MF59 + Peptide Vaccines
MF59 is a squalene-based oil-in-water emulsion adjuvant that enhances immune responses to peptide vaccines. It is used in licensed influenza vaccines and researched for HIV, cancer, and pandemic preparedness applications.
Overview
MF59 was developed in the 1990s as a next-generation vaccine adjuvant to overcome the limitations of aluminum salts, the only adjuvant licensed for human use for over 70 years. First described by Ott et al. (1995), MF59 consists of uniformly sized (~160 nm) oil-in-water droplets that create an immunostimulatory environment at the injection site. Unlike alum, which primarily enhances antibody responses, MF59 activates both innate and adaptive immunity, producing robust CD4+ T-cell responses alongside high-titer, cross-reactive antibodies. MF59 received its first regulatory approval in 1997 as part of Fluad (adjuvanted trivalent influenza vaccine) in Italy, and has since been licensed in over 30 countries. More than 100 million doses have been administered to date O'Hagan et al. (2012).
Mechanism of Action
MF59 enhances immune responses through multiple complementary mechanisms that distinguish it from traditional adjuvants:
-
Immunocompetent cell recruitment: MF59 creates a transient local immunostimulatory environment at the injection site, recruiting monocytes, macrophages, and granulocytes within hours of administration. This cellular influx is significantly greater than that induced by alum or incomplete Freund's adjuvant Calabro et al. (2011).
-
Antigen-presenting cell activation: MF59 promotes differentiation of monocytes into dendritic cells and enhances antigen uptake and processing by APCs. MF59-activated DCs show upregulated expression of MHC class II, CD86, and CD40, improving antigen presentation to T cells Seubert et al. (2008).
-
Cytokine and chemokine induction: At the injection site, MF59 induces production of CCL2 (MCP-1), CCL3 (MIP-1alpha), CCL4 (MIP-1beta), and CXCL8 (IL-8), creating a chemokine gradient that attracts immune cells. ATP release from muscle cells acts as an endogenous danger signal Vono et al. (2013).
-
Enhanced antigen uptake: MF59 promotes receptor-mediated and macropinocytic uptake of co-administered antigens by APCs, increasing the efficiency of antigen delivery to draining lymph nodes by approximately 100-fold compared to unadjuvanted antigen Dupuis et al. (1998).
-
Germinal center enhancement: MF59 promotes robust germinal center reactions in draining lymph nodes, leading to improved affinity maturation, broader epitope recognition, and generation of long-lived plasma cells and memory B cells Khurana et al. (2011).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Pandemic Preparedness
MF59 played a critical role during the 2009 H1N1 pandemic. Focetria, an MF59-adjuvanted monovalent H1N1 vaccine, achieved seroprotection after a single dose in >90% of adults, enabling dose-sparing strategies critical during pandemic antigen shortages. MF59 adjuvantation allowed a 4-fold reduction in antigen dose while maintaining equivalent immunogenicity Clark et al. (2009).
For H5N1 avian influenza preparedness, MF59-adjuvanted vaccines demonstrated seroconversion in 50-86% of subjects at antigen doses as low as 3.75 microg, compared to no detectable response at 45 microg without adjuvant Bernstein et al. (2008). This dose-sparing capacity is critical for pandemic planning, potentially expanding vaccine supply 4-8 fold.
HIV Peptide Vaccines
MF59 has been extensively evaluated as an adjuvant for HIV peptide and subunit vaccines. In the RV144 correlate analyses, oil-in-water emulsions similar to MF59 were identified as critical for inducing the V1V2-specific IgG antibodies correlated with protection. MF59-adjuvanted HIV gp120 vaccines consistently induced higher and more durable antibody responses than alum-adjuvanted formulations, with significantly enhanced CD4+ T-cell responses Ott et al. (2013).
In HIV peptide vaccine studies, MF59 enhanced binding antibody responses to multiple HIV envelope epitopes and promoted antibody-dependent cellular cytotoxicity (ADCC) activity, a correlate of protection in the RV144 trial Wiehe et al. (2014).
Cancer Peptide Vaccines
MF59 has been investigated as an adjuvant for cancer peptide vaccines targeting tumor-associated antigens. In melanoma studies, MF59-adjuvanted MAGE-A3 peptide vaccine induced antigen-specific CD4+ T-cell responses in a subset of patients with resected melanoma Vantomme et al. (2004). MF59's ability to enhance CD4+ T-cell help is particularly relevant for cancer vaccines, as CD4+ responses are critical for sustaining cytotoxic T-lymphocyte activity against tumors.
CD4+ T-Cell Enhancement
A distinguishing feature of MF59 compared to alum is its superior induction of CD4+ T-cell responses. In head-to-head clinical comparisons, MF59-adjuvanted influenza vaccines induced 2-4 fold higher frequencies of antigen-specific CD4+ T cells compared to alum-adjuvanted or unadjuvanted vaccines. These CD4+ T cells displayed a polyfunctional Th1 phenotype, producing IFN-gamma, TNF-alpha, and IL-2 simultaneously Galli et al. (2009).
Cross-Reactive Antibody Induction
MF59's most clinically significant advantage is its ability to elicit antibodies that recognize antigenically drifted virus strains. In influenza vaccination studies, MF59-adjuvanted vaccines induced antibodies with broader epitope recognition than unadjuvanted vaccines, covering antigenically distinct strains within the same subtype. This cross-reactivity results from MF59's enhancement of germinal center reactions and somatic hypermutation, producing antibodies with higher affinity and broader specificity Khurana et al. (2011).
Influenza Vaccination in the Elderly
The primary clinical application of MF59 is in Fluad, the adjuvanted influenza vaccine for adults aged 65 and older. Immunosenescence reduces conventional vaccine efficacy to 30-40% in elderly populations. MF59-adjuvanted influenza vaccines produce significantly higher seroconversion rates and geometric mean titers compared to unadjuvanted vaccines in this age group. A meta-analysis of 64 clinical studies involving over 30,000 elderly subjects demonstrated that MF59-adjuvanted vaccines were 63% more effective at preventing influenza-related hospitalization than conventional vaccines Domnich et al. (2017).
MF59 is particularly effective at generating cross-reactive antibodies against drifted influenza strains. In elderly subjects, MF59-adjuvanted vaccine induced seroprotective titers against heterologous H3N2 strains in 40-50% of recipients, compared to <15% with unadjuvanted vaccine Del Giudice et al. (2018).
Comparison with Aluminum Adjuvants
MF59 and alum adjuvants differ fundamentally in mechanism and immunological outcome:
- Mechanism: Alum acts primarily through NLRP3 inflammasome activation and antigen depot formation. MF59 acts through chemokine-mediated immune cell recruitment and enhanced antigen uptake without inflammasome involvement Seubert et al. (2008).
- Antibody responses: MF59 induces higher-titer, more broadly neutralizing antibodies with greater cross-reactivity to variant antigens.
- T-cell responses: MF59 generates significantly stronger CD4+ T-cell responses with Th1 polarization; alum preferentially drives Th2 responses.
- Reactogenicity: MF59 produces more injection-site pain and erythema than alum, but systemic adverse events are comparable.
- Antigen interaction: MF59 does not require antigen adsorption (unlike alum), simplifying vaccine formulation and preserving antigen conformation.
Safety Profile
MF59 has an extensive safety record spanning nearly three decades and over 100 million administered doses. The most common adverse events are mild, transient injection-site reactions (pain, erythema, induration) occurring in 30-50% of recipients, typically resolving within 2-3 days. Systemic reactions (myalgia, headache, fatigue) occur at slightly higher rates than with unadjuvanted vaccines but are generally mild. Post-marketing surveillance data from Fluad, including a study of over 170,000 elderly subjects, found no association with Guillain-Barre syndrome, autoimmune disorders, or other serious adverse events Pellegrini et al. (2009). Squalene is a naturally occurring lipid found in human sebum and liver, and anti-squalene antibodies observed in some recipients are not associated with adverse clinical outcomes. MF59 is not licensed for use in children under 6 months; pediatric safety data in children 6-23 months show an acceptable safety profile comparable to adults Vesikari et al. (2011).
Pharmacokinetic Profile
- Half-life
- Not applicable (depot-forming adjuvant)
Quick Start
- Route
- Intramuscular injection
Molecular Structure
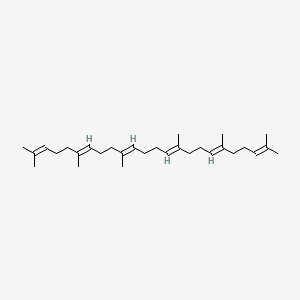
- Formula
- C30H50
- Weight
- 410.7 Da
- PubChem CID
- 638072
- Exact Mass
- 410.3913 Da
- LogP
- 11.6
- TPSA
- 0 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 0
- Rotatable Bonds
- 15
- Complexity
- 578
Identifiers (SMILES, InChI)
InChI=1S/C30H50/c1-25(2)15-11-19-29(7)23-13-21-27(5)17-9-10-18-28(6)22-14-24-30(8)20-12-16-26(3)4/h15-18,23-24H,9-14,19-22H2,1-8H3/b27-17+,28-18+,29-23+,30-24+
YYGNTYWPHWGJRM-AAJYLUCBSA-NResearch Indications
Vaccine Adjuvant
Fluad is licensed in 30+ countries for adults 65+. Meta-analysis of 20 trials (>10,000 elderly) confirmed greater immunogenicity vs non-adjuvanted vaccines, especially in those with low pre-immunization titers or chronic diseases.
Fluad Pediatric approved for infants 6-23 months. MF59 adjuvant overcomes immature immune responses in young children, improving seroconversion rates significantly.
MF59-adjuvanted vaccines (Celtura for H1N1, Audenz for H5N1) enable dose-sparing strategies and broader cross-reactive immunity against heterologous strains during pandemics.
Immunocompromised Populations
MF59-adjuvanted influenza vaccine showed improved immunogenicity and seroprotection in HIV-infected patients compared to standard vaccines.
Chronic kidney disease patients on hemodialysis achieved better antibody responses with MF59-adjuvanted vaccines after failing standard immunization.
Research Protocols
intramuscular Injection
Clinical Research Protocols Influenza Vaccine Studies in the Elderly In the pivotal Fluad clinical trials, elderly subjects (65+ years) received a single 0.5 mL intramuscular dose of MF59-adjuvanted trivalent influenza vaccine containing 15 microg hemagglutinin per strain. Pandemic H5N1 Dose-Rangin
Interactions
Peptide Interactions
A distinguishing feature of MF59 compared to alum is its superior induction of CD4+ T-cell responses. In head-to-head clinical comparisons, MF59-adjuvanted influenza vaccines induced 2-4 fold higher frequencies of antigen-specific CD4+ T cells compared to alum-adjuvanted or unadjuvanted vaccines....
What to Expect
What to Expect
Within 6 hours of intramuscular injection, MF59 induces release of ATP and other danger-associated molecular patterns (DAMPs) from muscle cells.
The most common adverse events are mild, transient injection-site reactions (pain, erythema, induration) occurring in 30-50% of recipients, typically...
At the lowest dose (3.75 microg + MF59), 55% of subjects achieved seroconversion by day 42, compared to 0% with 45 microg unadjuvanted antigen.
A: MF59-adjuvanted influenza vaccines (Fluad Pediatric) have been evaluated in clinical trials enrolling over 20,000 children aged 6 months to 5...
Continued use as directed
Quality Indicators
What to look for
- Human clinical trials conducted
- Naturally occurring compound
- Extensive peer-reviewed research base
Caution
- Injection site reactions reported
Red flags
- Significant side effect risk noted
Frequently Asked Questions
References (17)
- [7]Khurana et al *Sci Sci. Transl. Med. (2011)
- [17]Vantomme et al *Cancer Immunol Cancer Immunol. Immunother. (2004)
- [9]Del Giudice et al *Hum Hum. Vaccin. Immunother. (2018)
- [10]Clark et al *Lancet*, 374(9708), 2089-2094 Lancet (2009)
- [13]Pellegrini et al *Vaccine*, 27(25-26), 3459-3468 Vaccine (2009)
- [14]Podda *Vaccine*, 19(17-19), 2673-2680 Vaccine (2001)
- [15]Keefer et al *AIDS Res AIDS Res. Hum. Retroviruses (1996)
- [16]Galli et al *Proc Proc. Natl. Acad. Sci. USA (2009)
- [19]
- [20]
- [1]
- [2]O'Hagan et al *Expert Rev Expert Rev. Vaccines (2012)
- [3]
- [4]Seubert et al *J J. Immunol. (2008)
- [5]Vono et al *FASEB J.*, 27(4), 1272-1281 FASEB J. (2013)
- [6]
- [18]Ko et al — MF59 adjuvant enhances breadth of antibody response to SARS-CoV-2 variants in preclinical models (2023)
Methylene Blue
Methylene Blue is a synthetic compound originally developed as a textile dye that functions as a mitochondrial enhancer and mild MAOI (monoamine oxidase inhibit
MGF
Non-pegylated IGF-1 splice variant produced locally in muscle tissue following mechanical stress. Has a very short half-life compared to PEG-MGF, requiring more