GW0742
A highly selective PPARδ agonist structurally related to Cardarine (GW501516), investigated for metabolic regulation, endurance enhancement, and anti-inflammatory effects.
Overview
GW0742 is a potent and highly selective agonist of peroxisome proliferator-activated receptor delta (PPARδ), developed by GlaxoSmithKline as a structural analog of the better-known GW501516 (Cardarine). Both compounds activate PPARδ, a nuclear receptor that plays a central role in fatty acid oxidation, energy expenditure, and metabolic homeostasis, but GW0742 exhibits even greater selectivity for the delta subtype over PPARα and PPARγ. This makes it a valuable pharmacological tool for studying PPARδ-mediated biology.
Preclinical research with GW0742 has demonstrated significant metabolic effects including enhanced fatty acid beta-oxidation in skeletal muscle, improved lipid profiles (increased HDL, decreased triglycerides and LDL), and anti-inflammatory activity through suppression of NF-kB-mediated cytokine production. In animal models, GW0742 has shown cardioprotective properties, reducing infarct size and improving cardiac function following ischemia-reperfusion injury. Its anti-inflammatory effects have also been investigated in models of dermatitis, colitis, and kidney injury, suggesting broad therapeutic potential beyond metabolic applications.
GW0742 is often compared directly to GW501516, and both compounds are used in research and performance-enhancement contexts for their endurance-boosting and fat-oxidizing properties. It is important to note that GW0742, like GW501516, has not been approved for human use by any regulatory agency. The carcinogenicity concerns that led to the discontinuation of GW501516 clinical trials apply to the broader PPARδ agonist class, and long-term safety data in humans is lacking. GW0742 remains an investigational compound and should be approached with appropriate caution.
Mechanism of Action
PPARdelta Nuclear Receptor Agonism
GW0742 is a potent and highly selective synthetic agonist of peroxisome proliferator-activated receptor delta (PPARdelta/PPARbeta), a ligand-activated nuclear receptor and transcription factor. Upon binding GW0742, PPARdelta undergoes a conformational change that promotes heterodimerization with the retinoid X receptor (RXR). This PPARdelta-RXR heterodimer recruits co-activator proteins (SRC-1, PGC-1alpha) and binds to peroxisome proliferator response elements (PPREs) in target gene promoters, activating transcription of metabolic gene networks (PMID: 15734739).
Fatty Acid Oxidation & Metabolic Reprogramming
The primary downstream effect of PPARdelta activation is a comprehensive shift toward fatty acid catabolism. GW0742 upregulates genes encoding fatty acid transport proteins (FATP, CD36), mitochondrial beta-oxidation enzymes (CPT1, MCAD, LCAD), and uncoupling proteins (UCP2, UCP3). It also promotes mitochondrial biogenesis through PGC-1alpha induction, increasing oxidative capacity in skeletal muscle and cardiac tissue. This metabolic reprogramming enhances endurance capacity and shifts fuel utilization from glucose toward fatty acids (PMID: 18432616).
Glucose Homeostasis & Insulin Sensitization
GW0742 improves glucose handling by upregulating pyruvate dehydrogenase kinase 4 (PDK4), which spares glucose by redirecting pyruvate away from oxidation toward gluconeogenic substrates. It also enhances insulin sensitivity in skeletal muscle via increased GLUT4 expression and improved insulin receptor signaling. In adipose tissue, PPARdelta activation suppresses lipogenic gene expression, reducing ectopic lipid accumulation that drives insulin resistance.
Anti-Inflammatory & Cardioprotective Effects
GW0742 exerts potent anti-inflammatory effects by trans-repression of NF-kB — the PPARdelta-RXR complex physically sequesters the p65 subunit of NF-kB, preventing its nuclear translocation and suppressing transcription of IL-6, TNF-alpha, MCP-1, and VCAM-1. In cardiac tissue, this anti-inflammatory action combined with enhanced fatty acid metabolism confers protection against ischemia-reperfusion injury and pathological cardiac remodeling.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Cardiovascular Performance:: Highly effective for runners, cyclists, and athletes looking to increase their VO2 max and aerobic threshold.. Fat Loss:: Noted for being more potent than Cardarine in its fat-burning action, especially when paired with a caloric deficit.. Rapid Action:: Users often report feeling the metabolic shift and endurance benefits within the first week of use.
- Highly effective for runners, cyclists, and athletes looking to increase their VO2 max and aerobic threshold.
- Noted for being more potent than Cardarine in its fat-burning action, especially when paired with a caloric deficit.
- Users often report feeling the metabolic shift and endurance benefits within the first week of use.
Safety Profile
Safety Profile: GW0742 (PPARδ Agonist)
Common Side Effects
- Headache and mild fatigue reported in preclinical and early human data
- Gastrointestinal upset (nausea, diarrhea, abdominal discomfort)
- Mild transaminase elevations (ALT/AST)
- Changes in lipid profile (alterations in HDL, LDL, and triglycerides)
- Muscle cramps or myalgia at higher doses
Serious Adverse Effects
- Carcinogenesis concern: Closely related PPARδ agonist GW501516 was withdrawn from development due to rapid cancer induction in animal models; GW0742 shares the same target and similar structural features, though some preclinical data suggest a different safety margin
- Hepatotoxicity: Liver enzyme elevations and potential hepatic injury, particularly with prolonged use or high doses
- Cardiovascular effects: PPARδ activation affects cardiac metabolism; theoretical risk of cardiac hypertrophy or other cardiac remodeling
- Reproductive toxicity: Animal studies with PPARδ agonists suggest developmental toxicity
- Long-term safety unknown: GW0742 has not completed full human clinical development
Contraindications
- Known or suspected malignancy of any type (carcinogenesis risk from PPARδ agonism)
- Active liver disease or elevated transaminases >2x ULN
- Pregnancy, planned pregnancy, or lactation (teratogenic potential)
- Women of childbearing potential without reliable contraception
- Severe cardiovascular disease (unknown cardiac remodeling risk)
- Pediatric use
Drug Interactions
- Statins and fibrates: PPARδ agonists modulate lipid metabolism; additive or unpredictable effects on lipid panel and hepatotoxicity risk
- CYP450 substrates: Metabolic profile not fully characterized; assume potential CYP3A4 involvement
- Anticoagulants: PPARδ activation may affect thrombotic pathways; monitor coagulation parameters
- Diabetes medications: May alter glucose and insulin sensitivity; monitor blood glucose
- Other PPAR agonists (thiazolidinediones, fibrates): Additive or synergistic effects on PPAR-mediated pathways
Population-Specific Considerations
- Pregnancy/Lactation: Strictly contraindicated; PPARδ agonists show developmental toxicity in animals
- Cancer patients or survivors: Avoid entirely; PPARδ agonism has demonstrated pro-carcinogenic potential in animal models
- Athletes: GW0742, like GW501516, is banned by WADA under the S4 Hormone and Metabolic Modulators category
- Hepatic impairment: Avoid or use with extreme caution; close LFT monitoring required
- Elderly: No specific data; increased susceptibility to hepatotoxicity and metabolic disruption
- Note: GW0742 is an investigational compound not approved for human use. All safety data should be considered preliminary
Pharmacokinetic Profile
Molecular Structure
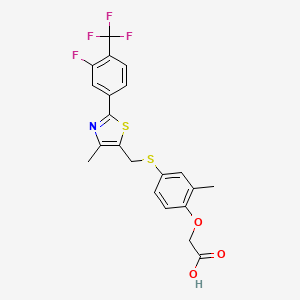
- Formula
- C21H17F4NO3S2
- Weight
- 471.5 Da
- PubChem CID
- 9934458
- Exact Mass
- 471.0586 Da
- LogP
- 6
- TPSA
- 113 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 10
- Rotatable Bonds
- 7
- Complexity
- 612
Identifiers (SMILES, InChI)
InChI=1S/C21H17F4NO3S2/c1-11-7-14(4-6-17(11)29-9-19(27)28)30-10-18-12(2)26-20(31-18)13-3-5-15(16(22)8-13)21(23,24)25/h3-8H,9-10H2,1-2H3,(H,27,28)
HWVNEWGKWRGSRK-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Heart Hypertrophy:: Some animal studies have raised concerns regarding potential heart enlargement, though human data is lacking.
- Carcinogenicity Concerns:: Like all PPAR-delta agonists, there are long-term theoretical concerns regarding cell proliferation, though user reports are anecdotal.
- Metabolic Shift:: Some users report initial lethargy as the body adjusts to burning fats over glucose.
References (1)
- [1]Emerging drugs affecting skeletal muscle function and mitochondrial biogenesis - Potential implications for sports drug testing programs
→ This research identifies GW0742 as part of a class of metabolic modulators that enhance mitochondrial function and physical performance, leading to its inclusion on prohibited substance lists in competitive sports.
GV1001
**GV1001** is a 16-amino acid peptide corresponding to residues 611-626 of human telomerase reverse transcriptase (hTERT), the catalytic subunit of telomerase.
Gymnema Sylvestre
Gymnema Sylvestre is a woody climbing shrub native to India and Africa, traditionally known as 'Gudmar' or 'sugar destroyer' for its unique ability to temporari