Pterostilbene
A dimethylated analog of resveratrol found in blueberries and grapes with dramatically superior bioavailability, activating SIRT1 and AMPK pathways to support cardiovascular health, neuroprotection, metabolic function, and longevity.
Overview
Pterostilbene (trans-3,5-dimethoxy-4'-hydroxystilbene) is a naturally occurring stilbenoid polyphenol structurally related to resveratrol, differing by the substitution of two hydroxyl groups with methoxy groups. This seemingly minor chemical modification has profound pharmacokinetic consequences: the methoxy groups increase lipophilicity, enhance membrane permeability, and dramatically reduce susceptibility to glucuronidation and sulfation — the phase II metabolic reactions that severely limit resveratrol's oral bioavailability (estimated at <1% for resveratrol versus approximately 80% for pterostilbene). Found naturally in blueberries, grapes, and the heartwood of Pterocarpus marsupium (Indian kijaat), pterostilbene has emerged as a potentially superior alternative to resveratrol for activating the same longevity-associated molecular targets.
Like resveratrol, pterostilbene activates SIRT1 (sirtuin 1) and AMPK (AMP-activated protein kinase), two master metabolic regulators linked to caloric restriction's health benefits. SIRT1 activation promotes mitochondrial biogenesis, enhances fatty acid oxidation, suppresses NF-kB-mediated inflammation, and supports DNA repair through deacetylation of key regulatory proteins including PGC-1alpha, p53, and FOXO transcription factors. Pterostilbene has demonstrated superior potency to resveratrol in several in vitro assays of SIRT1 activation and AMPK phosphorylation. In animal models, pterostilbene supplementation has reduced atherosclerotic lesion formation, lowered LDL cholesterol and triglycerides, improved insulin sensitivity, reduced adiposity, and extended lifespan in some organisms. Its anti-inflammatory profile includes potent inhibition of iNOS, COX-2, and pro-inflammatory cytokines through NF-kB suppression.
Neuroprotective effects represent another area of significant interest. Pterostilbene crosses the blood-brain barrier efficiently (aided by its lipophilicity), and animal studies have demonstrated improvements in working memory, reversal of age-related cognitive decline, and protection against neuroinflammation and oxidative damage. The compound also shows anticancer activity in preclinical models, inducing apoptosis and cell cycle arrest in breast, colon, prostate, and lung cancer cell lines through mechanisms involving p53 activation, Akt/mTOR inhibition, and autophagy induction. Typical supplemental dosing ranges from 50-250 mg/day. Pterostilbene complements other polyphenolic compounds including quercetin, fisetin, and piceatannol, and pairs well with NAD+ precursors like NMN for comprehensive sirtuin pathway support.
Mechanism of Action
Mechanism of Action: Pterostilbene
Pterostilbene (trans-3,5-dimethoxy-4-hydroxystilbene) is a naturally occurring stilbenoid found in blueberries and Pterocarpus marsupium. Its two methoxy groups confer significantly greater lipophilicity and oral bioavailability (80% vs. ~20% for resveratrol) while maintaining potent biological activity.
SIRT1 and AMPK Signaling
Pterostilbene allosterically activates SIRT1, enhancing NAD+-dependent deacetylation of key substrates including PGC-1α (promoting mitochondrial biogenesis), FOXO3a (activating stress resistance genes), and p53 (modulating cell cycle control). Concurrent AMPK activation increases cellular energy sensing, promotes autophagy through ULK1 phosphorylation, and inhibits mTOR signaling.
Anti-inflammatory Mechanisms
Pterostilbene inhibits NF-κB activation at multiple levels: blocking IKKβ kinase activity, preventing IκBα phosphorylation and proteasomal degradation, and reducing p65 nuclear translocation. It also suppresses NLRP3 inflammasome assembly, reducing IL-1β and IL-18 maturation. COX-2 and iNOS expression are markedly decreased.
Antioxidant Defense
Through Nrf2 stabilization and nuclear translocation, pterostilbene upregulates a battery of antioxidant and detoxification genes. Direct radical scavenging activity targets superoxide, hydroxyl radicals, and peroxynitrite. It also chelates transition metals (Fe²⁺, Cu²⁺), preventing Fenton reaction-mediated oxidative damage.
Metabolic Effects
PPARα activation by pterostilbene enhances hepatic and muscle fatty acid oxidation while suppressing SREBP-1c-mediated lipogenesis. It improves insulin signaling by enhancing IRS-1 phosphorylation and GLUT4 translocation. These effects contribute to reductions in triglycerides, LDL-C, and fasting glucose observed in clinical studies.
Neuroprotective Properties
Pterostilbene crosses the blood-brain barrier efficiently due to its lipophilicity. It reduces amyloid-β aggregation, inhibits tau hyperphosphorylation via GSK-3β modulation, and enhances hippocampal brain-derived neurotrophic factor (BDNF) expression, supporting cognitive function and neuroplasticity.
Research
Reported Effects
Synergistic Effects:: Most effective when combined with NAD+ boosters like NR or NMN, with users reporting the combination works better than either alone. Bioavailability Advantage:: Users choose pterostilbene over resveratrol due to its superior absorption and lipophilicity, noting it's more cost-effective despite higher price. Long-term Benefits:: Users report sustained benefits over months to years, particularly for anti-aging goals, with some showing measurable improvements in biomarkers. Individual Variation:: Effectiveness varies significantly between users, with some reporting dramatic results while others notice minimal changes
- Most effective when combined with NAD+ boosters like NR or NMN, with users reporting the combination works better than either alone
- Users choose pterostilbene over resveratrol due to its superior absorption and lipophilicity, noting it's more cost-effective despite higher price
- Users report sustained benefits over months to years, particularly for anti-aging goals, with some showing measurable improvements in biomarkers
- Effectiveness varies significantly between users, with some reporting dramatic results while others notice minimal changes
Safety Profile
Safety Profile: Pterostilbene
Common Side Effects
- Generally well tolerated at typical supplement doses (50–250 mg/day)
- Mild gastrointestinal discomfort including nausea and stomach upset
- Mild headache reported in some clinical trial participants
- Potential increase in LDL cholesterol observed in some studies at higher doses (100–250 mg/day), particularly when taken without concurrent grape extract
Serious Adverse Effects
- Clinically significant increases in LDL cholesterol (+8–18%) observed in randomized trials at 100–250 mg twice daily; lipid monitoring recommended
- Theoretical risk of bleeding potentiation due to antiplatelet properties at high doses
- No significant hepatotoxicity or nephrotoxicity reported in human studies to date
- Allergic reactions possible in individuals sensitive to stilbene compounds or resveratrol
Contraindications
- Known allergy to pterostilbene, resveratrol, or related stilbene polyphenols
- Pre-existing hypercholesterolemia (particularly elevated LDL) without medical supervision
- Active bleeding disorders or upcoming surgery (discontinue 2 weeks prior)
- Pregnancy and lactation (no human safety data available)
Drug Interactions
- Anticoagulants/Antiplatelets (warfarin, aspirin, clopidogrel): May potentiate anticoagulant effects; monitor INR and bleeding signs
- Statins and lipid-lowering agents: Paradoxical LDL increase may counteract statin therapy; monitor lipid panels
- CYP1A2 and CYP3A4 substrates: Pterostilbene may inhibit these enzymes in vitro; clinical significance uncertain but caution with narrow therapeutic index drugs
- Antidiabetic agents: May enhance hypoglycemic effects; monitor blood glucose
- Chemotherapy agents: Potential for both synergistic and antagonistic interactions; use only under oncologist supervision
Population-Specific Considerations
- Elderly: Generally well tolerated; monitor lipid panels and renal function
- Pediatric: No established safety or dosage data; not recommended for children
- Dyslipidemia patients: Monitor LDL cholesterol closely; consider co-administration with grape seed extract to mitigate LDL increases
- Diabetic patients: Monitor blood glucose due to potential hypoglycemic effects
- Hormone-sensitive conditions: Weak estrogenic activity reported in vitro; use cautiously in hormone-sensitive cancers
Pharmacokinetic Profile
Pterostilbene — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Most commonly used at 50-150mg daily, often as part of combination products with NR
Molecular Structure
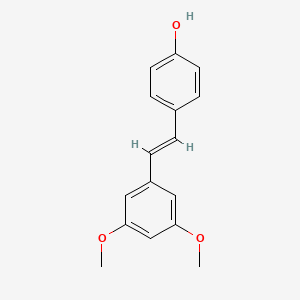
- Formula
- C16H16O3
- Weight
- 256.30 Da
- PubChem CID
- 5281727
- Exact Mass
- 256.1099 Da
- LogP
- 3.8
- TPSA
- 38.7 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 3
- Rotatable Bonds
- 4
- Complexity
- 270
Identifiers (SMILES, InChI)
InChI=1S/C16H16O3/c1-18-15-9-13(10-16(11-15)19-2)4-3-12-5-7-14(17)8-6-12/h3-11,17H,1-2H3/b4-3+
VLEUZFDZJKSGMX-ONEGZZNKSA-NSafety Profile
Common Side Effects
- Minimal Reported:: Very few direct side effects attributed to pterostilbene specifically in user reports
- Stack Interactions:: Users occasionally report difficulty determining which compound in complex stacks causes effects or side effects
- Cost Concerns:: High cost is the main complaint, with users seeking more affordable alternatives or questioning cost-effectiveness
- Limited Standalone Data:: Most users take it in combination, making it difficult to isolate specific side effects
References (8)
- [3]A randomized placebo-controlled trial of nicotinamide riboside and pterostilbene supplementation in experimental muscle injury in elderly individuals
→ Clinical trial examining NR and pterostilbene supplementation effects on muscle regeneration and stem cell function in elderly individuals following muscle injury.
- [7]Dietary Pterostilbene for MTA1-Targeted Interception in High-Risk Premalignant Prostate Cancer
→ Dietary pterostilbene showed promise for cancer prevention in high-risk premalignant prostate cancer through targeted molecular mechanisms, supporting its potential for nutritional intervention.
- [2]Pterostilbene Targets Hallmarks of Aging in the Gene Expression Landscape in Blood of Healthy Rats
→ Pterostilbene supplementation in healthy rats modulated gene expression related to key aging hallmarks, suggesting anti-aging effects at the molecular level through epigenetic mechanisms.
- [4]Pterostilbene, a dimethylated analog of resveratrol, promotes energy metabolism in obese rats
→ Study found pterostilbene significantly suppressed abdominal fat accumulation, increased oxygen consumption and energy expenditure, and promoted fat metabolism over carbohydrate metabolism in obese rats.
- [5]Pterostilbene-induced changes in gut microbiota composition in relation to obesity
→ Pterostilbene supplementation induced beneficial changes in gut microbiota composition associated with improvements in obesity-related metabolic parameters.
- [8]Pterostilbene enhances reproductive outcomes and oocyte quality in aged mice without adverse effects
→ Long-term pterostilbene supplementation improved reproductive outcomes, increased implantation and live birth rates in aged mice, and enhanced oocyte quality without affecting estrous cycles or offspring health.
- [1]Dietary Supplementation With NAD+-Boosting Compounds in Humans: Current Knowledge and Future Directions
→ Review examining NAD+ precursor supplementation including nicotinamide riboside co-administered with pterostilbene, finding these compounds are common candidates for improving physiologic function in aging and age-related diseases.
- [6]AMPK-Dependent Epigenetic Regulation of Metabolism Mediates the Anti-Cancer Action of Pterostilbene in Hepatocellular Carcinoma
→ Pterostilbene demonstrated anti-cancer effects in hepatocellular carcinoma through AMPK-dependent epigenetic regulation of metabolism, suggesting potential therapeutic applications.
PTD-DBM
**PTD-DBM** (Protein Transduction Domain-fused Dishevelled Binding Motif) is a cell-permeable peptide engineered to activate the canonical Wnt/beta-catenin sign
PTH 1-34 (Teriparatide / Forteo)
**Teriparatide (PTH 1-34)** is a recombinant peptide consisting of the first 34 amino acids of the 84-amino acid human parathyroid hormone. Marketed as Forteo (