Rapamycin
A macrolide compound originally isolated from soil bacteria on Easter Island that inhibits the mTOR kinase, approved as an immunosuppressant for organ transplantation and now under intense investigation as the most promising pharmacological agent for extending mammalian lifespan.
Overview
Rapamycin (sirolimus) is a macrocyclic lactone produced by Streptomyces hygroscopicus, first isolated in 1972 from soil samples collected on Rapa Nui (Easter Island) — the origin of its name. Initially developed as an antifungal agent, rapamycin's profound immunosuppressive properties redirected its clinical development, leading to FDA approval in 1999 as Rapamune for prevention of organ transplant rejection. Its mechanism centers on formation of a complex with the intracellular protein FKBP12, which then binds to and inhibits mTOR complex 1 (mTORC1) — the mechanistic target of rapamycin. mTORC1 is a master regulator of cell growth, protein synthesis, and metabolism, integrating signals from nutrients (particularly amino acids and glucose), growth factors (insulin/IGF-1), and cellular energy status (ATP/AMP ratio) to control anabolic processes.
The longevity implications of mTOR inhibition have made rapamycin arguably the most significant compound in aging research. In 2009, the NIA Interventions Testing Program demonstrated that rapamycin extended median and maximum lifespan in genetically heterogeneous mice when started at 20 months of age — equivalent to approximately 60 human years — making it the first pharmacological agent to extend lifespan in a mammalian species when initiated late in life. Subsequent studies have confirmed lifespan extension across multiple mouse strains, both sexes, and various dosing regimens, with extension ranging from 9-26%. The mechanisms underlying this longevity effect are multifaceted: mTORC1 inhibition activates autophagy (the cellular recycling program that declines with age), reduces senescent cell accumulation, improves stem cell function, reduces chronic inflammation ("inflammaging"), enhances immune function in aged organisms (paradoxically, given its immunosuppressive role at high doses), and shifts cellular metabolism from growth-promoting to maintenance-promoting programs.
Clinical translation of rapamycin for longevity is actively being pursued. Low-dose, intermittent rapamycin protocols (typically 1-6 mg once weekly) are being tested in human aging trials, including the landmark PEARL (Participatory Evaluation of Aging with Rapamycin for Longevity) trial. This intermittent dosing strategy aims to capture mTORC1 inhibition benefits while minimizing effects on mTORC2 (which mediates most immunosuppressive side effects) and avoiding metabolic complications seen with chronic high-dose use (hyperglycemia, hyperlipidemia, impaired wound healing). Rapamycin's longevity effects synergize conceptually with other interventions targeting overlapping pathways, including metformin (AMPK activation), NAD+ precursors (sirtuin activation), senolytic compounds like fisetin and quercetin (senescent cell clearance), and spermidine (autophagy induction). Rapamycin analogs (rapalogs) including everolimus and temsirolimus are used in oncology for their antiproliferative effects.
Mechanism of Action
Mechanism of Action: Rapamycin
Rapamycin (sirolimus) is a macrolide compound originally isolated from Streptomyces hygroscopicus, discovered on Easter Island (Rapa Nui). It is a highly specific allosteric inhibitor of mechanistic target of rapamycin complex 1 (mTORC1).
Molecular Mechanism of mTORC1 Inhibition
Rapamycin first binds to the cytoplasmic immunophilin FKBP12 (FK506-binding protein 12) with high affinity (Kd ~0.2 nM). The rapamycin-FKBP12 complex then binds to the FRB (FKBP12-rapamycin binding) domain of mTOR, allosterically inhibiting mTORC1 kinase activity. Importantly, rapamycin does not directly inhibit mTOR catalytic activity but rather disrupts mTORC1 substrate recruitment. Acute rapamycin exposure selectively inhibits mTORC1; prolonged exposure can also disrupt mTORC2 assembly in some cell types.
Downstream Signaling Effects
mTORC1 inhibition blocks phosphorylation of two critical substrates: (1) p70 S6 kinase 1 (S6K1): dephosphorylation inactivates ribosomal protein S6 and eIF4B, reducing ribosomal biogenesis and translation of 5'TOP mRNAs encoding ribosomal proteins and elongation factors; (2) 4E-BP1: hypophosphorylated 4E-BP1 sequesters eIF4E, preventing eIF4F complex assembly and cap-dependent translation initiation. Together, these effects profoundly reduce protein synthesis, cell growth, and proliferation.
Autophagy Induction
Active mTORC1 phosphorylates ULK1 at Ser757, disrupting the ULK1-AMPK interaction and preventing autophagy initiation. Rapamycin relieves this inhibition, allowing AMPK-mediated ULK1 activation at Ser317/Ser777. Activated ULK1 phosphorylates Beclin-1 and ATG14, initiating phagophore formation and autophagosome maturation. Enhanced autophagy clears damaged mitochondria (mitophagy), protein aggregates (aggrephagy), and dysfunctional organelles, a key mechanism underlying rapamycin's longevity effects.
Immunosuppressive Mechanisms
Rapamycin blocks IL-2 and IL-15-stimulated T-cell proliferation by preventing mTORC1-dependent cell cycle progression through G1 to S phase. Unlike calcineurin inhibitors, it does not block initial T-cell activation or IL-2 production but rather prevents the proliferative response. Notably, rapamycin promotes FOXP3+ regulatory T cell (Treg) differentiation while suppressing Th1 and Th17 effector responses, creating a relatively tolerogenic immune profile.
Longevity and Anti-aging
mTORC1 integrates nutrient, growth factor, and energy signals to drive anabolic processes. Rapamycin mimics aspects of caloric restriction by reducing mTORC1 activity, which: reduces cellular senescence burden, decreases the SASP, enhances stem cell function, improves proteostasis through autophagy, and reduces age-related inflammation (inflammaging). Rapamycin extends lifespan in multiple model organisms, including a 10-15% extension in genetically heterogeneous mice (NIA Interventions Testing Program).
Metabolic Considerations
Chronic mTORC1 inhibition can disrupt mTORC2-mediated Akt activation, potentially impairing glucose tolerance and lipid metabolism. This has led to intermittent or pulsed dosing strategies in longevity applications, which maintain mTORC1 inhibition while allowing mTORC2 recovery.
Research
Reported Effects
Animal Evidence Strong:: Rapamycin is the only drug consistently shown to extend lifespan across multiple mammalian species in rigorous testing. Human Data Limited:: Human longevity data is still emerging, with some concerning recent findings about epigenetic aging acceleration. Individual Variation:: Effects appear highly variable between individuals, with some reporting benefits for energy and recovery while others experience negative effects. Dosing Matters:: Intermittent dosing (e.g., once weekly) may provide benefits while reducing side effects compared to daily use
- Rapamycin is the only drug consistently shown to extend lifespan across multiple mammalian species in rigorous testing
- Human longevity data is still emerging, with some concerning recent findings about epigenetic aging acceleration
- Effects appear highly variable between individuals, with some reporting benefits for energy and recovery while others experience negative effects
- Intermittent dosing (e.g., once weekly) may provide benefits while reducing side effects compared to daily use
Safety Profile
Safety Profile: Rapamycin (Sirolimus)
Common Side Effects
- Hyperlipidemia: Elevated triglycerides (40–60% of patients) and cholesterol are very common and dose-dependent
- Hematologic effects: Thrombocytopenia, anemia, and leukopenia due to bone marrow suppression
- Peripheral edema and joint pain
- Mouth ulcers (aphthous stomatitis), one of the most common and bothersome side effects
- Acne-like rash and skin changes
- Gastrointestinal symptoms: diarrhea, nausea, and abdominal pain
- Impaired wound healing
Serious Adverse Effects
- Immunosuppression: Increased risk of opportunistic infections (pneumonia, UTIs, viral reactivation including CMV and BK virus) and potentially certain malignancies (especially skin cancers and lymphoma)
- Pneumonitis: Interstitial lung disease / sirolimus-associated pneumonitis; potentially life-threatening; presents as cough, dyspnea, and pulmonary infiltrates
- Nephrotoxicity: Can exacerbate renal injury, especially when combined with calcineurin inhibitors
- Hepatotoxicity: Elevated liver enzymes; rare cases of hepatic necrosis
- Impaired fertility: Oligospermia and testicular toxicity in males; menstrual irregularities in females; generally reversible upon discontinuation
- Metabolic syndrome: Insulin resistance, new-onset diabetes, hyperglycemia
- Lymphedema and angioedema: Reported particularly in renal transplant patients
Contraindications
- Known hypersensitivity to sirolimus, everolimus, or other mTOR inhibitors
- Active, untreated infections (immunosuppression will worsen outcome)
- Severe hepatic impairment (significantly altered drug metabolism)
- Pregnancy (FDA Category C; teratogenic in animals) and lactation
- Live vaccines during treatment (impaired immune response)
- Lung transplant recipients (increased risk of bronchial anastomotic dehiscence)
Drug Interactions
- CYP3A4 and P-glycoprotein inhibitors (ketoconazole, itraconazole, erythromycin, clarithromycin, grapefruit juice): Markedly increase sirolimus levels; dose reduction or avoidance required
- CYP3A4 inducers (rifampin, phenytoin, carbamazepine, St. John's Wort): Markedly decrease sirolimus levels; may lead to subtherapeutic exposure
- Calcineurin inhibitors (cyclosporine, tacrolimus): Synergistic nephrotoxicity; careful therapeutic drug monitoring required
- Statins and fibrates: Increased risk of rhabdomyolysis when combined with sirolimus-induced hyperlipidemia treatment
- ACE inhibitors: Increased risk of angioedema when used concurrently with sirolimus
- Vaccines: Live vaccines contraindicated; inactivated vaccines may have reduced immunogenicity
Population-Specific Considerations
- Elderly: Increased susceptibility to infections and impaired wound healing; monitor blood counts, lipids, and renal function frequently
- Pediatric: Used in transplant settings; growth impairment possible with chronic use; careful therapeutic drug monitoring required
- Renal transplant patients: Standard of care but requires close monitoring; target trough levels typically 4–12 ng/mL
- Longevity/anti-aging use: Intermittent low-dose protocols (e.g., 1–6 mg weekly) are being studied; long-term safety in healthy individuals is not established; immunosuppressive risks persist even at low doses
- Fertility considerations: Counsel patients of reproductive age; reversible gonadal toxicity documented in both sexes
- Diabetic patients: Monitor HbA1c and fasting glucose; may worsen glycemic control
Pharmacokinetic Profile
Rapamycin — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- Typical transplant doses are much higher (1-5mg daily) than what's being explored for longevity
Molecular Structure
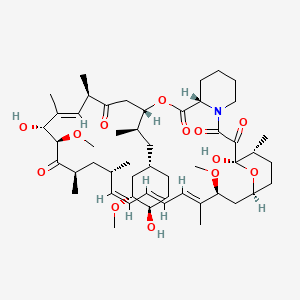
- Formula
- C51H79NO13
- Weight
- 914.2 Da
- PubChem CID
- 5284616
- Exact Mass
- 913.5551 Da
- LogP
- 6
- TPSA
- 195 Ų
- H-Bond Donors
- 3
- H-Bond Acceptors
- 13
- Rotatable Bonds
- 6
- Complexity
- 1760
Identifiers (SMILES, InChI)
InChI=1S/C51H79NO13/c1-30-16-12-11-13-17-31(2)42(61-8)28-38-21-19-36(7)51(60,65-38)48(57)49(58)52-23-15-14-18-39(52)50(59)64-43(33(4)26-37-20-22-40(53)44(27-37)62-9)29-41(54)32(3)25-35(6)46(56)47(63-10)45(55)34(5)24-30/h11-13,16-17,25,30,32-34,36-40,42-44,46-47,53,56,60H,14-15,18-24,26-29H2,1-10H3/b13-11+,16-12+,31-17+,35-25+/t30-,32-,33-,34-,36-,37+,38+,39+,40-,42+,43+,44-,46-,47+,51-/m1/s1
QFJCIRLUMZQUOT-HPLJOQBZSA-NSafety Profile
Common Side Effects
- Immune Suppression:: Primary concern is suppression of immune function, which can increase infection risk especially with long-term use
- Glucose Metabolism:: Can cause glucose intolerance or insulin resistance in some users, though effects may be benign and similar to fasting
- Epigenetic Concerns:: Recent study showed potential acceleration of aging across 16 epigenetic clocks, leading prominent users like Bryan Johnson to discontinue
- Mouth Sores:: Commonly reported side effect includes canker sores and oral ulcers with regular use
References (5)
- [2]Fasting and rapamycin: diabetes versus benevolent glucose intolerance
→ Discusses how rapamycin can induce a form of glucose intolerance similar to fasting that may be benevolent rather than harmful, while also improving insulin sensitivity and reducing diabetic complications.
- [3]Rapamycin as a potential repurpose drug candidate for the treatment of COVID-19
→ Explores rapamycin's potential therapeutic benefits for COVID-19 through immune modulation, mitochondrial support, and anti-inflammatory effects.
- [4]Pharmaceutical Intervention of Aging
→ Reviews rapamycin alongside other compounds like metformin and resveratrol as promising pharmacological interventions targeting aging pathways and age-related diseases.
- [5]Regulatory effects of hemp seed/evening primrose oil supplement in comparison with rapamycin on mTORC2 and IL-10 genes in experimental autoimmune encephalomyelitis
→ Compares rapamycin's effects with dietary supplements on immune regulation and gene expression in an autoimmune disease model.
- [1]Rapamycin, the only drug that consistently demonstrated to increase mammalian longevity. An update
→ Comprehensive review confirming rapamycin is the only drug that has consistently demonstrated lifespan extension across multiple mammalian species in rigorous testing protocols.
RANTES / CCL5-Derived Peptides
RANTES (Regulated upon Activation, Normal T cell Expressed and Secreted), systematically renamed CCL5 (C-C motif chemokine ligand 5), is a 68-amino acid chemoki
Rapastinel
Rapastinel (formerly GLYX-13) is an NMDA receptor modulator with glycine-site partial agonist properties that acts as a positive allosteric modulator. It has be