Fasoracetam
Fasoracetam is a synthetic nootropic compound belonging to the racetam family, known for its unique ability to modulate glutamate, GABA, and cholinergic systems simultaneously. It has been investigated for potential cognitive enhancement and as a treatment for ADHD in adolescents with specific glutamatergic gene variants.
Overview
Fasoracetam (NS-105/LAM-105) is a piperidinone-derived racetam originally developed by Nippon Shinyaku in the 1990s for the treatment of vascular dementia. Although initial clinical trials were discontinued due to insufficient efficacy in that indication, the compound was later licensed by Aevi Technologies (formerly NeuroFix) for investigation in attention deficit hyperactivity disorder (ADHD), specifically in patients carrying mutations in metabotropic glutamate receptor genes.
The pharmacological profile of fasoracetam is distinctive among racetams. It acts as an upregulator of GABA-B receptors, a modulator of metabotropic glutamate receptors (mGluRs), and an enhancer of cholinergic activity through high-affinity choline uptake stimulation. This triple mechanism of action differentiates it from simpler racetams like piracetam and may explain its anxiolytic properties alongside cognitive enhancement. Preclinical studies have demonstrated improvements in learning and memory tasks, as well as reductions in learned helplessness behaviors in animal models.
A Phase II clinical trial in adolescents with ADHD and confirmed mGluR gene mutations showed promising results, with improvements in ADHD symptom scores and minimal side effects. The compound is generally well-tolerated, with headache and mild gastrointestinal discomfort being the most commonly reported adverse effects. Fasoracetam remains an investigational compound and is not currently approved by any regulatory agency for clinical use.
Mechanism of Action
Metabotropic Glutamate Receptor Modulation
Fasoracetam (NFC-1, NS-105) is a synthetic racetam-class nootropic that acts as a positive allosteric modulator of metabotropic glutamate receptors (mGluRs), particularly mGluR1 (Group I) and mGluR5 subtypes. By enhancing glutamatergic signaling through these Gq-coupled receptors, fasoracetam activates phospholipase C (PLC), increasing inositol trisphosphate (IP3) and diacylglycerol (DAG) production, which amplifies intracellular calcium release and protein kinase C (PKC) activity in postsynaptic neurons (PMID: 10338108).
Cholinergic Enhancement
Fasoracetam significantly upregulates high-affinity choline uptake (HACU) in the hippocampus and cortex, the rate-limiting step for acetylcholine (ACh) synthesis. This increases ACh availability at muscarinic and nicotinic receptors, enhancing cholinergic neurotransmission critical for learning, memory encoding, and attentional processes. Unlike direct cholinesterase inhibitors, fasoracetam's modulation of upstream choline transport may produce a more physiological enhancement of cholinergic tone (PMID: 10338108).
GABAergic Modulation
Fasoracetam upregulates GABA-B receptor expression in cortical regions, potentially through transcriptional mechanisms. GABA-B receptors are Gi/o-coupled GPCRs that inhibit adenylyl cyclase and activate inwardly rectifying potassium channels (GIRKs), providing inhibitory tone that counterbalances excitatory signaling. This property has led to clinical investigation in ADHD patients harboring mutations in the glutamate decarboxylase (GAD) and GABA-B receptor genes (PMID: 29039583).
Synaptic Plasticity & Cognitive Effects
Through combined glutamatergic and cholinergic enhancement, fasoracetam facilitates long-term potentiation (LTP) in hippocampal CA1 circuits. It reverses scopolamine-induced amnesia and baclofen-induced memory deficits in animal models, demonstrating bidirectional rescue of cholinergic and GABAergic disruptions.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
ADHD Management:: Multiple users with diagnosed ADHD report it as one of the most effective nootropics they've tried, particularly when pharmaceutical options failed or weren't tolerated. Tolerance Prevention:: Commonly stacked with phenibut to prevent tolerance buildup and enhance effects through GABA-B receptor sensitization. Build-Up Period:: Effects are noticed immediately but continue to build over 1-2 weeks of consistent use, requiring daily administration rather than cycling for sustained benefits. Individual Response:: Clinical trials used high doses (80mg+) for ADHD, and users report higher doses (40-80mg) are more effective than lower doses for noticeable cognitive benefits
- Multiple users with diagnosed ADHD report it as one of the most effective nootropics they've tried, particularly when pharmaceutical options failed or weren't tolerated
- Commonly stacked with phenibut to prevent tolerance buildup and enhance effects through GABA-B receptor sensitization
- Effects are noticed immediately but continue to build over 1-2 weeks of consistent use, requiring daily administration rather than cycling for sustained benefits
- Clinical trials used high doses (80mg+) for ADHD, and users report higher doses (40-80mg) are more effective than lower doses for noticeable cognitive benefits
Safety Profile
Safety Profile: Fasoracetam
Common Side Effects
- Headache, often attributed to increased acetylcholine demand (may respond to choline supplementation)
- Mild fatigue or sedation, particularly at higher doses
- Gastrointestinal upset including nausea and abdominal discomfort
- Insomnia or disrupted sleep when taken later in the day
- Mild anxiety or jitteriness during initial use
- Decreased appetite
- Dry mouth
Serious Adverse Effects
- Limited human safety data; most information from Phase I trials and anecdotal reports
- Potential for GABAergic upregulation leading to altered seizure threshold with abrupt discontinuation after prolonged use
- Rare reports of significant mood changes (depressive symptoms or emotional blunting)
- Theoretical risk of cholinergic excess at very high doses (excessive salivation, bradycardia, GI hypermotility)
- No completed Phase III trials; long-term safety profile not established
- Preliminary ADHD trial (children with mGluR mutations) showed generally favorable safety but sample size was small
Contraindications
- Known hypersensitivity to racetam-class compounds
- Epilepsy or seizure disorders (may modulate seizure threshold via GABA-B upregulation)
- Severe hepatic impairment (metabolic pathway not fully characterized)
- Severe renal impairment (elimination route not fully established)
- Pregnancy and breastfeeding (no reproductive toxicity data available)
- Children under 18 (except under clinical trial supervision for mGluR-related ADHD)
Drug Interactions
- GABAergic drugs (benzodiazepines, gabapentin, baclofen): Fasoracetam upregulates GABA-B receptors; may potentiate or alter response to GABAergic medications; dose adjustments may be needed
- Phenibut and GHB: Particular concern; fasoracetam's GABA-B upregulation may restore sensitivity to these substances after tolerance; risk of unpredictable potentiation
- Cholinergic drugs (donepezil, rivastigmine): Additive cholinergic effects; monitor for excess cholinergic stimulation
- Anticholinergics: Fasoracetam may counteract anticholinergic drugs, reducing their efficacy
- Stimulants (amphetamines, methylphenidate): May modulate glutamatergic-dopaminergic interactions; combined effects unpredictable
- Other racetams (piracetam, aniracetam): Overlapping mechanisms; excessive cognitive stimulation possible
Population-Specific Considerations
- Adults (nootropic use): Most common user demographic; typical doses 20-100 mg/day; start low and titrate; cycle use recommended (4 weeks on, 1-2 weeks off)
- Children with ADHD (mGluR mutations): Studied in a Phase II trial at doses of 50-800 mg/day; showed tolerability but efficacy data pending
- Elderly: No specific data; caution warranted due to altered pharmacokinetics and increased sensitivity to cognitive-modulating compounds
- Renal/Hepatic impairment: Insufficient data; avoid in severe impairment
- Phenibut/GHB users: Anecdotal reports suggest fasoracetam reverses tolerance to GABA-B agonists; this creates risk of accidental overdose if resuming prior doses
- Pregnant/Lactating: No data; strictly avoid
- Note: Fasoracetam was originally developed by Nippon Shinyaku for vascular dementia but failed Phase III; current interest primarily in ADHD and nootropic communities
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Most users report taking 10-80mg daily, with 40-80mg being the range where ADHD and anxiety benefits become pronounced
Molecular Structure
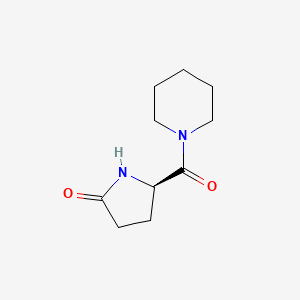
- Formula
- C10H16N2O2
- Weight
- 196.25 Da
- PubChem CID
- 198695
- Exact Mass
- 196.1212 Da
- LogP
- 0
- TPSA
- 49.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 1
- Complexity
- 247
Identifiers (SMILES, InChI)
InChI=1S/C10H16N2O2/c13-9-5-4-8(11-9)10(14)12-6-2-1-3-7-12/h8H,1-7H2,(H,11,13)/t8-/m1/s1
GOWRRBABHQUJMX-MRVPVSSYSA-NSafety Profile
Common Side Effects
- Minimal Reported:: Users consistently note few to no side effects at standard doses, making it well-tolerated compared to other racetams
- Headaches:: Some users report headaches, which may be alleviated by adding a choline source to the stack
- Tolerance Concerns:: While used to prevent tolerance to other substances, long-term fasoracetam use itself has limited data on dependency or tolerance development
- Individual Variability:: A minority of users report feeling no effects or finding it less effective than expected, suggesting individual response variation
References (3)
- [1]Fasoracetam in adolescents with ADHD and glutamatergic gene network variants disrupting mGluR neurotransmitter signaling
→ Clinical trial demonstrated that fasoracetam improved ADHD symptoms in adolescents with specific genetic variants affecting glutamate signaling, providing evidence for personalized medicine approaches in ADHD treatment.
- [2]Beyond stimulants: a systematic review of randomised controlled trials assessing novel compounds for ADHD
→ Systematic review identified fasoracetam among novel non-stimulant options in development for ADHD treatment, representing alternatives for patients who don't respond to or tolerate traditional stimulants.
- [3]A Study of Fasoracetam's Solid State Forms: A Potential Anti-Alzheimer Pharmaceutical
→ Research examined the chemical stability and formulation properties of fasoracetam for potential use in treating Alzheimer's disease, identifying challenges with its low melting point and hydrated forms.
Fadogia Agrestis
Fadogia Agrestis is a shrub native to Africa, traditionally used in herbal medicine as an aphrodisiac and for its purported testosterone-boosting properties. Th
Fat Blaster
Fat Blaster is a lipotropic injection blend combining L-Carnitine, Methionine-Inositol-Choline (MIC), B vitamins (B6, B12), and NADH. This enhanced formulati...