Piracetam
The original racetam nootropic and prototype cognitive enhancer, a cyclic derivative of GABA that modulates glutamatergic neurotransmission and membrane fluidity to support memory, learning, and neuroprotection without sedation or stimulation.
Overview
Piracetam (2-oxo-1-pyrrolidine acetamide) is the founding member of the racetam class of nootropics, synthesized in 1964 by Romanian chemist Corneliu Giurgea, who also coined the term "nootropic" to describe its unique pharmacological profile — enhancing cognition without the sedation, stimulation, or toxicity associated with psychotropic drugs. Despite being derived from GABA, piracetam does not bind GABA receptors and shows no GABAergic activity. Instead, its primary mechanism involves allosteric modulation of AMPA-type glutamate receptors, enhancing excitatory neurotransmission and facilitating long-term potentiation (LTP), the synaptic process underlying learning and memory formation. Piracetam also modulates NMDA receptors and improves cell membrane fluidity by interacting with phospholipid head groups, which may enhance signal transduction and neurotransmitter receptor function.
Piracetam has been extensively studied in clinical trials spanning several decades. In elderly populations with cognitive decline, meta-analyses have shown statistically significant improvements in memory and global cognitive function compared to placebo. It is approved as a prescription medication in many European and Asian countries for indications including age-related cognitive decline, post-stroke aphasia and recovery, myoclonus (particularly cortical myoclonus, where high-dose piracetam is a recognized treatment), vertigo, dyslexia, and sickle cell disease (where it reduces red blood cell sickling and painful crises by improving membrane deformability). Its safety profile is notably benign — piracetam is water-soluble, shows no significant hepatic metabolism, is renally excreted largely unchanged, and has an extremely high LD50 in animal studies.
Piracetam's relatively modest potency compared to newer racetams has led to the development of numerous derivatives including phenylpiracetam, aniracetam, oxiracetam, pramiracetam, coluracetam, and fasoracetam — each with enhanced potency or distinct receptor selectivity. Typical nootropic dosing ranges from 1,200-4,800 mg/day, often divided into two or three doses. Piracetam is commonly stacked with choline sources such as alpha-GPC or CDP-choline to support acetylcholine synthesis and mitigate occasional headaches attributed to choline depletion. In the United States, piracetam occupies a regulatory gray area — it is not FDA-approved and cannot be marketed as a dietary supplement, yet it is not a controlled substance.
Mechanism of Action
Piracetam (2-oxo-1-pyrrolidine acetamide) is the founding compound of the racetam class of nootropics, structurally derived from GABA but lacking direct GABAergic activity. Its mechanism is multifaceted and primarily manifests under conditions of neuronal impairment. At the molecular level, piracetam binds to a unique allosteric site on AMPA-type glutamate receptors, as demonstrated by X-ray crystallography of the GluA2 ligand-binding domain. This positive allosteric modulation enhances glutamatergic excitatory neurotransmission, facilitating long-term potentiation (LTP) and synaptic plasticity, which are fundamental to learning and memory formation.
A central mechanism involves piracetam's interaction with membrane phospholipids. It restores fluidity to aged or damaged neuronal membranes by intercalating with the polar head groups of the phospholipid bilayer. This normalization of membrane dynamics improves the function of membrane-bound proteins including receptors, ion channels, and neurotransmitter transporters. Through this membrane effect, piracetam indirectly enhances cholinergic neurotransmission by increasing muscarinic acetylcholine receptor density and improving acetylcholine turnover in the hippocampus, without directly binding to cholinergic receptors.
Piracetam also exhibits significant neuroprotective and rheological properties. It stabilizes mitochondrial function and enhances cerebral energy metabolism by increasing ATP synthesis and glucose/oxygen utilization under hypoxic conditions. Its rheological effects include reducing blood viscosity, decreasing platelet aggregation, and improving erythrocyte deformability, collectively enhancing cerebral microcirculation. These properties make piracetam particularly effective in age-related cognitive decline, post-stroke recovery, and conditions involving impaired cerebral blood flow.
Research
Reported Effects
Cumulative Benefits:: Effects often improve over weeks.. Subtle Action:: Not immediately noticeable like stimulants.. Synergy:: Works well with choline and other racetams.
- Effects often improve over weeks.
- Not immediately noticeable like stimulants.
- Works well with choline and other racetams.
Safety Profile
Common side effects include headaches, insomnia, nervousness, agitation, and gastrointestinal discomfort. It is contraindicated in individuals with severe renal impairment, Huntington's disease, or cerebral hemorrhage. Piracetam may enhance the effects of anticoagulants and thyroid hormones, so medical supervision is advised when used concurrently with these medications.
Pharmacokinetic Profile
Piracetam — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- 1.6-4.8g daily, divided into 2-3 doses.
Molecular Structure
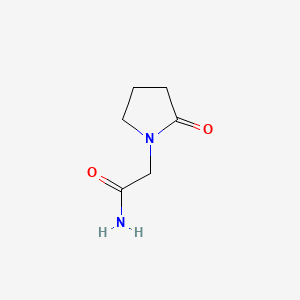
- Formula
- C6H10N2O2
- Weight
- 142.16 Da
- PubChem CID
- 4843
- Exact Mass
- 142.0742 Da
- LogP
- -1.3
- TPSA
- 63.4 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 2
- Rotatable Bonds
- 2
- Complexity
- 167
Identifiers (SMILES, InChI)
InChI=1S/C6H10N2O2/c7-5(9)4-8-3-1-2-6(8)10/h1-4H2,(H2,7,9)
GMZVRMREEHBGGF-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Headaches:: Common without adequate choline.
- Insomnia:: Possible if taken late in day.
- Brain Fog:: Paradoxically possible at wrong doses.
- Extremely Safe:: Excellent safety record over decades.
References (10)
- [8]Efficacy and safety of compound porcine cerebroside and ganglioside injection versus piracetam on cognition
→ Large controlled trial comparing CPCGI injection effectiveness to piracetam for cognitive and functional outcomes in mild to moderate traumatic brain injury.
- [5]Cognitive effects of piracetam in adults with memory impairment: A systematic review and meta-analysis
→ Analysis of multiple clinical trials found no significant difference in memory enhancement between those taking piracetam and the placebo group.
- [6]Physicochemical investigations of nootropic drug piracetam for enhanced solubilization
→ Study demonstrated that mixing surfactants improves drug solubilization, showing that the process is spontaneous and entropically favorable.
- [1]Piracetam for fetal distress in labour
→ One study suggested piracetam might reduce the likelihood of a caesarean section, but there was no significant impact on neonatal health.
- [2]Piracetam for acute ischaemic stroke
→ Research found no significant evidence to support its benefit, with a slight, but not statistically significant, trend towards increased early death.
- [3]Piracetam for reducing the incidence of painful sickle cell disease crises
→ Review of three trials determined evidence was insufficient and of poor quality despite some potential benefits noted in certain trials.
- [4]Piracetam in the treatment of cortical myoclonus
→ Individualized high dosages (7-24g daily) can be very effective with minimal side effects, with improvements observed at up to 45g daily.
- [7]Effect of Piracetam and Iron Treatment on Heart Rate Variability in Patients With Breath-Holding Spell
→ Research observed significant improvements in HRV parameters and a reduction in the number of breath-holding episodes with piracetam treatment.
- [9]Docosahexaenoic Acid Plus Piracetam Versus Piracetam Alone for Treatment of Breath-Holding Spells
→ Adding DHA to piracetam significantly reduced the frequency and severity of breath-holding spells with 16% experiencing spells versus 50% with piracetam alone.
- [10]Piracetam mitigates nephrotoxicity induced by cisplatin via the AMPK-mediated signaling pathways
→ Investigation found piracetam improved kidney function, reduced tissue damage and inflammation against chemotherapy-induced kidney damage.
Piperine
Piperine is an alkaloid compound extracted from black pepper (Piper nigrum) that acts as a bioavailability enhancer by inhibiting drug-metabolizing enzymes and
Piritinol
Piritinol (also known as pyrithioxin or pyritinol) is a nootropic compound consisting of two pyridoxine (vitamin B6) molecules bound by a disulfide bridge. It a