Folinic Acid (Leucovorin)
Folinic acid is a biologically active form of folate that bypasses the need for dihydrofolate reductase activation, used clinically to counteract methotrexate toxicity and support methylation and nucleotide synthesis pathways.
Overview
Folinic acid, also known as leucovorin or 5-formyltetrahydrofolate, is a reduced and metabolically active form of folic acid that does not require enzymatic reduction by dihydrofolate reductase (DHFR) to enter the folate cycle. This property makes it clinically invaluable as a "rescue" agent following high-dose methotrexate chemotherapy, where it restores folate-dependent biosynthetic pathways in healthy cells while preserving the drug's anti-tumor effects.
Beyond its established role in oncology, folinic acid supports critical one-carbon metabolism reactions including DNA synthesis, amino acid interconversion, and methylation processes. Unlike folic acid, it can be utilized directly by cells regardless of polymorphisms in the MTHFR gene, making it a preferred supplemental form for individuals with impaired folate metabolism. It has been studied in the context of neural tube defect prevention, neuropsychiatric conditions, and cerebral folate deficiency syndromes.
Research in autism spectrum disorder has generated particular interest, with some clinical trials suggesting that folinic acid supplementation may improve verbal communication in children with identified folate receptor autoantibodies. It is also used adjunctively with 5-fluorouracil in colorectal cancer protocols, where it enhances the cytotoxic effect of the chemotherapy agent by stabilizing its binding to thymidylate synthase.
Mechanism of Action
Reduced Folate Bypass Mechanism
Folinic acid (5-formyltetrahydrofolate, leucovorin) is a metabolically active reduced form of folic acid that bypasses the enzymatic step catalyzed by dihydrofolate reductase (DHFR). Unlike folic acid, it does not require DHFR-mediated reduction to enter the folate cycle, making it critical as a "rescue" agent after methotrexate therapy, which inhibits DHFR (PMID: 27599753).
One-Carbon Transfer Reactions
Once administered, folinic acid is rapidly converted to 5,10-methylenetetrahydrofolate by serine hydroxymethyltransferase and subsequently to 5-methyltetrahydrofolate (5-MTHF) by methylenetetrahydrofolate reductase (MTHFR). These folate coenzymes serve as one-carbon donors for critical biosynthetic reactions including thymidylate synthesis (via thymidylate synthase, essential for DNA replication), purine biosynthesis (supplying C2 and C8 of the purine ring), and methionine regeneration from homocysteine via methionine synthase (PMID: 22419553).
Synergy with 5-Fluorouracil
In oncology, folinic acid potentiates the cytotoxicity of 5-fluorouracil (5-FU) by stabilizing the ternary complex of 5-FdUMP, thymidylate synthase, and 5,10-methylenetetrahydrofolate. This enhanced inhibition of thymidylate synthase depletes intracellular dTMP pools, causing thymineless death in rapidly dividing cancer cells (PMID: 8898171).
Methylation & Neurotransmitter Support
By replenishing tetrahydrofolate pools, folinic acid supports the methionine-SAMe cycle, facilitating S-adenosylmethionine (SAMe)-dependent methylation of DNA, histones, neurotransmitters, and phospholipids. This is particularly relevant in individuals with MTHFR polymorphisms or cerebral folate deficiency, where folinic acid can cross the blood-brain barrier via folate receptor alpha (PMID: 19924226).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Depression Augmentation:: Most effective when combined with SSRIs rather than as monotherapy, with response rates approximately double that of placebo. Dosage Dependent:: 15 mg/day showed significantly better outcomes than 7.5 mg/day in clinical trials for depression. Individual Variation:: Effectiveness varies considerably between individuals, with approximately one-third showing significant response. MTHFR Mutations:: Particularly beneficial for individuals with genetic polymorphisms affecting folate metabolism
- Most effective when combined with SSRIs rather than as monotherapy, with response rates approximately double that of placebo
- 15 mg/day showed significantly better outcomes than 7.5 mg/day in clinical trials for depression
- Effectiveness varies considerably between individuals, with approximately one-third showing significant response
- Particularly beneficial for individuals with genetic polymorphisms affecting folate metabolism
Safety Profile
Safety Profile: Folinic Acid (Leucovorin)
Common Side Effects
- Gastrointestinal: nausea, vomiting, diarrhea, and stomatitis (especially during 5-FU rescue protocols)
- Skin rash, urticaria, or pruritus in approximately 2–5% of patients
- Insomnia and agitation reported with high-dose IV administration
- Fever and malaise, particularly in chemotherapy combination regimens
- Injection site reactions (pain, erythema, induration) with IM or IV administration
Serious Adverse Effects
- Seizures: reported in patients receiving high-dose IV leucovorin, particularly those with pre-existing CNS conditions or receiving intrathecal methotrexate
- Severe hypersensitivity: anaphylaxis, angioedema, and Stevens-Johnson syndrome reported rarely
- Enhanced 5-fluorouracil toxicity: when used as a chemotherapy potentiator, folinic acid significantly increases GI toxicity (severe diarrhea, mucositis) and myelosuppression
- Masking of B12 deficiency: may correct megaloblastic anemia while allowing irreversible neurological damage from untreated B12 deficiency to progress
- Thrombocytopenia and leukopenia when combined with antifolate chemotherapy
- Rare reports of encephalopathy at very high parenteral doses
Contraindications
- Pernicious anemia or undiagnosed B12 deficiency: leucovorin may mask hematologic signs while neurological damage progresses
- Known hypersensitivity to folinic acid or any formulation component
- Intrathecal administration is contraindicated (neurotoxic by this route)
- Should not be used to counteract the effects of methotrexate in the treatment of meningeal leukemia
Drug Interactions
- 5-Fluorouracil (5-FU): intentionally combined in oncology, but dramatically increases 5-FU toxicity (GI, hematologic); requires careful dosing and monitoring
- Methotrexate: used as rescue agent; timing is critical — delayed leucovorin administration reduces efficacy of rescue
- Trimethoprim-sulfamethoxazole: folinic acid may reduce antimicrobial efficacy by providing folate to bacteria; folic acid (not folinic acid) is preferred for folate supplementation during TMP-SMX therapy
- Phenobarbital, phenytoin, primidone: folinic acid may reduce anticonvulsant plasma levels, increasing seizure risk
- Capecitabine: similar interaction as 5-FU; enhanced toxicity profile
- Glucarpidase (Voraxaze): do not administer leucovorin within 2 hours of glucarpidase
Population-Specific Considerations
- Pregnant women: leucovorin is FDA Category C; used when benefit outweighs risk, primarily in methotrexate rescue during ectopic pregnancy treatment
- Breastfeeding: folate is secreted in breast milk; high-dose leucovorin should be used with caution; discuss with oncologist if on chemotherapy
- Pediatric patients: dosing for methotrexate rescue is weight-based; monitor renal function and methotrexate levels closely
- Elderly: increased susceptibility to enhanced 5-FU toxicity; dose reductions often required
- Renal impairment: delayed methotrexate clearance may require extended leucovorin rescue; monitor serum methotrexate levels until below 0.05 μM
- Hepatic impairment: metabolism may be altered; monitor for toxicity signs with combination chemotherapy
Pharmacokinetic Profile
Molecular Structure
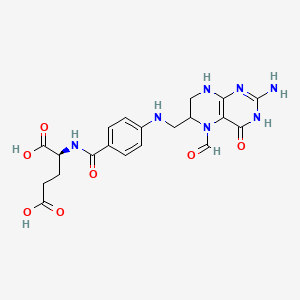
- Formula
- C20H23N7O7
- Weight
- 473.4 Da
- PubChem CID
- 135403648
- Exact Mass
- 473.1659 Da
- LogP
- -1.2
- TPSA
- 216 Ų
- H-Bond Donors
- 7
- H-Bond Acceptors
- 10
- Rotatable Bonds
- 9
- Complexity
- 911
Identifiers (SMILES, InChI)
InChI=1S/C20H23N7O7/c21-20-25-16-15(18(32)26-20)27(9-28)12(8-23-16)7-22-11-3-1-10(2-4-11)17(31)24-13(19(33)34)5-6-14(29)30/h1-4,9,12-13,22H,5-8H2,(H,24,31)(H,29,30)(H,33,34)(H4,21,23,25,26,32)/t12?,13-/m0/s1
VVIAGPKUTFNRDU-ABLWVSNPSA-NSafety Profile
Common Side Effects
- Generally Well-Tolerated:: Clinical trials reported adverse event rates comparable to placebo
- Minimal Side Effects:: No significant safety concerns identified in randomized controlled trials at therapeutic doses
- Individual Reactions:: Some individuals may experience mild gastrointestinal effects or headaches
- Drug Interactions:: May interact with anticonvulsants and other medications affecting folate metabolism
References (4)
- [3]Folate supplementation as a beneficial add-on treatment in relieving depressive symptoms: A meta-analysis of meta-analyses
→ This comprehensive meta-analysis of meta-analyses confirmed that folate supplementation serves as a beneficial adjunctive treatment for relieving depressive symptoms across multiple studies.
- [4]Biological, dietetic and pharmacological properties of vitamin B9
→ This review comprehensively discusses folate's biochemical properties, natural dietary sources, effects of food processing, industrial production methods, fortification strategies, and physiological roles in human health including prevention of pathological states related to deficiency.
- [2]l-Methylfolate as Adjunctive Therapy for SSRI-Resistant Major Depression: Results of Two Randomized, Double-Blind, Parallel-Sequential Trials
→ Two trials demonstrated that l-methylfolate at 15 mg/day as an adjunct to SSRI treatment produced a 32% response rate in SSRI-resistant depression compared to 15% with placebo, with a number needed to treat of approximately six and good tolerability.
- [1]Folate Augmentation of Treatment--Evaluation for Depression (FolATED): randomised trial and economic evaluation
→ This randomized controlled trial examined folate supplementation as an augmentation strategy for depression treatment, evaluating both clinical effectiveness and cost-effectiveness in patients with depressive symptoms.
FMN (Flavin Mononucleotide)
Flavin mononucleotide (FMN), also known as riboflavin-5′-phosphate, is the bioactive form of Vitamin B2 that serves as a vital prosthetic group for mitochondria
Follicle-Stimulating Hormone (FSH)
Follicle-stimulating hormone (FSH) is a heterodimeric glycoprotein hormone produced by gonadotroph cells of the anterior pituitary that plays an indispensable r