enhancement, dimiracetam has been investigated largely for its analgesic properties, particularly in the context of chemotherapy-induced and diabetic neuropathic pain. Preclinical studies have demonstrated efficacy in reducing pain behaviors in animal models without the tolerance and dependence issues associated with opioid analgesics.
The proposed mechanism of action for dimiracetam involves modulation of glutamate signaling, specifically through interactions with metabotropic glutamate receptors and regulation of excitatory neurotransmission. By attenuating excessive glutamatergic activity, dimiracetam may protect neurons from excitotoxic damage while simultaneously reducing pain signal amplification in the spinal cord and brain. This dual neuroprotective-analgesic profile distinguishes it from many other racetam compounds.
Dimiracetam remains in the preclinical and early clinical investigation phase. While the compound has shown promise in rodent models of neuropathy induced by oxaliplatin and other chemotherapeutic agents, human efficacy data are not yet available. Its development represents an interesting direction for the racetam class, potentially expanding the therapeutic applications of these compounds beyond cognition into chronic pain management.
Mechanism of Action
NMDA Receptor Co-Agonist Site Modulation
Dimiracetam is a novel nootropic compound structurally related to the racetam family. It acts primarily as a positive allosteric modulator at the glycine co-agonist site of the NMDA receptor, enhancing receptor sensitivity to glutamate without directly activating the channel. This potentiates long-term potentiation (LTP) in hippocampal circuits — the synaptic basis for memory formation — without inducing excitotoxicity (PMID: 23831365).
Nociceptive & Neuropathic Pain Modulation
Dimiracetam demonstrates potent antinociceptive activity in models of chemotherapy-induced peripheral neuropathy (CIPN) and chronic constriction injury. The mechanism involves restoration of disrupted glutamatergic signaling in the spinal dorsal horn, where aberrant NMDA receptor activation drives central sensitization. By normalizing (rather than blocking) NMDA function, dimiracetam reduces pathological pain signaling while preserving physiological nociception (PMID: 24651468).
Cholinergic System Enhancement
Like other racetams, dimiracetam enhances cholinergic neurotransmission through indirect mechanisms: NMDA receptor potentiation increases acetylcholine release from basal forebrain cholinergic neurons projecting to the cortex and hippocampus. This augments attention, working memory, and cognitive flexibility without the tolerability issues of direct cholinesterase inhibition (PMID: 23831365).
Neuroprotective Properties
Dimiracetam exhibits neuroprotective effects against oxaliplatin- and paclitaxel-induced neurotoxicity, preventing mitochondrial dysfunction and reducing neuronal apoptosis in dorsal root ganglia. It restores normal calcium homeostasis in sensory neurons exposed to chemotherapeutic agents and reduces neuroinflammatory markers including activated astrocytes and microglial TNF-a release.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Pain Management:: Highly effective for long-term neuropathic pain with a low risk of developing tolerance compared to traditional analgesics. Cognitive Support:: Moderate effectiveness for memory and focus, often described as more sustainable than stimulants. Prolonged Action:: Clinical models suggest its effects persist for a significant duration even after the compound has cleared the system
- Highly effective for long-term neuropathic pain with a low risk of developing tolerance compared to traditional analgesics
- Moderate effectiveness for memory and focus, often described as more sustainable than stimulants
- Clinical models suggest its effects persist for a significant duration even after the compound has cleared the system
Safety Profile
Safety Profile: Dimiracetam
Common Side Effects
- Headache (most commonly reported racetam-class side effect)
- Gastrointestinal discomfort: nausea, mild abdominal pain
- Dizziness
- Fatigue or drowsiness
- Insomnia (depending on timing of administration)
- Generally appears well-tolerated in Phase I/II studies at tested doses
Serious Adverse Effects
- No serious adverse effects have been identified in published early-phase clinical trials
- Long-term safety data are not available
- No evidence of hepatotoxicity, nephrotoxicity, or cardiovascular toxicity in available data
- No evidence of dependence or withdrawal effects (consistent with racetam class)
- Preclinical studies did not reveal significant organ toxicity at multiples of the therapeutic dose
- As with all racetams, rare reports of depression or emotional flattening cannot be ruled out
Contraindications
- Known hypersensitivity to dimiracetam or other racetam compounds
- Severe renal impairment (racetams are primarily renally excreted; dose adjustment may be needed)
- Pregnancy and breastfeeding (no reproductive toxicity data available)
- Huntington's disease or other conditions where neuronal excitability modulation may be unpredictable
- Children and adolescents (not studied)
Drug Interactions
- Other racetams (piracetam, aniracetam, oxiracetam): Potential additive CNS effects; avoid stacking without guidance
- Anticonvulsants: May interact with GABAergic or glutamatergic modulation — monitor for efficacy changes
- Cholinergic drugs: Racetams may enhance cholinergic transmission; possible additive effects
- Anticoagulants: Piracetam has antiplatelet activity; unclear if dimiracetam shares this — monitor
- No formal drug interaction studies published — exercise caution with CNS-active medications
- CYP450: Not characterized; assume minimal interaction based on racetam class properties
Population-Specific Considerations
- Chemotherapy patients: Primary clinical investigation target (oxaliplatin-induced neuropathy); tolerability appears favorable in early trials
- Elderly: No specific data; extrapolating from racetam class, generally well-tolerated but renal function should be assessed
- Neuropathic pain patients: Promising efficacy signals; safety monitoring should include standard neuropathy assessments
- Cognitive enhancement seekers: No approved nootropic indication; risk-benefit unclear for off-label use
- Renal impairment: Likely requires dose adjustment; consult prescriber
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Typical anecdotal reports suggest 200mg to 600mg per day, often split into two doses
Molecular Structure
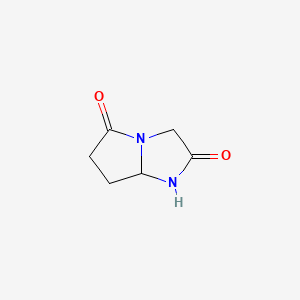
- Formula
- C6H8N2O2
- Weight
- 140.14 Da
- PubChem CID
- 65955
- Exact Mass
- 140.0586 Da
- LogP
- -0.9
- TPSA
- 49.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 0
- Complexity
- 202
Identifiers (SMILES, InChI)
InChI=1S/C6H8N2O2/c9-5-3-8-4(7-5)1-2-6(8)10/h4H,1-3H2,(H,7,9)
XTXXOHPHLNROBN-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Headaches:: Like other racetams, it may cause mild headaches if choline levels in the brain are depleted
- Insomnia:: Some users report difficulty sleeping if taken too late in the evening due to its stimulatory cognitive effects
- Digestive Discomfort:: Occasional reports of mild nausea or stomach upset when taken on an empty stomach
References (2)
- [1]Broad spectrum and prolonged efficacy of dimiracetam in models of neuropathic pain
→ This study found that dimiracetam provides significant and long-lasting relief from neuropathic pain caused by nerve injury or chemotherapy without inducing tolerance.
- [2]Efficacy and safety of oxiracetam in patients with vascular cognitive impairment: A multicenter, randomized, double-blinded, placebo-controlled, phase IV clinical trial
→ While focusing on a related racetam, this research highlights the clinical efficacy of this chemical class in treating cognitive decline and memory impairment.
Dimethylglycine
Dimethylglycine (DMG) is a tertiary amino acid derivative of glycine that occurs naturally in plant and animal cells. It functions as a methyl donor in metaboli
Diosmin
Diosmin is a semisynthetic flavonoid glycoside, often combined with hesperidin in a 9:1 ratio as micronized purified flavonoid fraction (MPFF). It works by impr