Thymogen
A synthetic dipeptide bioregulator (L-glutamyl-L-tryptophan) that functions as a potent immunomodulator by stimulating T-lymphocyte differentiation and enhancing both cellular and humoral immune responses.
Thymogen (EW dipeptide) is a Khavinson bioregulator consisting of glutamic acid and tryptophan, originally isolated from calf thymus extracts (Thymalin) in the late 1980s. Developed by Professor Vladimir Khavinson, it has been registered in Russia since 1990 in multiple forms including injectable solution, nasal spray, and topical cream. Thymogen modulates both humoral and cellular immunity, activates T-cell differentiation, and has demonstrated geroprotective (anti-aging) and antitumor activities in research studies.
Mechanism of Action
Thymogen works through multiple immunomodulatory mechanisms: (1) activates T-cell differentiation and T-cell recognition of peptide-MHC complexes, (2) induces changes in intracellular cyclic nucleotide composition, (3) activates neutrophilic chemotaxis and phagocytosis, (4) normalizes T-lymphocyte concentrations and ratios (CD3+, CD4+, CD8+), (5) stimulates production of immunoglobulins (IgA, IgG, IgE, IgM), and (6) enhances lymphocyte differentiation receptor expression. Research suggests Thymogen may interact specifically with the AACG DNA sequence, affecting gene expression. The peptide is rapidly distributed to thymus, lymph nodes, liver, adrenals, and kidneys.
Reconstitution Calculator
Thymogen
Thymogen (EW dipeptide) is a Khavinson bioregulator consisting of glutamic acid

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Safety Profile
Safety Profile: Thymogen
Common Side Effects
- Generally well tolerated in clinical studies, primarily conducted in Russia and Eastern European countries
- Mild nasal irritation or rhinitis when administered intranasally (the most common route)
- Mild injection site reactions (pain, redness) with parenteral administration
- Occasional headache and mild fatigue
- Mild allergic skin reactions including transient rash or pruritus
Serious Adverse Effects
- Limited Western clinical trial data; safety profile primarily based on Russian pharmacological literature
- Rare allergic reactions including urticaria and angioedema
- Theoretical risk of immune overstimulation in patients with autoimmune conditions, though clinical evidence is sparse
- No significant hepatotoxicity, nephrotoxicity, or hematological toxicity reported in available literature
- Long-term safety data beyond 10-day treatment courses are limited
Contraindications
- Known hypersensitivity to thymogen (L-glutamyl-L-tryptophan) or any formulation excipients
- Active autoimmune diseases (may exacerbate immune dysregulation)
- Pregnancy and lactation (insufficient safety data)
- Severe allergic conditions without medical supervision
- Concurrent use with other immunostimulants without medical oversight
Drug Interactions
- Immunosuppressants (cyclosporine, tacrolimus, methotrexate): May counteract immunosuppressive therapy due to immune-stimulating properties
- Other thymic peptides (thymalin, thymulin): Additive immunostimulatory effects; avoid concurrent use without medical supervision
- Corticosteroids: May partially antagonize the immunostimulatory effects of thymogen
- Vaccines: Theoretical enhancement of immune response to vaccines; timing should be discussed with a physician
- No significant CYP450 interactions reported
Population-Specific Considerations
- Elderly: Primary use population for immunosenescence; generally well tolerated at standard doses
- Pediatric: Used in Russian clinical practice for recurrent infections in children; dosing adjusted by age
- Immunocompromised patients: Primary indication, but requires medical supervision to calibrate immune response
- Post-surgical patients: Used in some protocols to reduce post-operative infection risk; ensure no conflict with other immunoactive medications
- Autoimmune patients: Generally contraindicated; immune stimulation may worsen disease activity
Pharmacokinetic Profile
- Half-life
- Not established
Quick Start
- Typical Dose
- 10-20mg daily during cycle
- Frequency
- Daily for 10-20 days, then break
- Cycle Length
- 10-20 day cycles (bioregulator protocol)
- Storage
- Injectable: 2-8°C. Nasal/topical: per product directions. Powder: room temp
Molecular Structure
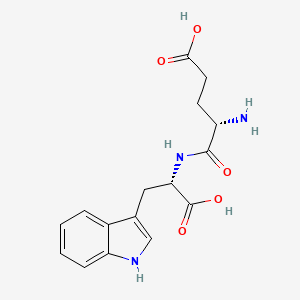
- Formula
- C16H19N3O5
- Weight
- 333 Da
- Length
- 2 amino acids
- PubChem CID
- 100094
- Exact Mass
- 333.1325 Da
- LogP
- -2.7
- TPSA
- 146 Ų
- H-Bond Donors
- 5
- H-Bond Acceptors
- 6
- Rotatable Bonds
- 8
- Complexity
- 484
Identifiers (SMILES, InChI)
InChI=1S/C16H19N3O5/c17-11(5-6-14(20)21)15(22)19-13(16(23)24)7-9-8-18-12-4-2-1-3-10(9)12/h1-4,8,11,13,18H,5-7,17H2,(H,19,22)(H,20,21)(H,23,24)/t11-,13-/m0/s1
LLEUXCDZPQOJMY-AAEUAGOBSA-NResearch Indications
Immune Support
Modulates both humoral and cellular immune responses for balanced immunity.
Activates T-cell differentiation and normalizes CD3+, CD4+, CD8+ populations.
Stimulates production of IgA, IgG, IgE, and IgM antibodies.
Studied for improving surgical outcomes through immune support.
Anti-Aging Research
Demonstrated geroprotector activity in aging research.
In combination with pineal peptides, showed significant mean lifespan increases in animal models.
Inhibits spontaneous carcinogenesis and shows robust antitumor effects in research.
Clinical Research Areas
Studied in Russia for regulating immune response in oncology patients.
Research interest in cardiovascular applications.
Investigated for metabolic and immune aspects of diabetes.
Research Protocols
subcutaneous Injection
Injectable form for intramuscular or subcutaneous administration. This is the original delivery method registered in Russia. Typical protocols involve 10-20 day cycles repeated 2-3 times per year.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Standard immune support | 10-20 mg | Daily for 10-20 days | —(Route: IM or SubQ) |
| Maintenance protocol | 20 mg | 2-3 cycles per year | —(Route: IM or SubQ) |
Reconstitution Guide (mg vial + mL BAC water)
- Clean work area and hands
- Reconstitute with bacteriostatic water
- Inject slowly down vial wall
- Gently swirl until dissolved
- Store refrigerated at 2-8°C
- Use within 4 weeks
intranasal Injection
Metered-dose nasal spray form registered in Russia. Convenient for non-injection administration with good absorption through nasal mucosa.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Nasal administration | As per product directions | Daily during cycle | —(Route: Intranasal spray) |
topical
0.05% cream formulation registered in Russia for external use. Applied to skin for local effects.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Topical use | Apply thin layer | As directed | —(Route: Topical cream) |
Interactions
Peptide Interactions
Combination showed significant lifespan increases and robust antitumor activity in research.
Thymogen was isolated from Thymalin; they share complementary thymus-supporting mechanisms.
Both are Khavinson thymus dipeptides; often combined in comprehensive immune protocols.
Different thymic peptides with complementary immune mechanisms.
Different structures and mechanisms; can be used together.
What to Expect
What to Expect
Rapid tissue distribution to thymus, lymph nodes, liver, kidneys
T-cell activation and differentiation begins
Immunoglobulin production stimulated; immune markers improve
Effects persist due to cellular and epigenetic changes
Cumulative geroprotective benefits with regular cycling
Safety Profile
Common Side Effects
- Generally well-tolerated
- Injection site reactions (mild)
- Minimal side effects reported in clinical use
Contraindications
- Active autoimmune diseases (use with caution)
- Organ transplant recipients on immunosuppression
- Known hypersensitivity to component amino acids
- Pregnancy or breastfeeding
Discontinue If
- Signs of allergic reaction
- Unusual immune symptoms
- Severe injection site reactions
Quality Indicators
What to look for
- White to off-white lyophilized powder
- Clear solution after reconstitution
- Certificate of analysis with purity verification
- Proper packaging and labeling
Caution
- Unknown source or unverified purity
Red flags
- Discolored powder
- Cloudy or particulate solution
- Damaged packaging or broken seal
References (4)
- [3]Peptide Bioregulators from Thymus and Pineal Gland (2003)
- [4]Thymogen: Mechanism of Action Studies
- [2]
- [1]
Thymalin
**Thymalin** is a synthetic dipeptide (L-glutamyl-L-tryptophan) originally isolated from bovine thymus tissue by Vladimir Khavinson and colleagues at the Saint
Thymopentin
**Thymopentin** (TP-5) is a synthetic pentapeptide with the sequence Arg-Lys-Asp-Val-Tyr, representing the minimal active fragment (residues 32-36) of the 49-am