Nefiracetam
A fat-soluble pyrrolidone nootropic of the racetam family that enhances long-term potentiation through simultaneous modulation of GABAergic, cholinergic, and glutamatergic neurotransmission, investigated for cognitive impairment, epilepsy, and post-stroke recovery.
Overview
Nefiracetam (N-(2,6-dimethylphenyl)-2-(2-oxopyrrolidin-1-yl)acetamide, DM-9384) is a lipophilic nootropic compound of the racetam family, structurally derived from piracetam with the addition of a 2,6-dimethylphenyl group that dramatically increases lipid solubility and blood-brain barrier penetration. Developed by Daiichi Pharmaceutical (now Daiichi Sankyo) in Japan, nefiracetam is distinguished from other racetams by its unique multi-target mechanism of action that simultaneously modulates three major neurotransmitter systems involved in memory and learning. Unlike piracetam or aniracetam, which primarily affect glutamatergic or cholinergic systems, nefiracetam acts as an integrated modulator of the GABAergic-cholinergic-glutamatergic triad that underlies long-term potentiation (LTP) — the cellular mechanism of memory formation.
The molecular pharmacology of nefiracetam involves several distinct receptor interactions. It potentiates GABA-A receptor currents through a novel mechanism independent of the benzodiazepine binding site, acting instead on the receptor's signal transduction via protein kinase C (PKC) and CaM kinase II pathways. Simultaneously, it enhances nicotinic acetylcholine receptor (nAChR) activity by directly interacting with the alpha4-beta2 subtype, increasing acetylcholine release and cholinergic transmission. At glutamatergic synapses, nefiracetam potentiates NMDA receptor function and enhances calcium influx through L-type voltage-dependent calcium channels — both critical for LTP induction and maintenance. This triple-modulator profile converges on sustained activation of PKC and CaM kinase II, which phosphorylate CREB and other transcription factors to drive the gene expression programs required for long-term memory consolidation.
Clinical investigations of nefiracetam have focused on post-stroke cognitive impairment, Alzheimer's disease, and epilepsy. Phase II trials in post-stroke patients demonstrated improvements in apathy, depression, and cognitive function at doses of 600–900 mg daily, though Phase III results were less consistent. In epilepsy research, nefiracetam showed anticonvulsant properties in kindling models without sedation — attributed to its GABAergic modulation. Typical research doses range from 150–900 mg daily in divided doses. Nefiracetam is generally well tolerated, with headache, nausea, and gastrointestinal discomfort as the most commonly reported side effects. A notable safety concern identified in preclinical studies was dose-dependent testicular toxicity in dogs (but not rats or monkeys), which contributed to the compound not receiving regulatory approval. It is often discussed alongside other advanced racetams such as aniracetam, oxiracetam, and coluracetam in nootropic research contexts.
Mechanism of Action
Nefiracetam is a pyrrolidone-derived nootropic that modulates neurotransmission through three primary molecular targets. First, it potentiates nicotinic acetylcholine receptor (nAChR) activity, particularly alpha4-beta2 subtypes, by interacting with the protein kinase C (PKC) phosphorylation site on the receptor, prolonging channel open time and enhancing cholinergic transmission in hippocampal circuits critical for memory formation. Second, nefiracetam activates L-type voltage-dependent calcium channels (L-VDCCs) through a direct interaction with the channel protein, increasing calcium influx that triggers CaMKII activation and long-term potentiation (LTP) in CA1 hippocampal neurons.
Third, nefiracetam enhances GABAergic neurotransmission by potentiating GABA-A receptor currents through a PKC-dependent mechanism, while simultaneously modulating the balance between excitatory and inhibitory signaling. It also facilitates glutamatergic transmission by enhancing AMPA receptor-mediated currents and promoting NMDA receptor function through glycine-site modulation. Nefiracetam activates CaMKII downstream signaling, which phosphorylates CREB (cAMP response element-binding protein), promoting transcription of brain-derived neurotrophic factor (BDNF) and other plasticity-related genes essential for long-term memory consolidation.
The convergence of cholinergic, GABAergic, and glutamatergic modulation gives nefiracetam a broad pro-cognitive profile. Therapeutically, it has been investigated for Alzheimer's disease, post-stroke cognitive impairment, and age-related memory decline. Its ability to enhance LTP through multiple complementary pathways rather than a single receptor target provides robust cognitive enhancement with reduced risk of excitotoxicity. Nefiracetam also demonstrates anticonvulsant properties through its GABAergic activity and neuroprotective effects via BDNF upregulation and calcium homeostasis maintenance.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Moderate Potency:: Users generally find nefiracetam less immediately noticeable than phenylpiracetam or noopept, with effects building over time rather than producing acute changes. Age-Related Benefits:: Research strongly supports effectiveness in older populations and those with existing cognitive impairments, with benefits persisting weeks after discontinuation. Stroke Recovery:: Multiple studies demonstrate significant improvements in spatial learning and memory function following cerebral ischemia at doses of 10-30 mg/kg. Combination Potential:: Shows synergistic effects when combined with cholinergics like physostigmine, suggesting value in stacking for enhanced cognitive benefits
- Users generally find nefiracetam less immediately noticeable than phenylpiracetam or noopept, with effects building over time rather than producing acute changes
- Research strongly supports effectiveness in older populations and those with existing cognitive impairments, with benefits persisting weeks after discontinuation
- Multiple studies demonstrate significant improvements in spatial learning and memory function following cerebral ischemia at doses of 10-30 mg/kg
- Shows synergistic effects when combined with cholinergics like physostigmine, suggesting value in stacking for enhanced cognitive benefits
Safety Profile
Side effects are typically mild and can include headaches, nervousness, and nausea, often linked to higher doses. It is contraindicated in individuals with severe kidney or liver impairment, as this can affect its metabolism and clearance. Animal studies have indicated a potential for testicular toxicity at very high doses, though this has not been replicated in human studies.
Pharmacokinetic Profile
Molecular Structure
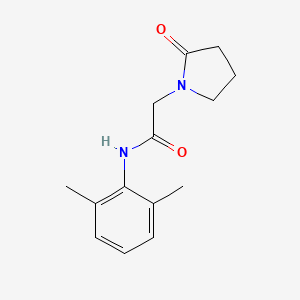
- Formula
- C14H18N2O2
- Weight
- 246.30 Da
- PubChem CID
- 71157
- Exact Mass
- 246.1368 Da
- LogP
- 1.4
- TPSA
- 49.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 3
- Complexity
- 320
Identifiers (SMILES, InChI)
InChI=1S/C14H18N2O2/c1-10-5-3-6-11(2)14(10)15-12(17)9-16-8-4-7-13(16)18/h3,5-6H,4,7-9H2,1-2H3,(H,15,17)
NGHTXZCKLWZPGK-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Kidney Concerns:: Major safety concern among users regarding potential nephrotoxicity, with at least one user reporting kidney pain after 100 mg oral doses
- Metabolic Issues:: Dog studies showed testicular toxicity from oral metabolites, leading users to avoid or minimize oral administration despite lack of human data
- Generally Mild:: When side effects occur, they are typically subtle and less pronounced than with stimulant nootropics or other racetams
- Individual Variation:: Some users report no side effects at moderate doses, while others experience discomfort, highlighting significant individual response differences
References (9)
- [2]Cellular mechanism of action of cognitive enhancers: effects of nefiracetam on neuronal Ca2+ channels
→ Nefiracetam produced a twofold increase in L-type calcium channel currents and facilitated recovery from opioid-induced inhibition of N-type calcium channels through interaction with inhibitory G proteins. This mechanism appears to underlie its cognitive-enhancing effects.
- [6]Effects of Nefiracetam on the amygdala-kindled seizures in rats
→ Nefiracetam decreased afterdischarge duration, seizure stage, and motor seizure duration in fully kindled rats in a dose-dependent manner at 25-100 mg/kg. However, it had minimal effects on the development of kindling.
- [8]Nefiracetam and physostigmine: separate and combined effects on learning in older rabbits
→ Combining nefiracetam (10 mg/kg) with low doses of physostigmine improved learning rates and magnitude beyond either drug alone in aged rabbits, suggesting potential for combination therapy in cognitive enhancement.
- [1]Effects of nefiracetam on amnesia animal models with neuronal dysfunctions
→ Nefiracetam improved learning and memory impairments across multiple amnesia models including scopolamine, bicuculline, and ethanol-induced deficits. It also improved cholinergic and monoaminergic neurotransmitter alterations in the hippocampus of brain-lesioned animals.
- [3]Quantitative effects of nefiracetam on spatial learning of rats after cerebral embolism
→ Nefiracetam (30 mg/kg for 21 days) completely ameliorated spatial learning impairments in embolized rats, with effects persisting after treatment cessation. The drug significantly reduced escape latency compared to vehicle controls.
- [7]Effects of nefiracetam on spatial memory function and acetylcholine and GABA metabolism in microsphere-embolized rats
→ Nefiracetam (10 mg/kg daily) effectively improved spatial learning dysfunction in stroke-model rats and was found to modify cholinergic and GABAergic systems in ischemic brain tissue.
- [9]Possible involvement of the activation of voltage-sensitive calcium channels in the ameliorating effects of nefiracetam
→ The anti-amnesic effects of nefiracetam on scopolamine-induced memory impairment were blocked by L-type and N-type calcium channel antagonists, confirming that activation of high-voltage-activated calcium channels is critical to its mechanism of action.
- [4]The long-term effects of nefiracetam on learning in older rabbits
→ Older rabbits treated with 10 mg/kg nefiracetam showed significant improvements in relearning ability up to 5 weeks after drug administration ended. Effects were observed even without continuous dosing.
- [5]Anticonvulsant and neuroprotective effects of the novel nootropic agent nefiracetam on kainic acid-induced seizures in rats
→ Nefiracetam dose-dependently protected against hippocampal damage from kainic acid-induced seizures without affecting behavioral seizures. At 25-100 mg/kg, it prevented degeneration and loss of pyramidal cells in hippocampal regions.
Nattokinase
Nattokinase is a fibrinolytic enzyme extracted from natto, a traditional Japanese fermented soybean food produced by Bacillus subtilis. It works by breaking dow
Neuropeptide Y (NPY)
**Neuropeptide Y (NPY)** is a 36-amino acid peptide amide and the most abundant neuropeptide in the mammalian central nervous system. First isolated from porcin