DGL (Deglycyrrhizinated Licorice)
DGL is a processed form of licorice root (Glycyrrhiza glabra) with glycyrrhizin removed, used to support gastrointestinal mucosal health without the aldosterone-like side effects of whole licorice.
Overview
Deglycyrrhizinated licorice (DGL) is derived from licorice root through a process that removes glycyrrhizin, the compound responsible for licorice's characteristic sweetness and its potential to cause hypertension, hypokalemia, and fluid retention. By eliminating glycyrrhizin, DGL retains the beneficial flavonoids and other bioactive constituents while dramatically improving its safety profile for long-term use.
DGL is primarily used to support the health of the gastrointestinal mucosa. Research suggests that its flavonoid compounds may stimulate the production of mucin, the protective glycoprotein lining the stomach and intestinal walls. This mechanism has made DGL a popular complementary approach for individuals dealing with gastric ulcers, gastroesophageal reflux, and functional dyspepsia.
DGL is commonly available in chewable tablet form, as oral contact with saliva is thought to enhance its efficacy by promoting the release of salivary compounds that support mucosal repair. Typical dosages range from 380 to 760 mg taken before meals. While generally well tolerated, DGL should not be confused with whole licorice extract, which carries significant risks when used chronically at high doses.
Mechanism of Action
Mucosal Defense Enhancement
Deglycyrrhizinated licorice (DGL) is a processed form of Glycyrrhiza glabra root from which glycyrrhizin (the mineralocorticoid-active component) has been removed, retaining the flavonoid and saponin fractions. The primary active compounds include liquiritigenin, isoliquiritigenin, glabridin, and licoricidin, which enhance gastrointestinal mucosal defense through multiple complementary mechanisms (PMID: 22235230).
Prostaglandin-Mediated Cytoprotection
DGL flavonoids stimulate prostaglandin E2 (PGE2) synthesis in gastric mucosal cells through upregulation of constitutive cyclooxygenase-1 (COX-1) activity. PGE2 activates EP3 and EP4 receptors on surface epithelial cells, stimulating mucus and bicarbonate secretion, enhancing mucosal blood flow, and promoting epithelial cell proliferation. This gastroprotective prostaglandin pathway operates independently of acid suppression (PMID: 8894209).
Mucin Secretion & Glycoprotein Synthesis
DGL increases both the quantity and quality of mucin glycoprotein secretion by goblet cells throughout the gastrointestinal tract. It stimulates mucin gene (MUC5AC, MUC6) expression and enhances post-translational glycosylation, producing a thicker, more viscous mucus barrier that resists pepsin and acid penetration. The increased mucus turnover improves the unstirred layer protecting the epithelial surface (PMID: 20190725).
Anti-Helicobacter & Anti-Inflammatory Activity
Glabridin and licoricidin exhibit direct antibacterial activity against Helicobacter pylori by disrupting bacterial membrane integrity and inhibiting bacterial urease activity. Isoliquiritigenin inhibits NF-kB and HMGB1 signaling, reducing mucosal inflammation. DGL also suppresses leukotriene synthesis by inhibiting 5-lipoxygenase, complementing its prostaglandin-enhancing effects.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Symptom Management:: Provides relief from acute reflux and gastritis symptoms but may not heal underlying damage completely for chronic conditions. Individual Variation:: Effectiveness varies significantly between users, with some achieving complete symptom resolution while others see minimal improvement. Time to Effect:: Users typically report effects within 3 days to 2 weeks, though sustained healing may require longer-term use. Limited Standalone Efficacy:: Research shows mixed results, with some clinical trials finding no significant benefit compared to placebo for ulcer healing
- Provides relief from acute reflux and gastritis symptoms but may not heal underlying damage completely for chronic conditions
- Effectiveness varies significantly between users, with some achieving complete symptom resolution while others see minimal improvement
- Users typically report effects within 3 days to 2 weeks, though sustained healing may require longer-term use
- Research shows mixed results, with some clinical trials finding no significant benefit compared to placebo for ulcer healing
Safety Profile
Safety Profile: DGL (Deglycyrrhizinated Licorice)
Common Side Effects
- Mild gastrointestinal effects: nausea, bloating, or increased bowel movements
- Headache (infrequent)
- Allergic reactions in individuals sensitive to licorice or legumes (Fabaceae family)
- Unpleasant taste (licorice flavor may cause nausea in sensitive individuals)
- Rarely, mild elevation in blood pressure if glycyrrhizin removal is incomplete
Serious Adverse Effects
- Residual glycyrrhizin risk: If manufacturing quality is poor, DGL products may contain enough glycyrrhizin to cause pseudoaldosteronism (hypertension, hypokalemia, edema)
- Severe allergic reaction or anaphylaxis (very rare)
- Theoretical risk of hormonal effects if glycyrrhizin contamination exceeds 3% of total content
- No organ toxicity reported with properly manufactured DGL products
- Prolonged high-dose use safety data are limited
Contraindications
- Known allergy to licorice or Fabaceae (legume) family plants
- Severe hypertension (precautionary, even though glycyrrhizin is reduced)
- Severe hypokalemia or conditions predisposing to potassium loss
- Cholestatic liver disease
- Pregnancy (precautionary — even low glycyrrhizin levels may affect fetal development)
Drug Interactions
- Diuretics (thiazides, loop diuretics): Even trace glycyrrhizin may exacerbate potassium loss
- Digoxin: Hypokalemia (if glycyrrhizin contamination) increases digoxin toxicity risk
- Corticosteroids: Potential additive mineralocorticoid effects with residual glycyrrhizin
- Antihypertensives: May slightly counteract effects if glycyrrhizin content is significant
- Warfarin: Some licorice compounds may affect CYP metabolism; monitor INR
- MAO inhibitors: Theoretical interaction (licorice flavonoids may have mild MAO-inhibitory activity)
- DGL interactions are significantly less concerning than whole licorice due to glycyrrhizin removal
Population-Specific Considerations
- GI patients: Primary use case (peptic ulcers, gastritis, GERD); chewable tablets before meals are the standard approach
- Hypertensive patients: Verify product certification for glycyrrhizin content (<3%); monitor blood pressure
- Elderly: Monitor potassium, especially if on diuretics or cardiac medications
- Pregnant women: Avoid unless specifically recommended by healthcare provider
- Children: Limited data; occasional use at low doses generally considered safe for ages 6+
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 760mg three times daily is the most commonly cited clinical dose, typically taken before meals
Molecular Structure
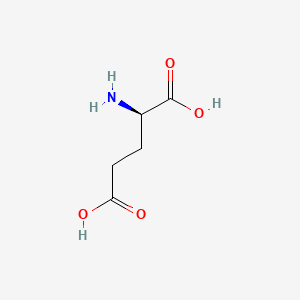
- Formula
- C5H9NO4
- Weight
- 147.13 Da
- PubChem CID
- 23327
- Exact Mass
- 147.0532 Da
- LogP
- -3.7
- TPSA
- 101 Ų
- H-Bond Donors
- 3
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 4
- Complexity
- 145
Identifiers (SMILES, InChI)
InChI=1S/C5H9NO4/c6-3(5(9)10)1-2-4(7)8/h3H,1-2,6H2,(H,7,8)(H,9,10)/t3-/m1/s1
WHUUTDBJXJRKMK-GSVOUGTGSA-NSafety Profile
Common Side Effects
- Minimal Direct Effects:: DGL is generally well-tolerated with few reported side effects due to removal of glycyrrhizin
- Rebound Symptoms:: Some users experience return of symptoms when discontinuing DGL, suggesting symptom management rather than cure
- Digestive Changes:: Occasional reports of altered bowel movements or mild nausea when first starting supplementation
- No Blood Pressure Issues:: Unlike regular licorice, DGL does not cause hypertension or potassium imbalances due to deglycyrrhization process
References (3)
- [1]Effectiveness of Nutritional Ingredients on Upper Gastrointestinal Conditions and Symptoms: A Narrative Review
→ Review of nutritional ingredients for upper GI complaints found limited robust evidence for most botanicals including DGL, though some ingredients showed potential for managing digestive symptoms when combined with other compounds.
- [2]Double-blind trial of deglycyrrhizinated liquorice in gastric ulcer
→ Clinical trial of 760mg DGL three times daily in patients with chronic gastric ulcers found no significant healing effect compared to placebo, with no demonstrated benefit for ulcer healing or symptom improvement.
- [3]A Brief Review of Nutraceutical Ingredients in Gastrointestinal Disorders: Evidence and Suggestions
→ Review examining nutraceutical ingredients for GI disorders found DGL among commonly used botanicals, though evidence quality varies and more rigorous studies are needed to establish efficacy.
Devil's Claw
Devil's claw (Harpagophytum procumbens) is a medicinal plant native to southern Africa, primarily used for its anti-inflammatory and analgesic properties. The p
DHA (Docosahexaenoic Acid)
Docosahexaenoic acid (DHA) is a primary omega-3 long-chain polyunsaturated fatty acid that serves as a structural component of the human brain, cerebral cortex,