Phenylpiracetam
A phenylated derivative of piracetam with significantly enhanced potency and additional psychostimulant properties, widely used as a nootropic for cognitive enhancement, physical performance, and cold tolerance.
Overview
Phenylpiracetam (also known as Phenotropil or Carphedon) is a racetam-class nootropic created by adding a phenyl group to the parent compound piracetam. This structural modification dramatically increases its lipophilicity and blood-brain barrier penetration, resulting in a compound estimated to be 20-60 times more potent than piracetam on a per-milligram basis. Developed in the early 1980s at the Russian Academy of Sciences, phenylpiracetam was originally designed to help cosmonauts cope with the stresses of space travel, including cognitive demands and extreme cold — a legacy that contributes to its reputation as a performance-enhancing agent for physically and mentally demanding environments.
Phenylpiracetam modulates multiple neurotransmitter systems. Like other racetams, it positively modulates AMPA and NMDA glutamate receptors, enhancing excitatory neurotransmission and long-term potentiation — processes central to learning and memory. Beyond its racetam heritage, the phenyl moiety confers additional dopaminergic and noradrenergic activity, producing mild psychostimulant effects including increased alertness, motivation, and physical stamina. This dual mechanism — cognitive enhancement plus stimulation — distinguishes it from milder racetams like piracetam or aniracetam. Phenylpiracetam also demonstrates anticonvulsant and anxiolytic properties in animal models, and it increases cold tolerance, likely through modulation of peripheral and central catecholamine pathways.
Due to its stimulant-like performance benefits, phenylpiracetam was banned by the World Anti-Doping Agency (WADA) and has appeared in doping violations in Olympic athletes. Typical nootropic dosing ranges from 100-300 mg per day, often divided into two doses. Tolerance to the stimulant effects can develop relatively quickly with daily use, so many users cycle it or reserve it for demanding tasks. It stacks well with choline sources like alpha-GPC or citicoline. Phenylpiracetam remains available as a prescription drug (Phenotropil) in Russia, though it is sold as an unregulated supplement in many other countries. Related racetams include pramiracetam, coluracetam, and oxiracetam.
Mechanism of Action
Mechanism of Action
Phenylpiracetam (also marketed as Phenotropil or fonturacetam) is piracetam with a phenyl group at the 4-position of the pyrrolidone ring. This single structural modification dramatically alters the pharmacological profile, adding potent psychostimulant activity to the base nootropic effects of the racetam scaffold.
Enhanced Racetam Core
The pyrrolidone core retains piracetam's AMPA receptor modulating activity. Phenylpiracetam slows AMPA receptor desensitization, prolonging excitatory postsynaptic potentials and facilitating LTP induction. This mechanism enhances learning, memory encoding, and recall. The phenyl group appears to increase binding affinity at the AMPA modulatory site compared to piracetam, contributing to phenylpiracetam's roughly 30-60 fold greater potency.
Catecholaminergic Activity
The most significant pharmacological difference from piracetam is phenylpiracetam's inhibition of dopamine and norepinephrine reuptake transporters. DAT inhibition increases dopamine signaling in reward and executive function circuits, while NET inhibition enhances noradrenergic tone for arousal and attention. These properties led WADA to ban phenylpiracetam as a stimulant, and they explain clinical reports of increased physical stamina, motivation, and cold tolerance.
Stereoselective Effects
Phenylpiracetam exists as R and S enantiomers with distinct pharmacological profiles. The R-enantiomer shows greater nootropic and antiamnestic activity, while the S-enantiomer has stronger anxiolytic and anticonvulsant effects. Commercially available phenylpiracetam is typically racemic, providing both profiles simultaneously.
Neuroprotective Properties
Phenylpiracetam protects against hypoxic, ischemic, and toxic brain injury in animal models. Mechanisms include reduced excitotoxicity (via GABA enhancement), improved cerebral blood flow, increased neuronal membrane fluidity, and enhanced antioxidant defenses. These properties have supported its clinical use in post-stroke rehabilitation in Russia.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Highly Potent:: Active at 100-200mg vs 2-4g for piracetam.. Fast Tolerance:: Loses effectiveness quickly with daily use.. Best Used Sparingly:: Reserved for important occasions.
- Active at 100-200mg vs 2-4g for piracetam.
- Loses effectiveness quickly with daily use.
- Reserved for important occasions.
Safety Profile
Safety Profile: Phenylpiracetam
Common Side Effects
- Insomnia and sleep disturbances (most common; strongly dose-dependent)
- Headache, particularly during initial use
- Increased blood pressure and heart rate
- Psychomotor agitation, restlessness, and irritability
- Nausea and gastrointestinal discomfort
- Skin warmth and flushing
- Rapid tolerance development with daily use
Serious Adverse Effects
- Hypertensive episodes in predisposed individuals
- Severe insomnia with prolonged use or high doses
- Allergic reactions including rash and angioedema (rare)
- Potential for psychological dependence due to stimulant properties
- Exacerbation of anxiety disorders and panic attacks
- Psychotic symptoms reported at supratherapeutic doses (rare)
Contraindications
- Known hypersensitivity to phenylpiracetam or racetam compounds
- Uncontrolled hypertension or severe cardiovascular disease
- Severe anxiety or panic disorder
- History of psychosis or bipolar disorder (manic phase)
- Severe hepatic or renal impairment
- Pregnancy and breastfeeding (no safety data)
- Children under 18 years
Drug Interactions
- CNS stimulants (caffeine, amphetamines, modafinil): Additive stimulant effects; avoid combination or use reduced doses
- Antihypertensives: May reduce efficacy of blood pressure medications
- Anticoagulants (warfarin): Reports of altered coagulation; monitor INR
- Antidepressants (SSRIs, SNRIs): Potential additive activating effects
- Other nootropics/racetams: Additive cognitive effects; dose adjustment may be needed
- Antiepileptic drugs: May affect seizure threshold
Population-Specific Considerations
- Cognitive enhancement: Typical dose 100-200 mg once or twice daily; take before noon to prevent insomnia; cycle 2-3 weeks on, 1-2 weeks off to prevent tolerance
- Athletes: Banned by WADA; classified as a stimulant; will trigger positive doping test
- Tolerance management: Rapid tolerance is well-documented; daily use quickly diminishes effects; intermittent dosing strongly recommended
- Note: Phenylpiracetam is the same compound as phenotropil/fonturacetam. Not approved by FDA or EMA. Originally developed as a Russian pharmaceutical for cosmonaut use.
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 100-300mg per dose.
Molecular Structure
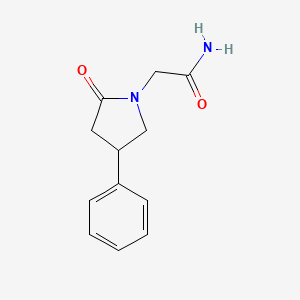
- Formula
- C12H14N2O2
- Weight
- 218.25 Da
- PubChem CID
- 132441
- Exact Mass
- 218.1055 Da
- LogP
- 0.1
- TPSA
- 63.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 3
- Complexity
- 285
Identifiers (SMILES, InChI)
InChI=1S/C12H14N2O2/c13-11(15)8-14-7-10(6-12(14)16)9-4-2-1-3-5-9/h1-5,10H,6-8H2,(H2,13,15)
LYONXVJRBWWGQO-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Insomnia:: Strong stimulation can disrupt sleep.
- Tolerance:: Rapid tolerance development.
- Overstimulation:: Can cause anxiety in sensitive individuals.
- Headaches:: Possible without adequate choline.
References (6)
- [2]New synthesis of β-aryl-GABA drugs
→ Researchers developed a novel cascade radical reaction method for synthesizing phenylpiracetam and related compounds.
- [4]Stereochemistry of phenylpiracetam and its methyl derivative: improvement of the pharmacological profile
→ Review emphasizing how molecular configuration of (R)-phenylpiracetam affects biological effectiveness for cognitive enhancement.
- [5]Single Enantiomer's Urge to Crystallize in Centrosymmetric Space Groups: Solid Solutions of Phenylpiracetam
→ Analysis revealed phenylpiracetam enantiomers form distinct polymorphs with different crystallographic properties.
- [6]On the formation of phenylpiracetam solid solutions: thermodynamic and structural considerations
→ Study characterized two rare solid solution types with distinct miscibility properties and phase transitions.
- [1]S-phenylpiracetam, a selective DAT inhibitor, reduces body weight gain without influencing locomotor activity
→ S-phenylpiracetam significantly decreased body weight gain and fat mass in obese rodent models.
- [3]Neuroprotective and anti-inflammatory activity of DAT inhibitor R-phenylpiracetam
→ R-phenylpiracetam demonstrated neuroprotective and anti-inflammatory effects through dopamine transporter interaction.
Phenylalanine
Phenylalanine is an essential amino acid that serves as a precursor to tyrosine and subsequently dopamine, norepinephrine, and epinephrine. It exists in three f
Phloretin
Phloretin is a natural dihydrochalcone flavonoid found primarily in apples and apple tree bark that exhibits anti-inflammatory, antioxidant, and neuroprotective