I3C
Indole-3-carbinol, a cruciferous vegetable compound that modulates estrogen metabolism and supports cellular detoxification through the aryl hydrocarbon receptor pathway.
Overview
Indole-3-carbinol (I3C) is a naturally occurring glucosinolate breakdown product formed when cruciferous vegetables — broccoli, cauliflower, cabbage, Brussels sprouts, and kale — are crushed, chewed, or digested. The parent compound glucobrassicin is hydrolyzed by the enzyme myrosinase to yield I3C, which then undergoes acid-catalyzed condensation in the stomach to produce a range of bioactive oligomeric products, most notably 3,3'-diindolylmethane (DIM). Both I3C and DIM are potent modulators of estrogen metabolism, shifting the ratio of estrogen metabolites away from the genotoxic 16α-hydroxyestrone toward the protective 2-hydroxyestrone pathway via induction of CYP1A1 and CYP1A2 enzymes.
The estrogen-modulatory effects of I3C have driven considerable research interest in hormone-dependent cancers. Epidemiological studies consistently associate higher cruciferous vegetable intake with reduced risks of breast, prostate, cervical, and colorectal cancers. Mechanistically, I3C activates the aryl hydrocarbon receptor (AhR), induces Phase I and Phase II detoxification enzymes (including glutathione S-transferases and UDP-glucuronosyltransferases), suppresses NF-κB-mediated inflammation, and promotes cell cycle arrest and apoptosis in cancer cell lines through modulation of CDK6, p21, and p27 pathways. Clinical trials in women with cervical intraepithelial neoplasia (CIN) demonstrated complete regression in nearly 50% of patients receiving 200–400 mg/day of I3C for 12 weeks.
Supplemental I3C is typically dosed at 200–400 mg/day and is commonly used in integrative protocols for estrogen dominance, PMS, fibrocystic breast changes, and as adjunctive cancer prevention. However, there is ongoing debate about whether I3C or its metabolite DIM is the preferred supplement, since I3C's gastric conversion produces variable and sometimes unpredictable metabolite profiles, while DIM supplementation provides the primary active metabolite directly. I3C also supports liver detoxification and pairs well with calcium-d-glucarate (which inhibits beta-glucuronidase to prevent estrogen reabsorption) and sulforaphane (another cruciferous compound with complementary Nrf2-activating properties) in comprehensive hormonal health and detoxification protocols.
Mechanism of Action
Indole-3-carbinol (I3C) is a glucosinolate breakdown product found in cruciferous vegetables such as broccoli, cabbage, and Brussels sprouts. In the acidic environment of the stomach, I3C is converted to its primary active metabolite, 3,3'-diindolylmethane (DIM), along with other oligomeric products. Both I3C and DIM are potent activators of the aryl hydrocarbon receptor (AhR), a transcription factor that regulates genes involved in xenobiotic detoxification (Phase I and Phase II enzymes), cell cycle control, and immune function.
A key mechanism of I3C and DIM is the modulation of estrogen metabolism. They promote a metabolic shift favoring the production of 2-hydroxyestrone, a less estrogenic metabolite, over 16-alpha-hydroxyestrone, which has stronger estrogenic and proliferative activity. This alteration in the estrogen metabolite ratio is considered a primary mechanism behind the protective effects against estrogen-sensitive cancers, particularly breast and cervical cancers.
I3C also exerts direct anti-cancer effects by inducing G1 phase cell cycle arrest through inhibition of cyclin-dependent kinases (CDKs) and by promoting apoptosis in cancer cells via modulation of the PI3K/Akt survival signaling pathway. Additionally, I3C inhibits the TGF-beta/Smad signaling pathway, which plays roles in fibrosis and tumor progression. These multi-target mechanisms make I3C a promising chemopreventive compound with activity against multiple cancer-related pathways.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Bioavailability Issues:: Multiple users and sources note that I3C has poor bioavailability in supplement form, with some suggesting food sources (broccoli, Brussels sprouts) may be more effective. Individual Variation:: Effectiveness varies significantly between users, with some experiencing noticeable benefits for estrogen-related issues while others see no changes. Food vs Supplement Debate:: Users frequently discuss whether consuming 300-400g of broccoli daily provides adequate I3C compared to supplements, with no definitive answer. I3C vs DIM Comparison:: Ongoing debate about whether I3C (which converts to DIM in the stomach) or taking DIM directly is more effective, with slight preference toward I3C for better absorption
- Multiple users and sources note that I3C has poor bioavailability in supplement form, with some suggesting food sources (broccoli, Brussels sprouts) may be more effective
- Effectiveness varies significantly between users, with some experiencing noticeable benefits for estrogen-related issues while others see no changes
- Users frequently discuss whether consuming 300-400g of broccoli daily provides adequate I3C compared to supplements, with no definitive answer
- Ongoing debate about whether I3C (which converts to DIM in the stomach) or taking DIM directly is more effective, with slight preference toward I3C for better absorption
Safety Profile
Safety Profile: Indole-3-Carbinol (I3C)
Common Side Effects
- Gastrointestinal symptoms: nausea, bloating, flatulence, and mild abdominal discomfort, especially at doses >400 mg/day
- Mild skin rashes in sensitive individuals
- Dizziness or lightheadedness at initiation
- Changes in urine odor (related to cruciferous vegetable metabolites)
- Loose stools or mild diarrhea
Serious Adverse Effects
- Estrogen modulation: at high doses, I3C and its condensation product DIM may excessively shift estrogen metabolism toward 2-hydroxyestrone, potentially causing symptoms of estrogen deficiency (fatigue, joint pain, mood changes)
- Paradoxical tumor promotion: some in vitro studies suggest I3C (not DIM) may promote tumor growth under certain hormonal conditions; clinical significance unclear but concerning at very high doses
- Hepatotoxicity: rare reports of elevated liver enzymes at sustained doses >800 mg/day
- Thyroid function interference: I3C is a goitrogen; high-dose supplementation may impair thyroid hormone synthesis, particularly in iodine-deficient individuals
Contraindications
- Hormone-sensitive cancers under active treatment (breast, ovarian, endometrial, prostate) — use only under oncologist supervision
- Hypothyroidism or iodine deficiency without thyroid monitoring
- Known hypersensitivity to cruciferous vegetables (Brassicaceae family)
- Concurrent use of tamoxifen or aromatase inhibitors without medical guidance
Drug Interactions
- CYP1A2 induction: I3C strongly induces CYP1A2, increasing metabolism of theophylline, clozapine, caffeine, and some antidepressants
- Tamoxifen / aromatase inhibitors: complex interaction — may enhance or antagonize anti-estrogen therapy depending on dose and individual metabolism
- Oral contraceptives: CYP1A2 induction may reduce estrogen levels and contraceptive efficacy
- Thyroid medications (levothyroxine): goitrogenic effects may increase levothyroxine requirements
- Acetaminophen: CYP1A2 induction may shift acetaminophen metabolism toward hepatotoxic pathways (NAPQI production)
Population-Specific Considerations
- Pregnancy / lactation: AVOID — significant estrogen-modulating effects may affect fetal development; no safety data in pregnant or lactating women
- Children / adolescents: not recommended; effects on developing endocrine system are unstudied
- Women with estrogen-related conditions (endometriosis, fibroids, PCOS): effects are dose-dependent and unpredictable; medical supervision required
- Men: high-dose I3C may reduce estradiol excessively, potentially affecting bone density and cardiovascular markers
- Thyroid patients: monitor TSH and free T4 regularly; may need thyroid dose adjustments
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Users commonly reference dosages without specific amounts, suggesting they rely on product labels or cruciferous vegetable consumption
Molecular Structure
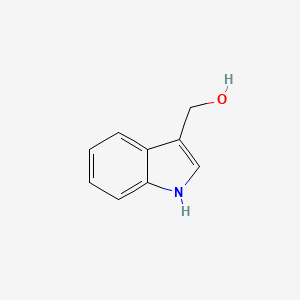
- Formula
- C9H9NO
- Weight
- 147.17 Da
- PubChem CID
- 3712
- Exact Mass
- 147.0684 Da
- LogP
- 1.1
- TPSA
- 36 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 1
- Rotatable Bonds
- 1
- Complexity
- 138
Identifiers (SMILES, InChI)
InChI=1S/C9H9NO/c11-6-7-5-10-9-4-2-1-3-8(7)9/h1-5,10-11H,6H2
IVYPNXXAYMYVSP-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Carcinogenic Concerns:: Several users express worry about potentially carcinogenic side effects from I3C supplementation, leading some to discontinue use
- DHT Blocking:: Reports that I3C may block DHT (dihydrotestosterone), which could negatively impact male characteristics and muscle building
- mTOR Inhibition:: Some users note concerns that I3C might block mTOR pathways, potentially interfering with muscle growth and recovery
- Minimal Direct Reports:: Despite concerns, few users report experiencing actual negative side effects from taking I3C, suggesting theoretical risks may outweigh practical ones
References (3)
- [1]Vegetable and fruit intake after diagnosis and risk of prostate cancer progression
→ Study examining how vegetable intake, particularly cruciferous vegetables containing I3C, may influence prostate cancer progression and overall cancer outcomes in diagnosed patients.
- [3]Implication of Trimethylamine N-Oxide (TMAO) in Disease: Potential Biomarker or New Therapeutic Target
→ Review examining TMAO's role in cardiovascular and neurological disorders, and how dietary interventions including cruciferous vegetable consumption may reduce TMAO levels and associated disease risks.
- [2]Effect of Choline Forms and Gut Microbiota Composition on Trimethylamine-N-Oxide Response in Healthy Men
→ Research on how different dietary compounds including those in cruciferous vegetables affect gut microbiota metabolism and TMAO production, which is linked to cardiovascular disease risk.
Hypericin
Hypericin is a naphthodianthrone compound found in St. John's Wort (Hypericum perforatum), primarily used for treating mild to moderate depression. It acts as a
Idebenone
Idebenone is a synthetic short-chain analogue of Coenzyme Q10 (CoQ10) that functions as a mitochondrial electron carrier and potent antioxidant. It transfers el