HMG
Human Menopausal Gonadotropin (hMG/Menotropins) is a combination of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) extracted from postmenopausal urine, used clinically for ovulation induction in IVF and treatment of male hypogonadotropic hypogonadism.
Overview
hMG is produced by extracting and purifying gonadotropins from pooled postmenopausal urine, which contains elevated FSH and LH levels due to the loss of ovarian negative feedback. Modern highly purified hMG preparations (HP-hMG, e.g., Menopur) undergo extensive chromatographic purification and supplement LH activity with hCG derived from the same urinary source, as native urinary LH is largely lost during purification. The resulting product provides FSH activity for follicular development alongside LH/hCG activity for steroidogenesis.
Lunenfeld (2004) published a landmark historical review documenting the 40-year journey from the first successful ovulation induction with hMG in 1961 to the development of recombinant gonadotropins, establishing the foundational role of hMG in reproductive medicine.
Mechanism of Action
hMG provides dual gonadotropin stimulation through two distinct receptor pathways:
- FSH component: Binds FSH receptors (FSHR) on granulosa cells (females) and Sertoli cells (males). FSHR is a Gs-coupled GPCR that activates adenylyl cyclase, increasing cAMP and activating PKA. In females, this drives follicular growth, granulosa cell proliferation, aromatase expression (CYP19A1), and inhibin B secretion. In males, FSH stimulates Sertoli cell proliferation and function, including production of androgen-binding protein (ABP), inhibin B, and support of spermatogenesis
- LH/hCG component: Binds LH/CG receptors (LHCGR) on theca cells (females) and Leydig cells (males). Activates cAMP/PKA cascade driving steroidogenesis — androgen production in theca cells (which is aromatized to estradiol by granulosa cells) and testosterone production in Leydig cells. The two-cell, two-gonadotropin model requires both FSH and LH for normal follicular estradiol production
- Synergistic action: The combination of FSH and LH in hMG recapitulates physiological gonadotropin signaling more closely than FSH alone, potentially improving oocyte quality through optimal steroidogenic milieu in the follicular microenvironment
Research
Male Infertility & Hypogonadotropic Hypogonadism
hMG is a first-line treatment for male infertility caused by hypogonadotropic hypogonadism (HH), typically in combination with hCG. The standard protocol initiates hCG alone (1500-2000 IU twice weekly) to stimulate Leydig cell testosterone production, followed by addition of hMG (75-150 IU three times weekly) after testosterone levels normalize, to drive spermatogenesis via FSH-mediated Sertoli cell stimulation. Treatment duration averages 6-18 months before sperm appear in the ejaculate. Studies report spermatogenesis induction rates of 70-90% in men with acquired HH.
Comparison with Recombinant FSH
The debate between urinary hMG and recombinant FSH (rFSH) has generated extensive clinical trial data. Key differences include:
- Batch consistency: rFSH offers more consistent bioactivity between batches; hMG has inherent biological variability
- LH activity: hMG provides endogenous LH/hCG activity; rFSH provides FSH alone (LH supplementation may be added separately)
- Immunogenicity: hMG carries theoretical risk of urinary protein contaminants; rFSH is produced in CHO cells with higher purity
- Clinical outcomes: Meta-analyses generally show equivalent live birth rates, though individual studies have suggested advantages for one or the other in specific patient populations
- Cost: hMG is typically less expensive than rFSH, influencing treatment accessibility globally
Ovulation Induction in Anovulatory Infertility
Beyond IVF, hMG is used for ovulation induction in anovulatory women, particularly those with WHO Group II anovulation (hypothalamic-pituitary dysfunction) who have failed clomiphene citrate. Low-dose step-up protocols (starting at 75 IU/day) minimize the risk of OHSS and multiple pregnancy while achieving monofollicular development in 60-70% of cycles.
IVF Protocols & Controlled Ovarian Stimulation
hMG is a primary agent for controlled ovarian stimulation (COS) in IVF. The Menopur Ovarian Stimulation Trial in Europe (MOST) and other large studies demonstrated that HP-hMG produces comparable clinical pregnancy rates to recombinant FSH, with some evidence suggesting improved embryo quality and implantation rates with hMG. The LH/hCG activity in hMG may provide a more physiological follicular environment by supporting theca cell androgen production, which serves as substrate for granulosa cell estradiol synthesis.
Al-Inany et al. (2009) conducted a Cochrane systematic review comparing hMG with recombinant FSH for ovarian stimulation in IVF, finding no significant difference in live birth rates but noting potential advantages of hMG in specific patient subgroups, particularly poor responders who may benefit from exogenous LH activity.
Safety Profile
hMG shares the safety profile common to exogenous gonadotropin therapy:
- Ovarian hyperstimulation syndrome (OHSS): The most significant risk, occurring in 1-5% of stimulated cycles (mild to moderate) and 0.5-2% (severe). The LH/hCG component may contribute to OHSS through VEGF-mediated vascular permeability increases. Risk is mitigated through careful dose titration and ultrasound monitoring
- Multiple pregnancy: Ovarian stimulation with hMG increases the risk of multiple gestation (twins: 15-20%, higher-order multiples: 1-3% in non-IVF cycles). Strict monitoring with cycle cancellation criteria reduces this risk
- Injection site reactions: Pain, redness, swelling, and bruising at injection sites. Subcutaneous administration is generally better tolerated than intramuscular
- Allergic reactions: Rare hypersensitivity to urinary-derived proteins. Risk is lower with highly purified preparations
- Ectopic pregnancy: Slightly increased risk compared to natural conception
- Ovarian torsion: Rare complication associated with enlarged ovaries from stimulation
Pharmacokinetic Profile
HMG — Pharmacokinetic Curve
Intramuscular injection, Subcutaneous injectionQuick Start
- Typical Dose
- 75-150 IU for ovulation induction; 150-300 IU for IVF
- Frequency
- Daily during stimulation phase (7-12 days)
- Route
- Intramuscular injection, Subcutaneous injection
- Cycle Length
- 7-12 days per cycle
- Storage
- Lyophilized: Room temperature. Reconstituted: 2-8°C, use immediately after reconstitution
Molecular Structure
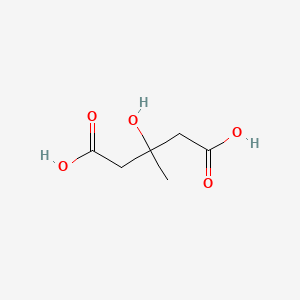
- Formula
- C6H10O5
- Weight
- 162.14 Da
- CAS
- 9002-68-0
- PubChem CID
- 1662
- Exact Mass
- 162.0528 Da
- LogP
- -1.2
- TPSA
- 94.8 Ų
- H-Bond Donors
- 3
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 4
- Complexity
- 158
Identifiers (SMILES, InChI)
InChI=1S/C6H10O5/c1-6(11,2-4(7)8)3-5(9)10/h11H,2-3H2,1H3,(H,7,8)(H,9,10)
NPOAOTPXWNWTSH-UHFFFAOYSA-NResearch Indications
Female Fertility (FDA-Approved)
Stimulates follicular development in anovulatory women.
Used in IVF protocols to stimulate multiple follicle development.
Effective for inducing ovulation in polycystic ovary syndrome patients.
Male Fertility
FSH promotes sperm production; LH stimulates testosterone for sperm development.
Treats male infertility due to insufficient gonadotropin production.
Testosterone Support
LH component stimulates testicular testosterone production.
Research Protocols
subcutaneous Injection
Human menopausal gonadotropin. 3x weekly, typically combined with hCG therapy.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Standard dose | 75 IU | 3x weekly (Mon/Wed/Fri) | Weeks 1-12+(Extendable to 16 weeks. 3.0 mL per dose exceeds standard 1 mL syringe — use 3 mL syringe or split into 3 separate injections.) |
Reconstitution Guide (75mg vial + 3mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 3.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 25 IU/mL
- For 75 IU dose: draw full 3.0 mL — use 3 mL syringe or split into 3 x 1.0 mL injections
- Store reconstituted vial refrigerated at 2-8°C
Interactions
Peptide Interactions
The debate between urinary hMG and recombinant FSH (rFSH) has generated extensive clinical trial data. Key differences include: - Batch consistency: rFSH offers more consistent bioactivity between batches; hMG has inherent biological variability - LH activity: hMG provides endogenous LH/hCG activ...
What to Expect
What to Expect
Follicular recruitment begins
Follicular growth continues; monitoring required
Follicles reach mature size; trigger ovulation
Ovulation occurs ~36 hours after hCG trigger
Safety Profile
Common Side Effects
- Injection site reactions
- Abdominal discomfort
- Ovarian enlargement
- Mood changes
- Headache
Contraindications
- Primary ovarian failure
- Uncontrolled thyroid or adrenal dysfunction
- Sex hormone-dependent tumors
- Abnormal uterine bleeding of unknown cause
- Ovarian cysts (not due to PCOS)
- Pregnancy
Discontinue If
- Severe abdominal pain or bloating
- Rapid weight gain
- Difficulty breathing
- Nausea and vomiting
- Decreased urination
Quality Indicators
What to look for
- White lyophilized powder
- Pharmaceutical grade product
- Proper cold chain maintained
- Clear solution after reconstitution
Caution
- Research-grade products lack quality assurance
Red flags
- Discolored powder or solution
- Particulates visible
- Exposed to temperature extremes
Frequently Asked Questions
References (20)
- [3]
- [1]
- [1]
- [2]HMG vs Recombinant FSH in PCOS Patients Undergoing IVF (2013)
- [3]Effect of HMG and HP-FSH on IVF Outcomes (1996)
- [4]Generic hMG vs Costly FSH for Ovulation Induction (2001)
- [6]Burgués S et al. hMG vs recombinant FSH in partners of men with obstructive and non-obstructive azoospermia. Reprod Biomed Online (2001)
- [19]Levi-Setti et al — HP-hMG versus rFSH in IVF: updated meta-analysis of live birth rates Reprod Biomed Online (2023)
- [20]Alviggi et al — LH activity in controlled ovarian stimulation: when, how much, and to whom? Fertil Steril (2022)
- [21]Bosch et al — Personalized ovarian stimulation: the role of gonadotropin formulation selection Hum Reprod Update (2023)
- [4]Bosch E et al. Highly purified hMG versus recombinant FSH in ovarian hyperstimulation with GnRH antagonists -- a randomized study. Hum Reprod (2008)
- [7]Warne DW et al. A combined analysis of data to identify predictive factors for spermatogenesis in men with hypogonadotropic hypogonadism. Hum Reprod (2009)
- [10]European Society of Human Reproduction and Embryology. Ovarian Stimulation for IVF/ICSI Guideline. (2019)
- [11]Nagels HE et al. Androgens (dehydroepiandrosterone or testosterone) for women undergoing assisted reproduction. Cochrane Database Syst Rev (2015)
- [22]
- [2]Al-Inany HG et al. Highly purified hMG versus recombinant FSH for ovarian stimulation in IVF: a Cochrane review. Cochrane Database Syst Rev (2008)
- [8]Andersen AN et al. Clinical outcome following stimulation with HP-hMG or rFSH in patients undergoing IVF. Reprod Biomed Online (2006)
- [9]Lehert P et al. Recombinant human follicle-stimulating hormone (r-hFSH) plus recombinant luteinizing hormone versus human menopausal gonadotropin (hMG) for ovarian stimulation during assisted reproductive technology. Cochrane Database Syst Rev (2014)
- [12]Ding T et al. hMG vs rFSH for ovarian stimulation in assisted reproduction cycles. Reprod Biol Endocrinol (2020)
- [5]Balasch J et al. The role of LH in human follicle development and ovarian function. Reprod Biomed Online (2010)
HMB
Beta-hydroxy-beta-methylbutyrate (HMB) is a metabolite of the amino acid leucine that helps reduce muscle protein breakdown and may enhance muscle protein synth
Honokiol
Honokiol is a bioactive polyphenol compound extracted from Magnolia bark that has been used in traditional medicine for centuries. It works primarily as a posit