Citicoline
Citicoline (CDP-choline) is an endogenous nucleotide that serves as an intermediate in the synthesis of phosphatidylcholine, a major component of cell membranes. It is widely used as a nootropic supplement to support cognitive function, memory, and neuroprotection.
Overview
Citicoline, also known as cytidine diphosphate-choline (CDP-choline), is a naturally occurring compound found in every cell of the body. It plays a critical role in the Kennedy pathway, the primary biosynthetic route for phosphatidylcholine, which is essential for maintaining the structural integrity of neuronal cell membranes. When taken as a supplement, citicoline is hydrolyzed into cytidine and choline, both of which cross the blood-brain barrier and are reassembled into CDP-choline in the brain.
Citicoline has been extensively studied for its cognitive-enhancing and neuroprotective properties. Clinical research has demonstrated benefits for attention, memory, and executive function in both healthy adults and individuals with age-related cognitive decline. It increases levels of key neurotransmitters including acetylcholine and dopamine, while also supporting mitochondrial function and reducing oxidative damage. In some countries, citicoline is prescribed as a pharmaceutical agent for stroke recovery and neurodegenerative conditions.
The compound is well-tolerated at typical doses of 250–500 mg per day, with studies using up to 2,000 mg daily showing a favorable safety profile. Citicoline is available in two common forms: as a free-form supplement and as the branded ingredient Cognizin. It is frequently combined with other nootropics such as racetams or uridine to create synergistic stacks targeting cholinergic and phospholipid pathways.
Mechanism of Action
Endogenous Nucleotide — Kennedy Pathway Intermediate
Citicoline (cytidine 5'-diphosphocholine, CDP-choline) is an endogenous intermediate in the Kennedy pathway of phosphatidylcholine (PC) biosynthesis. Following oral administration, citicoline is hydrolyzed to cytidine and choline in the intestinal mucosa and liver, absorbed separately into the systemic circulation, and resynthesized intracellularly by CTP:phosphocholine cytidylyltransferase (CCT) — the rate-limiting enzyme in PC synthesis. Oral bioavailability exceeds 90%, with peak plasma choline levels at 3-5 hours and cytidine/uridine levels at 4-6 hours post-dose (PMID: 15591001).
Phospholipid Membrane Restoration
Citicoline's primary neuroprotective mechanism involves restoration of phospholipid membrane integrity in ischemic and damaged neurons. During stroke and neurodegeneration, phospholipase A2 (PLA2) activation releases arachidonic acid and lysophosphatidylcholine, compromising membrane bilayer structure. Citicoline provides both the polar head group (choline) and energetic substrate (CTP from cytidine) to reacylate lysophospholipids and synthesize new PC, restoring membrane structure and function. It also enhances synthesis of phosphatidylethanolamine, phosphatidylserine, and sphingomyelin, collectively rebuilding all major membrane phospholipid classes (PMID: 16420464).
Cholinergic & Dopaminergic Enhancement
Citicoline increases acetylcholine synthesis by providing choline substrate to choline acetyltransferase (ChAT) in cholinergic neurons, with MRS studies confirming increased brain choline levels after oral dosing. It also enhances dopamine release in the striatum by increasing tyrosine hydroxylase activity and D2 receptor density. The cytidine component crosses the blood-brain barrier and is converted to uridine, which is phosphorylated to UTP — a P2Y receptor agonist that stimulates neurite outgrowth, synaptogenesis, and dendritic spine formation in cortical neurons (PMID: 18816480).
Mitochondrial & Anti-Apoptotic Effects
Citicoline stabilizes mitochondrial cardiolipin content, maintaining electron transport chain complex assembly and preventing cytochrome c release under ischemic conditions. It also inhibits caspase-3 activation and reduces PARP cleavage, providing anti-apoptotic protection in stroke penumbra neurons (PMID: 22465051).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Individual Response:: Effectiveness varies dramatically between users—some describe it as life-changing while others report zero noticeable effects, suggesting genetic or biochemical differences in response. Synergistic Effects:: Works better when combined with other supplements like Huperzine A (for acetylcholine metabolism), racetams, or comprehensive nootropic stacks rather than as a standalone supplement. Duration for Results:: Benefits may take weeks to manifest rather than providing immediate effects, with some users noting subtle improvements that become more apparent over extended use. Comparison to Other Forms:: Users who tried both citicoline and Alpha-GPC report mixed preferences, with citicoline being less likely to cause depression compared to Alpha-GPC for some individuals
- Effectiveness varies dramatically between users—some describe it as life-changing while others report zero noticeable effects, suggesting genetic or biochemical differences in response
- Works better when combined with other supplements like Huperzine A (for acetylcholine metabolism), racetams, or comprehensive nootropic stacks rather than as a standalone supplement
- Benefits may take weeks to manifest rather than providing immediate effects, with some users noting subtle improvements that become more apparent over extended use
- Users who tried both citicoline and Alpha-GPC report mixed preferences, with citicoline being less likely to cause depression compared to Alpha-GPC for some individuals
Safety Profile
Safety Profile: Citicoline (CDP-Choline)
Common Side Effects
- Headache (most commonly reported, ~3–5% in clinical trials)
- Insomnia and restlessness, particularly with afternoon or evening dosing
- Gastrointestinal symptoms: nausea, diarrhea, and abdominal discomfort (dose-related, more common above 1000 mg/day)
- Mild dizziness and transient blood pressure fluctuation
- Rare skin rash or flushing
Serious Adverse Effects
- Cholinergic overstimulation: at high doses (>2000 mg/day) or when stacked with other cholinergics, symptoms may include excessive salivation, lacrimation, bradycardia, and bronchoconstriction
- Mood destabilization: case reports of mania/hypomania in individuals with bipolar disorder, likely due to dopaminergic and cholinergic modulation
- Hypotension: vagal-mediated blood pressure drops, particularly in elderly patients or those on antihypertensives
- Long-term safety data is reassuring—clinical trials up to 12 months at 2000 mg/day show no significant organ toxicity; the ICTUS stroke trial (n=2298) confirmed a favorable safety profile
Contraindications
- Hypersensitivity to citicoline or cytidine/choline components
- Caution in bipolar disorder or psychotic disorders (dopaminergic potentiation)
- Caution in Parkinson's disease patients on levodopa (may alter dopamine metabolism)
- Severe bradycardia or sick sinus syndrome (vagotonic risk)
Drug Interactions
- Levodopa/carbidopa: citicoline may enhance dopaminergic effects; can improve efficacy but also increase dyskinesias—requires dose monitoring
- Cholinesterase inhibitors (donepezil, rivastigmine): additive cholinergic effects; risk of cholinergic crisis at combined high doses
- Anticholinergic medications: citicoline may reduce effectiveness of drugs like oxybutynin, diphenhydramine, or benztropine
- Selegiline / MAO-B inhibitors: theoretical additive dopaminergic stimulation
- Racetams (piracetam, aniracetam): commonly stacked in nootropic protocols; synergistic but increases cholinergic burden—monitor for headache and GI symptoms
Population-Specific Considerations
- Stroke recovery: most robust evidence base; 500–2000 mg/day is well-tolerated in post-stroke populations, though the ICTUS trial showed no significant efficacy benefit over placebo
- Elderly / cognitive decline: well-tolerated in geriatric populations; 500–1000 mg/day studied in MCI and Alzheimer's with favorable safety
- Pregnancy / lactation: choline is essential during pregnancy, but citicoline specifically has not been studied; prefer dietary choline or standard choline supplements
- Children: limited data; some studies in pediatric head trauma at 100–200 mg/day showed acceptable tolerability
- Glaucoma patients: some evidence of neuroprotective benefit at 500–1000 mg/day; no safety concerns identified
- Substance use disorders: studied for cocaine and methamphetamine dependence; 2000 mg/day tolerated but not standard of care
Pharmacokinetic Profile
Citicoline — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Most commonly used at 250-500mg daily, with clinical studies using 500mg/day showing positive results for memory and cognitive function
Molecular Structure
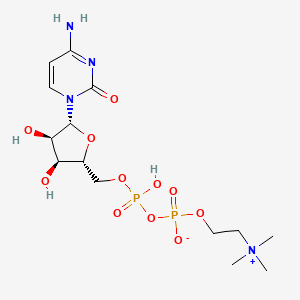
- Formula
- C14H26N4O11P2
- Weight
- 488.32 Da
- PubChem CID
- 13804
- Exact Mass
- 488.1073 Da
- LogP
- -4
- TPSA
- 214 Ų
- H-Bond Donors
- 4
- H-Bond Acceptors
- 11
- Rotatable Bonds
- 10
- Complexity
- 821
Identifiers (SMILES, InChI)
InChI=1S/C14H26N4O11P2/c1-18(2,3)6-7-26-30(22,23)29-31(24,25)27-8-9-11(19)12(20)13(28-9)17-5-4-10(15)16-14(17)21/h4-5,9,11-13,19-20H,6-8H2,1-3H3,(H3-,15,16,21,22,23,24,25)/t9-,11-,12-,13-/m1/s1
RZZPDXZPRHQOCG-OJAKKHQRSA-NSafety Profile
Common Side Effects
- Minimal Side Effects:: Generally well-tolerated with few reported adverse effects in both clinical trials and user experiences, particularly at standard doses
- Sleep Disruption:: Some users report difficulty sleeping if taken too late in the day due to its stimulating cognitive effects
- Individual Sensitivities:: A minority of users found it worsened their symptoms or caused headaches, though this appears uncommon compared to other nootropics
- Depression Risk (Alpha-GPC):: Users specifically choosing citicoline over Alpha-GPC cite that the latter caused depression, suggesting citicoline may be better tolerated for mood-sensitive individuals
References (8)
- [1]Citicoline and Memory Function in Healthy Older Adults: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial
→ In 100 healthy adults aged 50-85 with age-associated memory impairment, 500mg/day of citicoline for 12 weeks significantly improved memory function compared to placebo, with no serious adverse events reported.
- [2]Application of Citicoline in Neurological Disorders: A Systematic Review
→ Systematic review of 47 studies found citicoline prevents dementia progression, enhances cognitive functions in healthy individuals, improves stroke prognosis, and stimulates nerve regeneration in animal models with unclear effects in brain trauma patients.
- [3]Is Citicoline Effective in Preventing and Slowing Down Dementia? A Systematic Review and Meta-Analysis
→ Meta-analysis demonstrated citicoline supplementation shows promise in preventing and slowing cognitive decline in dementia patients, though researchers call for more standardized trials to establish optimal dosing protocols.
- [4]Efficacy of citicoline as a supplement in glaucoma patients: A Systematic review
→ First systematic review investigating citicoline's therapeutic potential in glaucoma found evidence of neuroprotective properties on retinal ganglion cells, offering a potential treatment strategy beyond intraocular pressure control.
- [5]Citicoline: A Superior Form of Choline?
→ Analysis reveals citicoline is significantly less toxic than choline on a molar mass basis and serves as an effective dietary source of both choline and cytidine, with authorized health claims for normal lipid metabolism and liver function.
- [6]Role of Citicoline in Patients With Mild Cognitive Impairment
→ Review of available literature shows citicoline consistently improves cognitive function in patients with mild cognitive impairment, especially of vascular origin, through diverse neuroprotective mechanisms of action.
- [7]Citicoline in addictive disorders: a review of the literature
→ Nine clinical studies on citicoline for addiction (primarily cocaine use) found it safe, well-tolerated, and potentially beneficial for reducing craving, withdrawal symptoms, and cognitive dysfunction while attenuating neurotoxic effects of drugs of abuse.
- [8]Therapeutic applications of citicoline for stroke and cognitive dysfunction in the elderly: a review of the literature
→ Review highlights citicoline's promise for elderly patients with cognitive deficits and early-stage Alzheimer's disease through cholinergic and neuroprotective actions, though stroke trial results remain inconclusive.
Cistanche (Cistanche deserticola)
Cistanche, specifically Cistanche deserticola and Cistanche tubulosa, is a parasitic desert plant rich in phenylethanoid glycosides like echinacoside and verbas
Citrulline (L-Citrulline)
L-Citrulline is a nonessential amino acid that serves as a potent precursor to L-arginine, effectively increasing nitric oxide synthesis in the body. It is prim