FMN (Flavin Mononucleotide)
FMN is a biomolecule derived from riboflavin (vitamin B2) that functions as a coenzyme in oxidoreductase reactions and serves as the precursor to FAD, playing a key role in mitochondrial electron transport and cellular energy metabolism.
Overview
Flavin mononucleotide (FMN), also known as riboflavin-5'-phosphate, is a phosphorylated derivative of riboflavin (vitamin B2) that serves as a prosthetic group for a variety of flavoprotein enzymes. It is produced in the body by the enzyme riboflavin kinase, which phosphorylates riboflavin at the 5' position. FMN is the direct precursor to FAD and plays a distinct role as a cofactor in its own right, particularly in Complex I (NADH dehydrogenase) of the mitochondrial electron transport chain.
As a redox cofactor, FMN cycles between its oxidized, semiquinone, and fully reduced (FMNH2) states, enabling it to participate in both one-electron and two-electron transfer reactions. This versatility makes it essential for enzymes involved in a broad range of biological processes, including bioluminescence in bacteria, nitric oxide synthesis, and the reduction of methemoglobin. FMN-dependent enzymes are also involved in the biosynthesis of other vitamins and cofactors.
Supplemental FMN has been investigated for its potential benefits in conditions linked to riboflavin insufficiency, including certain mitochondrial myopathies and migraine prophylaxis. Because FMN is water-soluble and more bioavailable than some forms of riboflavin, it is sometimes preferred in clinical and nutraceutical applications. Its role in supporting Complex I function makes it particularly relevant to research on mitochondrial dysfunction and neurodegenerative conditions.
Mechanism of Action
Coenzyme Structure & Redox Properties
Flavin mononucleotide (FMN), also known as riboflavin-5'-phosphate, is the phosphorylated form of riboflavin (vitamin B2) and serves as an essential redox cofactor for numerous flavoenzymes. FMN contains the isoalloxazine ring system that undergoes reversible one- or two-electron reductions: FMN (oxidized) → FMNH· (semiquinone radical) → FMNH2 (fully reduced). This ability to accept and donate electrons in single or paired steps makes FMN uniquely versatile in biological electron transfer reactions (PMID: 24655016).
Mitochondrial Complex I (NADH Dehydrogenase)
FMN's most critical role is as the primary electron acceptor in Complex I (NADH:ubiquinone oxidoreductase) of the mitochondrial electron transport chain. FMN bound at the NADH-binding site of Complex I accepts a hydride ion (two electrons + one proton) from NADH, becoming FMNH2. These electrons are then passed through a chain of seven iron-sulfur (Fe-S) clusters to ubiquinone (CoQ10), coupled with the translocation of four protons across the inner mitochondrial membrane. This is the largest proton-pumping complex in the ETC and contributes ~40% of the proton motive force driving ATP synthesis (PMID: 20675419).
Other FMN-Dependent Enzymes
NADH-cytochrome b5 reductase uses FMN to reduce cytochrome b5, essential for methemoglobin reduction, fatty acid desaturation, and cytochrome P450-mediated steroidogenesis. Pyridoxamine 5'-phosphate oxidase (PNPO) requires FMN to synthesize the active form of vitamin B6 (pyridoxal 5'-phosphate, PLP), linking riboflavin status to vitamin B6 metabolism. L-amino acid oxidase and D-amino acid oxidase both use FMN for oxidative deamination (PMID: 22113863).
Biosynthesis & Relationship to FAD
FMN is synthesized from riboflavin by riboflavin kinase (EC 2.7.1.26) in a reaction requiring ATP. FMN then serves as the substrate for FAD synthase, which adenylates FMN to produce FAD. Thus, FMN occupies a central position in flavin metabolism, serving both as an independent coenzyme and as the obligate precursor to FAD.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Mitochondrial Rescue:: Highly effective in clinical contexts for treating mitochondrial diseases and ischemia/reperfusion injury.. Bioavailability:: Considered more effective for those with riboflavin kinase deficiencies who cannot convert standard B2 efficiently.. Fast-Acting:: Because it does not require the same metabolic conversion steps as riboflavin, it is utilized more rapidly by cells.
- Highly effective in clinical contexts for treating mitochondrial diseases and ischemia/reperfusion injury.
- Considered more effective for those with riboflavin kinase deficiencies who cannot convert standard B2 efficiently.
- Because it does not require the same metabolic conversion steps as riboflavin, it is utilized more rapidly by cells.
Safety Profile
Safety Profile: FMN (Flavin Mononucleotide)
Common Side Effects
- Generally very well tolerated as a bioactive form of vitamin B2 (riboflavin-5'-phosphate)
- Bright yellow-orange discoloration of urine (harmless; indicates riboflavin excretion)
- Mild gastrointestinal symptoms: nausea, bloating, or diarrhea at doses exceeding 100 mg/day
- Occasional facial flushing, especially with intravenous administration
- Mild photosensitivity (FMN is a photosensitizer); increased sunburn risk with high-dose supplementation
Serious Adverse Effects
- Extremely rare at physiological doses due to water-soluble nature and efficient renal excretion
- Theoretical risk of phototoxic reactions at very high doses or with concurrent photosensitizing medications
- Anaphylactic reactions have been reported rarely with injectable forms (likely excipient-related)
- High-dose IV administration may cause transient numbness, tingling, or itching at the injection site
- No known cumulative toxicity; excess is rapidly excreted in urine
Contraindications
- Known hypersensitivity to riboflavin, FMN, or any formulation excipients
- Caution with injectable forms in patients with history of anaphylaxis to B-vitamin injections
- No absolute contraindications for oral supplementation at standard doses (10–50 mg/day)
Drug Interactions
- Tetracycline antibiotics: riboflavin and FMN may reduce tetracycline absorption; separate by 2+ hours
- Anticholinergic drugs: may reduce riboflavin absorption by slowing GI transit and altering pH
- Probenecid: may reduce renal tubular secretion of riboflavin, potentially increasing levels
- Tricyclic antidepressants (amitriptyline, imipramine): may impair riboflavin metabolism and increase FMN requirements
- Methotrexate: riboflavin may support folate metabolism; theoretical interaction, though generally considered beneficial
- Phenobarbital and phenytoin: may accelerate riboflavin metabolism, increasing requirements
- Photosensitizing drugs (tetracyclines, fluoroquinolones, thiazides): additive photosensitivity risk
Population-Specific Considerations
- Pregnant women: FMN is generally considered safe; riboflavin requirements increase during pregnancy (RDA 1.4 mg/day); higher therapeutic doses should be supervised
- Breastfeeding: riboflavin is secreted in breast milk; supplementation at moderate doses is considered safe and may benefit both mother and infant
- Infants/children: riboflavin deficiency should be corrected; supplemental FMN doses should be age-appropriate (pediatric RDA: 0.3–1.3 mg/day depending on age)
- Elderly: increased risk of riboflavin deficiency due to reduced absorption; FMN supplementation often well tolerated
- Renal impairment: water-soluble B vitamins accumulate in severe renal failure; monitor in dialysis patients
- Migraine patients: high-dose riboflavin (400 mg/day) used prophylactically; FMN form may offer better bioavailability with fewer GI side effects
Pharmacokinetic Profile
Molecular Structure
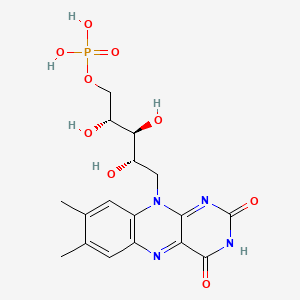
- Formula
- C17H21N4O9P
- Weight
- 456.3 Da
- PubChem CID
- 643976
- Exact Mass
- 456.1046 Da
- LogP
- -2.6
- TPSA
- 202 Ų
- H-Bond Donors
- 6
- H-Bond Acceptors
- 10
- Rotatable Bonds
- 7
- Complexity
- 843
Identifiers (SMILES, InChI)
InChI=1S/C17H21N4O9P/c1-7-3-9-10(4-8(7)2)21(15-13(18-9)16(25)20-17(26)19-15)5-11(22)14(24)12(23)6-30-31(27,28)29/h3-4,11-12,14,22-24H,5-6H2,1-2H3,(H,20,25,26)(H2,27,28,29)/t11-,12+,14-/m0/s1
FVTCRASFADXXNN-SCRDCRAPSA-NSafety Profile
Common Side Effects
- Chromaturia:: Harmless bright yellow or neon-orange discoloration of urine due to excess riboflavin excretion.
- Digestive Comfort:: Generally well-tolerated, though high doses may cause mild gastrointestinal upset in sensitive individuals.
- Rapid Absorption:: Unlike standard B2, its high solubility can lead to faster but shorter-lived peaks in serum levels if not taken with food.
References (2)
- [2]Flavin mononucleotide
→ An analysis of FMN as a stronger oxidizing agent than NAD, highlighting its unique ability to participate in both one- and two-electron transfers within cells.
- [1]Riboflavin metabolism: role in mitochondrial function
→ This review details how riboflavin derivatives like FMN are indispensable for mitochondrial energy production and the maintenance of the electron transport chain.
Fisetin
Fisetin is a naturally occurring flavonoid found in fruits and vegetables (strawberries, apples, grapes) with senolytic properties, meaning it selectively elimi
Folinic Acid (Leucovorin)
Folinic acid (also known as leucovorin) is a reduced form of folate (vitamin B9) that can be directly utilized by the body without requiring conversion by the e