Melanotan II
A synthetic cyclic heptapeptide analog of alpha-MSH that non-selectively activates melanocortin receptors, producing skin tanning, appetite suppression, and pro-erectile effects, widely used in research and non-medical tanning contexts.
Overview
Melanotan II (MT-II) is a synthetic cyclic heptapeptide developed at the University of Arizona as a more potent, shorter analog of alpha-melanocyte-stimulating hormone (alpha-MSH). Its cyclic structure — incorporating a lactam bridge between positions 4 and 10 — confers enhanced receptor binding affinity, metabolic stability, and broader melanocortin receptor activation compared to the linear Melanotan I. MT-II is a non-selective agonist at MC1R (melanogenesis), MC3R (energy homeostasis), MC4R (appetite, sexual function, erectile response), and MC5R (exocrine secretion), producing a diverse pharmacological profile that extends well beyond skin pigmentation.
The multi-receptor activity of Melanotan II has generated research interest across several therapeutic domains. Its MC1R-mediated tanning effect is the most visible outcome, producing significant melanogenesis and darkening of skin and hair. The MC4R-mediated pro-erectile effect led to the development of bremelanotide (PT-141), a derivative that was FDA-approved in 2019 as Vyleesi for hypoactive sexual desire disorder in premenopausal women — the first melanocortin-based drug approved for sexual dysfunction. MT-II's appetite-suppressive effects via MC3R/MC4R have been explored in obesity research, though development for this indication has not advanced to approval. In preclinical studies, melanocortin receptor activation has also demonstrated anti-inflammatory and neuroprotective effects, adding further dimensions to MT-II's pharmacological interest.
Despite its investigational status and lack of regulatory approval for cosmetic tanning, Melanotan II has become widely available through online peptide suppliers and is self-administered subcutaneously by individuals seeking UV-free tanning. This unregulated use raises significant safety concerns: reported adverse effects include nausea, facial flushing, fatigue, changes in existing moles (nevi) that complicate melanoma screening, and priapism from the pro-erectile effect. There are also case reports linking MT-II to melanoma development, though a causal relationship has not been established. The typical self-administration protocol involves a loading phase of 0.25–0.5 mg subcutaneously daily for 1–2 weeks, followed by maintenance dosing of 0.5 mg once or twice weekly. Medical supervision is strongly recommended, and dermatological monitoring of nevi is essential for anyone using melanocortin agonists.
Mechanism of Action
Melanotan II (MT-II) is a synthetic cyclic heptapeptide analog of alpha-MSH with the sequence Ac-Nle-c[Asp-His-D-Phe-Arg-Trp-Lys]-NH2. Unlike the MC1R-selective Melanotan I, MT-II is a non-selective agonist that activates melanocortin receptors MC1R, MC3R, MC4R, and MC5R with varying affinities, producing a broader spectrum of physiological effects. At MC1R on melanocytes, it triggers the same cAMP/PKA/MITF/tyrosinase cascade as Melanotan I, stimulating eumelanin production and skin darkening without UV exposure.
The central nervous system effects of MT-II are mediated primarily through MC3R and MC4R in the hypothalamus. MC4R activation in the paraventricular nucleus (PVN) stimulates pro-opiomelanocortin (POMC) neuronal circuits that suppress appetite and increase energy expenditure through sympathetic nervous system activation. MC4R signaling also activates oxytocinergic pathways in the PVN that project to the spinal cord, mediating erectile function through increased parasympathetic outflow to penile vasculature. This mechanism involves nitric oxide (NO) release from cavernosal endothelium and smooth muscle relaxation via the NO/cGMP pathway, producing the pro-erectile effects that led to development of the related drug bremelanotide. MC3R activation in the arcuate nucleus modulates energy homeostasis and feeding behavior through distinct but overlapping circuits.
The non-selective receptor profile of MT-II accounts for both its broad effects and its side effect profile. MC3R/MC4R activation can produce nausea through area postrema stimulation, facial flushing via peripheral vasodilation, and fatigue. Chronic MC1R stimulation may promote melanocytic nevus darkening. The compound's cyclic structure provides resistance to enzymatic degradation, giving it a longer duration of action than linear melanocortin peptides.
Reconstitution Calculator
Melanotan II
Melanotan II is a synthetic analog of alpha-melanocyte-stimulating hormone (α-MS

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Safety Profile
Common side effects include nausea, facial flushing, spontaneous erections, and darkening of moles. Health authorities advise against its use due to serious risks such as the potential to promote melanoma development, kidney dysfunction, and cardiovascular issues. Product quality from unregulated sources is not guaranteed, and its sale is restricted in many countries.
Pharmacokinetic Profile
Melanotan II — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Loading: Start 0.25mg daily, increase to 0.5-1mg; Maintenance: 0.5-1mg 2-3x weekly
- Frequency
- Loading phase: Daily for first week, then tanning maintenance 2-3x weekly or as needed for sexual enhancement
- Cycle Length
- 4-8 weeks loading phase for tanning, then ongoing maintenance as needed
- Storage
- Refrigerate at 2-8°C wrapped in aluminum foil (light-sensitive); use reconstituted solution within 4 weeks
Molecular Structure
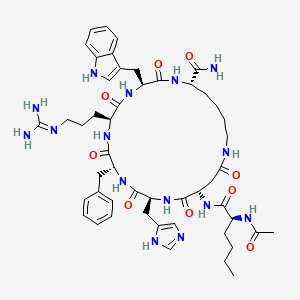
- Formula
- C50H69N15O9
- Weight
- 1024.18 Da
- Length
- 7 amino acids
- PubChem CID
- 92432
- Exact Mass
- 1023.5403 Da
- LogP
- 0
- TPSA
- 385 Ų
- H-Bond Donors
- 13
- H-Bond Acceptors
- 11
- Rotatable Bonds
- 17
- Complexity
- 1950
Identifiers (SMILES, InChI)
InChI=1S/C50H69N15O9/c1-3-4-16-36(59-29(2)66)44(69)65-41-25-42(67)55-20-11-10-18-35(43(51)68)60-47(72)39(23-31-26-57-34-17-9-8-15-33(31)34)63-45(70)37(19-12-21-56-50(52)53)61-46(71)38(22-30-13-6-5-7-14-30)62-48(73)40(64-49(41)74)24-32-27-54-28-58-32/h5-9,13-15,17,26-28,35-41,57H,3-4,10-12,16,18-25H2,1-2H3,(H2,51,68)(H,54,58)(H,55,67)(H,59,66)(H,60,72)(H,61,71)(H,62,73)(H,63,70)(H,64,74)(H,65,69)(H4,52,53,56)/t35-,36-,37-,38+,39-,40-,41-/m0/s1
JDKLPDJLXHXHNV-MFVUMRCOSA-NResearch Indications
Skin Health
Stimulates natural melanin production for tanning without requiring UV exposure.
Increased melanin provides natural SPF protection against sun damage.
May help address certain pigmentation disorders.
Sexual Health
Improves sexual desire in both men and women through MC4R activation.
80% response rate in psychogenic erectile dysfunction studies.
73% of women reported arousal within 24 hours in clinical trials.
Metabolic
MC4R activation in hypothalamus reduces appetite (15% caloric intake reduction in studies).
Enhanced fat oxidation through metabolic pathway activation.
Research Protocols
intranasal Injection
Nasal spray offers convenient administration with rapid absorption, though with less research data.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Nasal administration | 1-2mg | 1-2x daily | —(Route: Nasal spray) |
topical
Topical application provides localized effects with minimal systemic absorption.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Localized tanning | 1-2mg/mL solution | 1-2x daily | —(Route: Topical application) |
subcutaneous Injection
Melanocortin receptor agonist for tanning research. Loading phase followed by maintenance dosing.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Week 1 | 250 mcg | Once daily | Week 1 |
| Week 2 | 500 mcg | Once daily | Week 2 |
| Week 3 | 750 mcg | Once daily | Week 3 |
| Loading phase | 1,000 mcg | Once daily | Weeks 4-8 |
| Maintenance | 500-1,000 mcg | 1-2x weekly | Ongoing(Do not exceed 2 mg/day. Loading: 6-8 weeks.) |
Reconstitution Guide (10mg vial + 3mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 3.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 3.33 mg/mL
- For 250 mcg dose: draw 7.5 units (0.075 mL)
- For 500 mcg dose: draw 15 units (0.15 mL)
- For 1,000 mcg dose: draw 30 units (0.30 mL)
- Store reconstituted vial refrigerated at 2-8°C
Interactions
What to Expect
What to Expect
Possible nausea, flushing, fatigue
Increased spontaneous erections (men), enhanced arousal
Noticeable skin darkening, reduced appetite
Significant tanning, stabilized sexual effects
Maintained tan with less frequent dosing
Safety Profile
Common Side Effects
- Nausea (pre-treatment with antiemetics recommended)
- Facial flushing
- Temporary blood pressure elevation
- Fatigue
- Spontaneous erections
Contraindications
- History of melanoma or dysplastic nevi
- Pregnancy or breastfeeding
- Cardiovascular conditions
- Uncontrolled hypertension
Discontinue If
- Severe persistent nausea or vomiting
- Chest pain or significant blood pressure elevation
- Mole changes (size, shape, color) - monitor closely
- Prolonged painful erections (priapism)
- Severe headaches or vision changes
- Allergic reactions (rash, swelling, breathing difficulty)
Quality Indicators
What to look for
- White to off-white lyophilized powder
- Clear to pale yellow solution when reconstituted
- Vacuum sealed vial with audible 'pop' when opening
Caution
- Protect from light exposure - MT-II is photosensitive
- Wrap vials in foil for storage
Red flags
- Brown or dark powder indicates oxidation/impurities
- Cloudy solution after mixing indicates degradation/contamination
References (8)
- [2]Melanogenesis & UV Protection Study (1999)
- [3]Female Sexual Arousal Disorder Trial (2004)
- [4]Appetite Reduction Study (2001)
- [5]Melanocortin Receptor Selectivity
- [6]Vitiligo Repigmentation Study
- [7]Cardiovascular Effects Assessment (2018)
- [8]Photoprotection in Fair-Skinned Individuals (2015)
- [1]
Melanotan I
Synthetic analog of α-MSH that selectively targets MC1 receptors for melanin stimulation. FDA-approved as SCENESSE implant for erythropoietic protoporphyria (EP
Melatonin
Melatonin is a naturally occurring neurohormone synthesized by the pineal gland that regulates circadian rhythms and the sleep-wake cycle. It acts as a chronobi