Galantamine
Galantamine is a naturally occurring alkaloid derived from snowdrop and daffodil bulbs, approved for the treatment of mild to moderate Alzheimer's disease. It acts as both a reversible acetylcholinesterase inhibitor and an allosteric modulator of nicotinic acetylcholine receptors, providing a dual mechanism for cognitive enhancement.
Overview
Galantamine (marketed as Razadyne/Reminyl) is a tertiary alkaloid originally isolated from the Caucasian snowdrop (Galanthus woronowii) and subsequently found in other Amaryllidaceae species including daffodils (Narcissus spp.) and red spider lilies (Lycoris radiata). It has a fascinating ethnobotanical history — its cognitive effects were first noted in Eastern European folk medicine, where snowdrop preparations were used to treat neurological conditions. Today it is produced both by extraction from cultivated daffodil bulbs and through chemical synthesis.
Galantamine's dual mechanism of action distinguishes it from other acetylcholinesterase (AChE) inhibitors used in Alzheimer's treatment. As a competitive and reversible AChE inhibitor, it increases acetylcholine availability in the synaptic cleft, compensating for the cholinergic deficit characteristic of Alzheimer's disease. Additionally, it acts as an allosteric potentiating ligand (APL) at nicotinic acetylcholine receptors, particularly the alpha-7 subtype, enhancing the receptor's response to acetylcholine. This nicotinic modulation may contribute to neuroprotective effects, improved attention and memory consolidation, and potentially disease-modifying activity beyond symptomatic relief.
Clinical trials have demonstrated that galantamine improves cognitive function, global assessment scores, and activities of daily living in patients with mild to moderate Alzheimer's disease, with benefits sustained over 12–36 months of treatment. It is also used off-label for vascular dementia and mixed dementia. Beyond clinical neurology, galantamine has gained niche popularity for lucid dreaming induction due to its enhancement of REM sleep cholinergic activity. Common side effects include nausea, vomiting, and diarrhea, which can be minimized with gradual dose titration. It should not be combined with other cholinesterase inhibitors or cholinergic drugs.
Mechanism of Action
Acetylcholinesterase Inhibition
Galantamine is a tertiary alkaloid originally isolated from Galanthus nivalis (snowdrop) that acts as a competitive, reversible inhibitor of acetylcholinesterase (AChE). It binds to the active site gorge of AChE with high selectivity over butyrylcholinesterase, slowing the hydrolysis of acetylcholine (ACh) in synaptic clefts. This increases ACh concentration and duration of action at both muscarinic and nicotinic receptors in the CNS, enhancing cholinergic neurotransmission (PMID: 12404554).
Allosteric Potentiation of Nicotinic Receptors
Uniquely among AChE inhibitors, galantamine functions as an allosteric potentiating ligand (APL) at nicotinic acetylcholine receptors (nAChRs), particularly the alpha7 and alpha4beta2 subtypes. It binds to a site distinct from the ACh binding site, inducing conformational changes that increase the probability of channel opening in response to ACh. This dual mechanism amplifies cholinergic signaling beyond what AChE inhibition alone achieves (PMID: 11157082).
Neuroprotective Downstream Effects
Alpha7 nAChR potentiation by galantamine activates the PI3K/Akt survival pathway and the JAK2/STAT3 signaling cascade in neurons and microglia. This reduces beta-amyloid-induced neurotoxicity, suppresses microglial release of pro-inflammatory cytokines (TNF-alpha, IL-1beta), and upregulates Bcl-2 anti-apoptotic protein expression. The alpha7 nAChR engagement also stimulates the cholinergic anti-inflammatory pathway via vagal afferents (PMID: 16271068).
Clinical Pharmacology
Galantamine crosses the blood-brain barrier readily and demonstrates dose-dependent improvements in cognitive function, attention, and behavioral symptoms in Alzheimer's disease. Its dual AChE-inhibitory and nicotinic-potentiating mechanism provides broader cholinergic enhancement than donepezil or rivastigmine, with particular benefits for attention and executive function mediated by prefrontal cortical nicotinic signaling (PMID: 15209681).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Alzheimer's Symptoms:: Highly effective for symptomatic relief of memory loss in confirmed Alzheimer's cases.. MCI Limitations:: Research suggests it is not effective for people with only mild cognitive impairment compared to a placebo.. Long-term Stability:: Clinical trials show that therapeutic responses can be maintained for at least 12 months with consistent dosing.
- Highly effective for symptomatic relief of memory loss in confirmed Alzheimer's cases.
- Research suggests it is not effective for people with only mild cognitive impairment compared to a placebo.
- Clinical trials show that therapeutic responses can be maintained for at least 12 months with consistent dosing.
Safety Profile
Common Side Effects
- Nausea and vomiting are the most commonly reported side effects, occurring in up to 20–30% of patients and often dose-limiting
- Diarrhea, abdominal pain, and decreased appetite
- Dizziness, headache, and fatigue
- Weight loss, which may be significant in some patients
- Insomnia and vivid or disturbing dreams
- Bradycardia (slowed heart rate), typically mild
- Muscle cramps and weakness
Serious Adverse Effects
- Severe bradycardia and heart block, particularly in patients with pre-existing cardiac conduction abnormalities or when combined with other rate-lowering agents
- Syncope (fainting) has been reported, especially during dose titration
- Gastrointestinal bleeding, particularly in patients with peptic ulcer disease or those on concurrent NSAIDs
- Seizures have been reported, though cholinesterase inhibitors may lower seizure threshold
- Severe skin reactions including Stevens-Johnson syndrome have been reported rarely
- Hepatotoxicity with elevated transaminases; liver function monitoring is recommended
- Worsening of parkinsonian symptoms in some patients
- Respiratory depression in patients with severe asthma or COPD
Contraindications
- Known hypersensitivity to galantamine or any excipients
- Severe hepatic impairment (Child-Pugh score 10–15) contraindicates use
- Severe renal impairment (CrCl < 9 mL/min) contraindicates use
- Active gastrointestinal bleeding or untreated peptic ulcer disease
- Urinary tract obstruction or bladder outflow obstruction
- Not approved for use in children
Drug Interactions
- Concurrent use with other cholinesterase inhibitors (donepezil, rivastigmine) is contraindicated due to additive cholinergic toxicity
- Beta-blockers, calcium channel blockers (verapamil, diltiazem), digoxin, and amiodarone may cause severe bradycardia or heart block when combined with galantamine
- Anticholinergic medications (oxybutynin, diphenhydramine, tricyclic antidepressants) directly oppose galantamine's mechanism and reduce efficacy
- Succinylcholine and similar neuromuscular blocking agents may have prolonged effects
- Strong CYP2D6 and CYP3A4 inhibitors (paroxetine, ketoconazole, erythromycin) significantly increase galantamine levels, requiring dose reduction
- NSAIDs increase gastrointestinal bleeding risk
FDA-Specific Information
- FDA-approved for mild to moderate Alzheimer disease; dosing should follow prescribing information (starting 8 mg/day, titrating to 16–24 mg/day)
- Extended-release formulation available for once-daily dosing
- Black box–equivalent warnings regarding cardiac conduction and GI bleeding
- Requires medical supervision; this is a prescription medication and should not be self-administered as a supplement
Pharmacokinetic Profile
Galantamine — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- 16 mg to 24 mg daily is the most common range for treating cognitive decline.
Molecular Structure
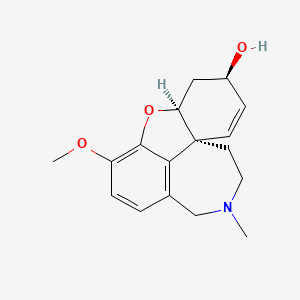
- Formula
- C17H21NO3
- Weight
- 287.35 Da
- PubChem CID
- 9651
- Exact Mass
- 287.1521 Da
- LogP
- 1.8
- TPSA
- 41.9 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 1
- Complexity
- 440
Identifiers (SMILES, InChI)
InChI=1S/C17H21NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,12,14,19H,7-10H2,1-2H3/t12-,14-,17-/m0/s1
ASUTZQLVASHGKV-JDFRZJQESA-NSafety Profile
Common Side Effects
- Gastrointestinal Distress:: Nausea and vomiting are the most frequently reported adverse effects, especially during the initial phase.
- Diarrhea:: Frequently cited in clinical trials as a common reaction to acetylcholinesterase inhibition.
- Sleep Disturbances:: Can cause vivid dreams or insomnia if taken too close to the initial onset of sleep.
References (2)
- [2]Galantamine in AD
→ A 12-month study demonstrated that galantamine significantly improved cognition and global function in mild to moderate Alzheimer's patients, with benefits maintained over a full year of treatment.
- [1]Galantamine for dementia due to Alzheimer's disease and mild cognitive impairment
→ This systematic review found that galantamine significantly improves cognitive function and daily living activities in Alzheimer's patients at doses of 16mg to 24mg daily, though it showed little benefit for mild cognitive impairment.
Galanin
Galanin is a neuromodulatory peptide first isolated from porcine intestinal extracts by Tatemoto et al. in 1983, named for its N-terminal glycine and C-terminal
Gamma-Tocopherol
Gamma-tocopherol is one of the eight naturally occurring forms of vitamin E, found abundantly in common dietary oils like soybean and corn oil. Unlike alpha-toc