Piritinol
A semi-synthetic pyridoxine (vitamin B6) dimer with nootropic and anti-inflammatory properties that enhances cerebral glucose metabolism and cholinergic neurotransmission, used clinically for cognitive impairment and rheumatoid arthritis.
Overview
Piritinol (pyritinol, pyridoxine disulfide) is a semi-synthetic compound created by linking two molecules of pyridoxine (vitamin B6) via a disulfide bridge. Developed in the 1960s by Merck, piritinol was designed to create a centrally active nootropic by enhancing blood-brain barrier penetration beyond what unmodified pyridoxine achieves. Despite its structural derivation from vitamin B6, piritinol does not function as a vitamin B6 supplement — it does not serve as a cofactor for pyridoxal phosphate-dependent enzymes. Instead, it exerts independent pharmacological effects primarily related to enhancement of cerebral glucose uptake and utilization, cholinergic facilitation, and anti-inflammatory activity.
Piritinol's nootropic mechanism centers on increasing glucose transport across the blood-brain barrier by upregulating GLUT1 transporter expression and activity. Since glucose is the brain's primary energy substrate, this enhancement supports neuronal metabolism during periods of increased cognitive demand or metabolic compromise (such as aging or neurodegenerative conditions). Piritinol also modulates cholinergic neurotransmission — increasing acetylcholine release and turnover in cortical and hippocampal regions — contributing to its memory-enhancing effects. Clinical trials have demonstrated cognitive benefits in elderly patients with mild to moderate dementia, including improvements in attention, concentration, memory, and psychomotor speed. In Germany and several other countries, piritinol has been prescribed as Encephabol for age-related cognitive decline and pediatric learning difficulties.
An unexpected but clinically valuable property of piritinol is its anti-inflammatory activity. It inhibits superoxide radical generation by activated neutrophils and suppresses lysosomal enzyme release, leading to its use as a disease-modifying agent in rheumatoid arthritis in some European countries. However, this same immune-modulating activity accounts for piritinol's most notable adverse effect profile, which includes rare but serious hepatotoxicity, cholestatic hepatitis, and autoimmune reactions. These risks have limited its widespread adoption compared to other nootropics like piracetam or citicoline. Piritinol is structurally and mechanistically distinct from the racetam family, offering an alternative approach to cognitive enhancement through metabolic rather than receptor-mediated pathways.
Mechanism of Action
Piritinol (pyritinol) is a semi-synthetic derivative of vitamin B6, consisting of two pyridoxine molecules linked by a disulfide bridge. Its primary mechanism of action involves enhancing cholinergic neurotransmission by increasing the high-affinity choline uptake into neurons, thereby boosting acetylcholine synthesis and release. This cholinergic facilitation is central to its cognitive-enhancing effects, as demonstrated in studies showing recovery of cortical cholinergic deficits caused by nucleus basalis lesions in rats (Bentura et al., 1994, Journal of Neural Transmission). Piritinol also increases cortical nitric oxide synthase (NOS) activity, which may contribute to synaptic plasticity and learning processes.
Beyond cholinergic modulation, piritinol exhibits significant antioxidant properties that protect neurons from oxidative stress and free radical damage. It enhances cerebral blood flow and improves glucose and oxygen utilization in the brain, supporting neuronal energy metabolism. The compound also serves as a precursor to dopamine synthesis, contributing to mood regulation. These multi-target neuroprotective effects—spanning cholinergic enhancement, antioxidant defense, improved cerebral perfusion, and dopaminergic support—underlie its clinical use in Europe for treating cognitive disorders including dementia and Alzheimer's disease, where it is marketed under brand names such as Encephabol.
Research
Reported Effects
Dose-Response Relationship:: Higher doses (600-1200mg daily) appear more effective than lower doses, with success rates increasing with both dose and duration of treatment. Time Course:: Cognitive benefits may peak at 8-16 weeks of treatment and potentially fade with longer-term use beyond 6 months. Population Specificity:: Appears more effective in populations with existing cognitive impairment (organic brain syndrome, Alzheimer's, aging) than as a general cognitive enhancer. Limited Modern Use:: Rarely used in current clinical practice compared to when initially studied in the 1970s-1990s, suggesting limited practical effectiveness or safety concerns
- Higher doses (600-1200mg daily) appear more effective than lower doses, with success rates increasing with both dose and duration of treatment
- Cognitive benefits may peak at 8-16 weeks of treatment and potentially fade with longer-term use beyond 6 months
- Appears more effective in populations with existing cognitive impairment (organic brain syndrome, Alzheimer's, aging) than as a general cognitive enhancer
- Rarely used in current clinical practice compared to when initially studied in the 1970s-1990s, suggesting limited practical effectiveness or safety concerns
Safety Profile
Side effects may include gastrointestinal disturbances, headache, dizziness, and skin rash. Rare but serious adverse effects include cholestatic hepatitis and autoimmune reactions such as pemphigus. It is contraindicated in individuals with severe hepatic or renal impairment, rheumatoid arthritis, or hypersensitivity to penicillamine-like compounds, and should be avoided during pregnancy.
Pharmacokinetic Profile
Quick Start
- Typical Dose
- 600mg twice daily (1200mg total) was commonly used in Alzheimer's and cognitive impairment studies
Molecular Structure
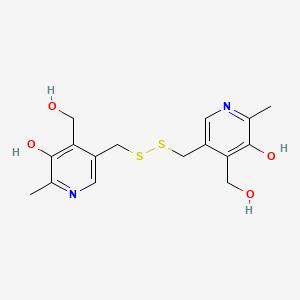
- Formula
- C16H20N2O4S2
- Weight
- 368.5 Da
- PubChem CID
- 14190
- Exact Mass
- 368.0864 Da
- LogP
- 1.2
- TPSA
- 157 Ų
- H-Bond Donors
- 4
- H-Bond Acceptors
- 8
- Rotatable Bonds
- 7
- Complexity
- 346
Identifiers (SMILES, InChI)
InChI=1S/C16H20N2O4S2/c1-9-15(21)13(5-19)11(3-17-9)7-23-24-8-12-4-18-10(2)16(22)14(12)6-20/h3-4,19-22H,5-8H2,1-2H3
SIXLXDIJGIWWFU-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Drug-Induced Pemphigus:: A serious autoimmune skin condition characterized by blistering, reported in multiple cases, typically superficial pemphigus associated with autoimmune disorders
- Sulfhydryl-Related Reactions:: Alterations in taste perception, mucocutaneous lesions, oral ulcers, and rash due to the active sulfhydryl group in the compound's structure
- Hematologic Effects:: Rare occurrences of neutropenia and thrombocytopenia reported with sulfhydryl compounds as a class
- Proteinuria:: Potential for immune-complex membranous glomerulopathy leading to protein in urine, requiring monitoring during long-term use
References (7)
- [3]Long-term effects of phosphatidylserine, pyritinol, and cognitive training in Alzheimer's disease. A neuropsychological, EEG, and PET investigation
→ In 70 Alzheimer's patients treated for 6 months, the combination of cognitive training with pyritinol (2x600mg/day) showed effects on brain function measures, though neuropsychological improvements were best documented at 8-16 weeks and faded toward treatment end.
- [1]Psychopharmacological effects of pyritinol in normal volunteers
→ A double-blind study in 12 healthy volunteers found that pyritinol (600-1200mg) produced significant improvements in Critical Flicker Fusion and Choice Reaction Time tests, with no significant side effects, suggesting cognitive enhancement effects.
- [4]Dose-effect relationship of orally administered Pyritinol in the chronic organic brain syndrome
→ A study of 161 patients with chronic organic brain syndrome found that treatment success rate increased significantly with higher doses and longer treatment duration, suggesting that recommended daily doses should be exceeded in severe cases.
- [5]Effects of nootropic drugs on brain cholinergic and dopaminergic transmission
→ Six-day administration of pyritinol (50mg/kg/day) in rats increased high-affinity choline uptake in the hippocampus without changing striatal dopamine levels, suggesting enhancement of acetylcholine synthesis.
- [6]Drug-induced pemphigus
→ A literature review identified pyritinol as responsible for 4 cases of drug-induced pemphigus, with superficial pemphigus seen in about 75% of cases, though prognosis was better than non-drug-induced pemphigus under immunosuppressive therapy.
- [7]Adverse effects profile of sulfhydryl compounds in man
→ Compounds with active sulfhydryl groups (including pyritinol) share a distinctive adverse reaction pattern including alterations in taste perception, mucocutaneous lesions, proteinuria, and pemphigus, though these reactions occur with variable frequency.
- [2]Effects of the chronic administration of pyrithioxin on behaviour and cholinergic function in young and aged rats
→ Chronic pyrithioxin administration (200-600 mg/kg over 6 weeks) enhanced passive avoidance retention in both young and old rats, and improved sensitivity to foot shock and water maze retention in aged animals, without affecting locomotor activity.
Piracetam
Piracetam is the original racetam and one of the first synthetic nootropics, developed in the 1960s. It enhances memory and cognitive function through modulatio
PNC-27
PNC-27 is an experimental anti-cancer peptide created by a supercomputer at SUNY Downstate Medical Center in 2000. It contains an HDM-2 binding domain from p53