Ferulic Acid
Ferulic acid is a naturally occurring hydroxycinnamic acid and potent antioxidant found abundantly in plant cell walls, grains, and seeds. It is widely used in skincare formulations for its photoprotective and anti-aging properties, and is being researched for cardiovascular, neuroprotective, and anti-inflammatory benefits.
Overview
Ferulic acid (4-hydroxy-3-methoxycinnamic acid) is a phenolic compound ubiquitous in the plant kingdom, found in high concentrations in rice bran, wheat bran, oats, coffee, and various fruits and vegetables. It plays a structural role in plant cell walls by cross-linking polysaccharides and lignin. As a dietary antioxidant, ferulic acid is one of the most abundant phenolic acids consumed in the human diet and demonstrates excellent free radical scavenging activity, particularly against reactive oxygen and nitrogen species.
In dermatology, ferulic acid has gained prominence as a key ingredient in topical antioxidant formulations. Its ability to stabilize and enhance the photoprotective effects of vitamins C and E has been well documented, with studies showing that the combination of 15% vitamin C, 1% vitamin E, and 0.5% ferulic acid provides approximately 8-fold protection against solar-simulated UV damage. This synergy has made ferulic acid a staple in premium anti-aging serums, where it helps reduce photodamage, hyperpigmentation, and fine lines.
Beyond skincare, preclinical research has revealed promising systemic benefits of ferulic acid. It has demonstrated cardioprotective effects through reduction of oxidative stress and improvement of endothelial function, neuroprotective properties via modulation of neuroinflammation and amyloid-beta toxicity, and antidiabetic activity through enhancement of insulin sensitivity and pancreatic beta-cell protection. Its excellent safety profile, good oral bioavailability relative to other polyphenols, and broad biological activity make it a compound of significant research interest across multiple therapeutic areas.
Mechanism of Action
Antioxidant Radical Scavenging
Ferulic acid (4-hydroxy-3-methoxycinnamic acid) is a ubiquitous hydroxycinnamic acid found abundantly in plant cell walls, particularly in rice bran, wheat, and oats. Its potent antioxidant activity derives from its phenylpropanoid structure: the hydroxyl group on the phenyl ring donates hydrogen atoms to free radicals, while the extended conjugated double bond system stabilizes the resulting phenoxyl radical through resonance delocalization across the vinyl and carboxyl groups. This structural feature gives ferulic acid a significantly lower bond dissociation enthalpy than simple phenolics, enabling efficient scavenging of hydroxyl, peroxyl, superoxide, and nitrogen dioxide radicals (PMID: 15753103).
Nrf2/ARE Pathway Activation
Ferulic acid activates the Nrf2 (nuclear factor erythroid 2-related factor 2) signaling pathway by modifying Keap1 cysteine residues, disrupting the Keap1-Nrf2 interaction and allowing Nrf2 nuclear translocation. Nuclear Nrf2 binds antioxidant response elements (ARE) to upregulate phase II detoxification and antioxidant enzymes including heme oxygenase-1 (HO-1), glutathione S-transferase (GST), NAD(P)H quinone dehydrogenase 1 (NQO1), and gamma-glutamylcysteine ligase (GCL), the rate-limiting enzyme in glutathione synthesis (PMID: 26383540).
Anti-Inflammatory Mechanisms
Ferulic acid inhibits NF-kB activation by suppressing IKK-beta phosphorylation and preventing IkB-alpha degradation. It directly inhibits COX-2 transcription and 5-lipoxygenase activity, reducing prostaglandin E2 and leukotriene synthesis. It also suppresses NLRP3 inflammasome assembly and downstream IL-1beta/IL-18 maturation (PMID: 25454459).
Photoprotective & Anti-Aging
Ferulic acid absorbs UV radiation (peak ~320 nm in the UVA/UVB range) and synergizes with vitamins C and E to provide enhanced photoprotection. It inhibits matrix metalloproteinases (MMP-1, MMP-9) by suppressing AP-1 transcription factor activity, preserving dermal collagen integrity (PMID: 16185284).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Clinical Evidence:: Well-supported by randomized controlled trials showing measurable improvements in lipid profiles within 6 weeks. Bioavailability Challenge:: Poor water solubility limits absorption, though it's absorbed better than some other phenolic acids. Dosage Response:: 1000mg daily appears effective for cardiovascular benefits in clinical studies. Synergistic Potential:: Often used in combination with other antioxidants in skincare and supplement formulations
- Well-supported by randomized controlled trials showing measurable improvements in lipid profiles within 6 weeks
- Poor water solubility limits absorption, though it's absorbed better than some other phenolic acids
- 1000mg daily appears effective for cardiovascular benefits in clinical studies
- Often used in combination with other antioxidants in skincare and supplement formulations
Safety Profile
Safety Profile: Ferulic Acid
Common Side Effects
- Generally well tolerated at typical oral supplementation doses (150–500 mg/day)
- Mild gastrointestinal discomfort: nausea, stomach upset, or loose stools, particularly on an empty stomach
- Topical formulations may cause mild skin irritation, redness, or stinging in sensitive individuals, especially at concentrations above 1%
- Occasional headache reported at higher oral doses
- Mild hypotension symptoms (dizziness, lightheadedness) in susceptible individuals due to vasodilatory properties
Serious Adverse Effects
- Hypoglycemia: ferulic acid enhances insulin sensitivity and may potentiate glucose-lowering effects of diabetes medications, leading to dangerously low blood sugar
- Bleeding risk: demonstrated antiplatelet activity in vitro; theoretical risk of excessive bleeding when combined with anticoagulants
- Hypotension: at high doses, may cause clinically significant blood pressure reduction in patients already on antihypertensives
- No human hepatotoxicity reports, but high-dose animal studies show mild transaminase elevation
- Allergic reactions are rare but possible in individuals sensitive to phenolic compounds
Contraindications
- Known hypersensitivity to ferulic acid or related hydroxycinnamic acids (caffeic acid, coumaric acid)
- Scheduled surgery (discontinue at least 2 weeks prior due to antiplatelet effects)
- Severe hypotension or patients on multiple antihypertensive medications
- Active bleeding disorders or concurrent anticoagulant therapy without medical supervision
Drug Interactions
- Antidiabetic medications (metformin, sulfonylureas, insulin): additive hypoglycemic effect; monitor blood glucose closely
- Anticoagulants/antiplatelets (warfarin, aspirin, clopidogrel): increased bleeding risk due to antiplatelet activity
- Antihypertensives (ACE inhibitors, ARBs, calcium channel blockers): additive blood pressure reduction
- Chemotherapy agents: ferulic acid's antioxidant properties may theoretically interfere with oxidative-stress-based chemotherapy mechanisms (doxorubicin, cisplatin)
- CYP450 substrates: ferulic acid may inhibit CYP3A4 and CYP2C9 at high concentrations, potentially altering metabolism of numerous drugs
Population-Specific Considerations
- Pregnant/breastfeeding women: insufficient human safety data; dietary intake from grains is safe, but concentrated supplements should be avoided
- Diabetic patients: monitor blood glucose more frequently when initiating supplementation
- Elderly: start at lower doses due to increased sensitivity to hypotensive and hypoglycemic effects; review concurrent medications
- Renal impairment: primarily metabolized hepatically and excreted renally; dose adjustment may be needed in severe renal insufficiency
- Children: no established pediatric dosing; not recommended as a supplement for children
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 1000mg daily used in clinical trials for cardiovascular and anti-inflammatory benefits
Molecular Structure
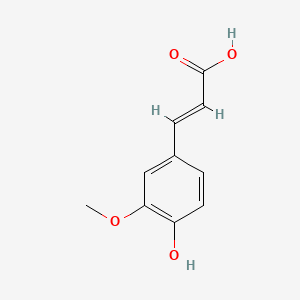
- Formula
- C10H10O4
- Weight
- 194.18 Da
- PubChem CID
- 445858
- Exact Mass
- 194.0579 Da
- LogP
- 1.5
- TPSA
- 66.8 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 3
- Complexity
- 224
Identifiers (SMILES, InChI)
InChI=1S/C10H10O4/c1-14-9-6-7(2-4-8(9)11)3-5-10(12)13/h2-6,11H,1H3,(H,12,13)/b5-3+
KSEBMYQBYZTDHS-HWKANZROSA-NSafety Profile
Common Side Effects
- Gastric Acid Concern:: May be degraded or weakened by stomach acid, potentially reducing effectiveness
- Limited Toxicity Data:: Noted as non-toxic compound in research, but long-term human safety data is limited
- Bioavailability Issues:: Poor water solubility may limit absorption and therapeutic effects
- Interaction Potential:: As with phenolic compounds, may interact with certain medications, though specific interactions not well documented
References (3)
- [1]Ferulic Acid: A Comprehensive Review
→ Comprehensive analysis showing ferulic acid demonstrates significant antioxidant, anti-inflammatory, antimicrobial, and anticancer properties, though clinical application is limited by poor water solubility and bioavailability challenges.
- [3]Ferulic Acid: Extraction, Estimation, Bioactivity and Applications for Human Health and Food
→ Review demonstrating ferulic acid exhibits antioxidant, anti-inflammatory, antimicrobial, anticancer, antidiabetic, cardiovascular and neuroprotective activities with applications in food preservation, pharmaceuticals, and cosmetics industries.
- [2]Ferulic Acid Supplementation Improves Lipid Profiles, Oxidative Stress, and Inflammatory Status in Hyperlipidemic Subjects
→ Randomized controlled trial found 1000mg daily ferulic acid significantly decreased total cholesterol (8.1%), LDL-C (9.3%), oxidative stress markers (24.5%), and inflammatory markers including hs-CRP (32.66%) in hyperlipidemic subjects over six weeks.
Fenugreek
Fenugreek (Trigonella foenum-graecum) is an herb whose seeds contain bioactive compounds including saponins, diosgenin, 4-hydroxyisoleucine, and various flavono
FGL (NCAM-Derived Peptide)
FGL is a synthetic 15-amino acid peptide derived from the second fibronectin type III (FnIII-2) domain of neural cell adhesion molecule (NCAM). It encompasses t