Sunifiram
An experimental ampakine nootropic structurally derived from piracetam but approximately 1,000 times more potent, enhancing glutamatergic AMPA receptor signaling, long-term potentiation, and memory consolidation at microgram-level doses.
Overview
Sunifiram (DM-235) is a synthetic piperazine-containing compound developed at the University of Firenze (Florence), Italy, as part of a structure-activity relationship program exploring simplified analogs of piracetam. Despite its structural origin in the racetam family, sunifiram is not technically classified as a racetam because it lacks the pyrrolidine nucleus — instead, it features a piperazine ring system. However, it shares the racetam class's fundamental mechanism of positive allosteric modulation of AMPA-type glutamate receptors, and it does so with extraordinary potency: sunifiram enhances AMPA receptor currents at concentrations approximately 1,000 times lower than piracetam, making it one of the most potent ampakine nootropics identified in preclinical research.
Sunifiram's mechanism involves binding to the AMPA receptor at the dimer interface between GluA1 and GluA2 subunits, stabilizing the channel in its open conformation and reducing receptor desensitization. This enhanced glutamatergic signaling facilitates long-term potentiation (LTP) in the hippocampus — the synaptic strengthening process essential for memory formation and consolidation. In animal studies, sunifiram reverses amnesia induced by scopolamine (a muscarinic antagonist), NBQX (an AMPA antagonist), and MK-801 (an NMDA antagonist), demonstrating broad anti-amnesic efficacy across multiple neurotransmitter systems. Additional mechanisms include activation of CaMKII (calcium/calmodulin-dependent protein kinase II) and PKCα — intracellular kinases that phosphorylate AMPA receptor subunits to enhance their conductance and membrane insertion — as well as increased glycine release that potentiates NMDA receptor function.
Sunifiram remains strictly an experimental research compound with no human clinical trials conducted to date. Despite this, it has gained popularity in the nootropic community, where users typically dose in the range of 4-10 mg sublingually (given its potency at microgram-to-milligram levels in animal models). The absence of human pharmacokinetic, safety, and efficacy data makes responsible dosing guidance impossible, and the compound's potent glutamatergic activity theoretically carries excitotoxicity risk at excessive doses. Sunifiram is often discussed alongside other ultra-potent nootropics including noopept (which operates through a different mechanism involving BDNF and NGF upregulation), unifiram (DM-232, a closely related structural analog with similar potency), and coluracetam (which enhances high-affinity choline uptake). It is not approved as a drug or supplement in any jurisdiction and is sold exclusively as a research chemical.
Mechanism of Action
Sunifiram (DM-235) is a synthetic piperazine-derived compound structurally related to the racetam family but approximately 1,000 times more potent than piracetam in enhancing cognitive function in animal models. Its primary mechanism involves modulation of glutamatergic neurotransmission at two key receptor subtypes: NMDA and AMPA receptors. At NMDA receptors, sunifiram acts at the glycine-binding site (the GluN1 subunit), enhancing receptor activation without directly stimulating it, thereby facilitating the NMDA receptor-dependent component of long-term potentiation (LTP) in the hippocampus.
Sunifiram's effects on AMPA receptors occur indirectly through activation of calcium/calmodulin-dependent protein kinase II (CaMKII) and protein kinase C alpha (PKCalpha). These kinases phosphorylate the AMPA receptor GluR1 subunit at Ser831, increasing single-channel conductance and promoting AMPA receptor trafficking to the postsynaptic membrane. This dual enhancement of both NMDA and AMPA receptor function produces a robust facilitation of LTP, the cellular mechanism underlying learning and memory formation.
In vivo studies have demonstrated that sunifiram reverses amnesia induced by scopolamine (anticholinergic), mecamylamine (nicotinic antagonist), and dizocilpine (NMDA antagonist) in passive avoidance and Morris water maze paradigms. The compound also increases acetylcholine release in the prefrontal cortex, suggesting secondary cholinergic facilitation. Sunifiram remains an experimental compound with no approved clinical applications, and human safety and efficacy data are essentially absent despite its popularity in the nootropics community.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Potency Concerns:: While research suggests high potency, user experiences are highly variable with many reporting minimal or negative effects. Combination Risks:: Frequently reported problems when stacked with other racetams, noopept, or stimulants, suggesting negative synergistic effects. Safety Profile:: Experienced users consistently warn it has never been studied in humans and at least one hospitalization has been reported. Preference for Alternatives:: Many users who tried both sunifiram and unifiram report unifiram as safer and more effective with better tolerability
- While research suggests high potency, user experiences are highly variable with many reporting minimal or negative effects
- Frequently reported problems when stacked with other racetams, noopept, or stimulants, suggesting negative synergistic effects
- Experienced users consistently warn it has never been studied in humans and at least one hospitalization has been reported
- Many users who tried both sunifiram and unifiram report unifiram as safer and more effective with better tolerability
Safety Profile
Safety Profile: Sunifiram
Common Side Effects
- Limited human safety data; effects reported primarily from anecdotal user reports
- Headache (common with ampakine-type compounds)
- Insomnia and overstimulation
- Anxiety and nervousness at higher doses
- Nasal congestion (when administered sublingually)
- Jaw clenching and bruxism reported by some users
Serious Adverse Effects
- No human clinical trials completed: Safety profile is largely unknown
- Theoretical excitotoxicity risk due to AMPA receptor potentiation (could lower seizure threshold)
- Unknown long-term neurotoxicity profile
- Unpredictable dose-response due to very high potency (active at microgram to low-milligram doses)
- Theoretical risk of neuronal damage from excessive glutamatergic signaling
Contraindications
- Epilepsy or seizure disorders (AMPA potentiation may lower seizure threshold)
- History of stroke or brain injury (excitotoxicity vulnerability)
- Pregnancy and lactation
- Children and adolescents
- Concurrent use of other ampakines or glutamate-modulating compounds
Drug Interactions
- Stimulants: Additive CNS excitation; increased seizure risk
- Other nootropics (racetams, ampakines): Overlapping glutamatergic mechanisms; unpredictable synergy
- Anticonvulsants: May reduce their efficacy
- SSRIs/SNRIs: Unknown interaction; theoretical concern for serotonin-glutamate crosstalk
Population-Specific Considerations
- Research compound only: Not approved for human use; available only through gray-market research chemical vendors
- Potency warning: Active at sub-milligram doses; accurate dosing requires precision scales and significant caution
- No established dosing protocol: User-reported doses vary widely (4–10 mg sublingually); no clinical guidance exists
- Risk-benefit: Very poor; no proven therapeutic benefit to offset unknown risks
- Nootropic community: Heavily discussed in online forums, but lack of clinical data makes all claims unverifiable
Pharmacokinetic Profile
Molecular Structure
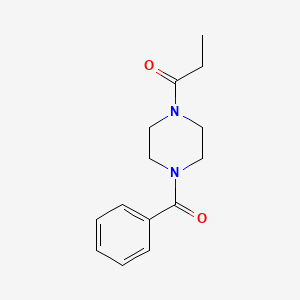
- Formula
- C14H18N2O2
- Weight
- 246.30 Da
- PubChem CID
- 4223812
- Exact Mass
- 246.1368 Da
- LogP
- 1.1
- TPSA
- 40.6 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 2
- Complexity
- 303
Identifiers (SMILES, InChI)
InChI=1S/C14H18N2O2/c1-2-13(17)15-8-10-16(11-9-15)14(18)12-6-4-3-5-7-12/h3-7H,2,8-11H2,1H3
DGOWDUFJCINDGI-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Sleep Disruption:: Insomnia and inability to sleep reported, particularly when taken later in day or combined with stimulants
- Anxiety and Akathisia:: Multiple reports of severe anxiety, restlessness, and agitation, especially with phenylpiracetam combination
- Headaches:: Massive headaches reported when combined with multiple racetams
- Excitotoxicity Risk:: Users and research warn about potential for excitotoxicity with chronic use, recommending NMDA antagonists if using
References (6)
- [1]Pharmacological characterization of DM232 (unifiram) and DM235 (sunifiram), new potent cognition enhancers
→ Sunifiram demonstrated cognition-enhancing properties four orders of magnitude more potent than piracetam, increasing acetylcholine release and hippocampal activity through AMPA receptor involvement without binding to major central receptors.
- [2]Novel nootropic drug sunifiram enhances hippocampal synaptic efficacy via glycine-binding site of N-methyl-D-aspartate receptor
→ Sunifiram enhanced NMDAR-dependent long-term potentiation in mouse hippocampal slices at 10-100 nM concentrations in a bell-shaped dose-response curve, with effects blocked by glycine-binding site antagonists.
- [3]DM235 (sunifiram): a novel nootropic with potential as a cognitive enhancer
→ Sunifiram prevented amnesia induced by scopolamine, mecamylamine, baclofen, and clonidine at doses of 0.001-0.1 mg/kg, showing comparable efficacy to established nootropics but at much lower doses.
- [4]Novel nootropic drug sunifiram improves cognitive deficits via CaM kinase II and protein kinase C activation in olfactory bulbectomized mice
→ Sunifiram improved spatial memory and hippocampal LTP in olfactory bulbectomized mice at 0.01-1.0 mg/kg doses through NMDAR-dependent mechanisms involving CaMKII and PKC activation.
- [5]AMPA-receptor activation is involved in the antiamnesic effect of DM 232 (unifiram) and DM 235 (sunifiram)
→ Both compounds reversed AMPA receptor antagonist-induced amnesia and increased excitatory synaptic transmission in rat hippocampus, confirming AMPA receptor involvement in their cognitive-enhancing effects.
- [6]Unifi nootropics from the lab to the web: a story of academic (and industrial) shortcomings
→ A cautionary review noting that sunifiram and unifiram were never protected by patent, have limited preclinical studies, unknown long-term toxicity, and yet are widely sold online despite never being studied in humans.
Suma
Suma (Pfaffia paniculata), also known as Brazilian ginseng, is an adaptogenic herb containing ecdysteroids, particularly ecdysterone, along with germanium, vita
Super Human Blend (CJC-1295 + Ipamorelin + BPC-157 + TB-500)
The Super Human Blend is a compounded multi-peptide formulation combining four peptides with complementary mechanisms: CJC-1295 (a growth hormone-releasing horm