Carvacrol
Carvacrol is a monoterpenoid phenol found predominantly in oregano and thyme essential oils. It possesses broad-spectrum antimicrobial activity along with anti-inflammatory and antioxidant properties.
Overview
Carvacrol (5-isopropyl-2-methylphenol) is a monoterpenoid phenol and one of the primary bioactive constituents of oregano (Origanum vulgare) and thyme (Thymus vulgaris) essential oils. It is an isomer of thymol and is recognized for its distinctive warm, pungent aroma. Carvacrol has been used historically in traditional medicine and is currently approved as a food flavoring agent by the FDA (GRAS status).
The antimicrobial properties of carvacrol have been extensively documented against a wide range of bacteria, fungi, and parasites. Its mechanism of action involves disruption of the bacterial cell membrane through interaction with membrane lipids, leading to increased permeability, leakage of cellular contents, and ultimately cell death. Carvacrol has demonstrated efficacy against methicillin-resistant Staphylococcus aureus (MRSA), Escherichia coli, Candida species, and various foodborne pathogens, generating interest in its application as a natural food preservative and alternative to conventional antibiotics.
In addition to its antimicrobial activity, carvacrol modulates inflammatory pathways by suppressing NF-kB signaling and reducing the production of pro-inflammatory cytokines such as TNF-alpha and IL-6. Preclinical studies have investigated its potential in gastrointestinal disorders, respiratory infections, and cancer, with several studies reporting anti-proliferative effects in tumor cell lines. Carvacrol is available commercially in oregano oil supplements and as a purified compound for research applications.
Mechanism of Action
Monoterpenoid Phenol — TRP Channel Modulation
Carvacrol (5-isopropyl-2-methylphenol) is a monoterpenoid phenol found predominantly in oregano (Origanum vulgare) and thyme (Thymus vulgaris) essential oils. It activates TRPV3 (transient receptor potential vanilloid 3) channels at low micromolar concentrations, inducing warm-sensing and vasodilatory responses. Carvacrol also activates TRPA1 channels (the "wasabi receptor"), contributing to its pungent sensory properties. At higher concentrations, it inhibits TRPM7 channels, which are involved in magnesium homeostasis and cell proliferation, potentially contributing to its anti-proliferative effects (PMID: 22226908).
Antimicrobial Mechanism — Membrane Disruption
Carvacrol exerts broad-spectrum antimicrobial activity by inserting into the phospholipid bilayer of bacterial cell membranes, disrupting lipid packing and increasing membrane permeability. This causes dissipation of the proton motive force (PMF), leakage of intracellular ATP, K+, and phosphate, and collapse of the transmembrane electrochemical gradient. In Gram-negative bacteria, carvacrol first disintegrates the outer membrane by chelating divalent cations (Ca2+, Mg2+) that stabilize lipopolysaccharide (LPS), then penetrates to the inner membrane. It also inhibits bacterial efflux pumps (AcrAB-TolC), enhancing susceptibility to conventional antibiotics (PMID: 22178164).
Anti-Inflammatory — NF-kB & COX-2 Suppression
Carvacrol suppresses NF-kB activation by inhibiting IKKbeta phosphorylation and IkBalpha degradation in macrophages and epithelial cells. This reduces transcription of pro-inflammatory mediators including COX-2, iNOS, TNF-alpha, IL-1beta, and IL-6. Carvacrol also activates PPARgamma in macrophages, promoting the anti-inflammatory M2 phenotype and increasing IL-10 production (PMID: 22664488).
GABAergic & Anxiolytic Properties
Carvacrol acts as a positive allosteric modulator of GABA_A receptors, enhancing chloride ion conductance and producing anxiolytic and sedative effects in animal models. It also inhibits acetylcholinesterase (AChE) activity, potentially enhancing cholinergic neurotransmission and cognitive function (PMID: 22579816).
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Reported Effects
Antimicrobial Efficacy:: Strong evidence for effectiveness against various bacterial and fungal strains in research settings. Anti-inflammatory Action:: Well-documented reduction of inflammatory markers and cytokines in experimental models. Safety Profile:: Generally considered safe with limited toxicity data in humans. Synergistic Potential:: Enhanced effectiveness when combined with other natural compounds
- Strong evidence for effectiveness against various bacterial and fungal strains in research settings
- Well-documented reduction of inflammatory markers and cytokines in experimental models
- Generally considered safe with limited toxicity data in humans
- Enhanced effectiveness when combined with other natural compounds
Safety Profile
Safety Profile: Carvacrol
Common Side Effects
- Gastrointestinal irritation (burning sensation, heartburn, nausea) in ~10-15% of users
- Oral/esophageal mucosal irritation when taken undiluted
- Skin irritation or contact dermatitis with topical application (essential oil)
- Mild diarrhea
- Unpleasant strong taste/odor
Serious Adverse Effects
- Severe mucosal burns: Undiluted oregano oil (high carvacrol content) can cause chemical burns to oral mucosa, esophagus, and stomach
- Hepatotoxicity: High-dose chronic use has shown hepatotoxic potential in animal studies
- Allergic reactions: Anaphylaxis possible in Lamiaceae-allergic individuals
- Hematologic effects: In vitro antiplatelet activity; excessive bleeding risk at high doses
- Embryotoxicity: Demonstrated teratogenic effects in animal models at high doses
Contraindications
- Known allergy to oregano, thyme, or Lamiaceae family plants
- Active peptic ulcer disease or severe gastritis (mucosal irritant)
- Pregnancy (potential uterotonic and embryotoxic effects)
- Bleeding disorders or pre-surgical patients
- Severe hepatic impairment
- Iron-deficiency anemia (may reduce iron absorption)
Drug Interactions
- Anticoagulants/Antiplatelets (warfarin, heparin, aspirin): Additive antiplatelet effects; increased bleeding risk
- Antidiabetic medications (metformin, insulin): Carvacrol has demonstrated hypoglycemic effects in animal studies; monitor blood glucose
- Antihypertensives: Additive hypotensive effects; carvacrol shows vasodilatory properties
- CYP450 substrates: May inhibit CYP3A4 and CYP2D6 in vitro; potential to increase plasma levels of co-administered drugs
- Iron/Mineral supplements: Phenolic compounds may chelate metals; separate by 2+ hours
- Antibiotics: Potential synergistic antimicrobial effects (may be beneficial but monitor)
Population-Specific Considerations
- Pregnancy: Contraindicated; animal data shows embryotoxic and uterotonic properties. Do not use supplemental doses
- Lactation: Insufficient safety data; avoid concentrated supplements. Small culinary amounts likely safe
- Pediatric: Not recommended for children under 6; essential oil ingestion poses particular risk in young children
- Elderly: Start with low doses; increased GI sensitivity and potential drug interactions with polypharmacy
- Topical use: Always dilute in carrier oil (max 2-3% concentration); patch test before widespread application
- Renal impairment: Limited data; use caution with chronic high-dose supplementation
Pharmacokinetic Profile
Molecular Structure
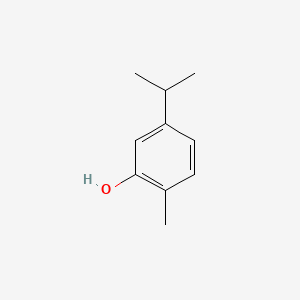
- Formula
- C10H14O
- Weight
- 150.22 Da
- PubChem CID
- 10364
- Exact Mass
- 150.1045 Da
- LogP
- 3.1
- TPSA
- 20.2 Ų
- H-Bond Donors
- 1
- H-Bond Acceptors
- 1
- Rotatable Bonds
- 1
- Complexity
- 120
Identifiers (SMILES, InChI)
InChI=1S/C10H14O/c1-7(2)9-5-4-8(3)10(11)6-9/h4-7,11H,1-3H3
RECUKUPTGUEGMW-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Toxicity Data:: Limited information on adverse effects in humans at supplemental doses
- Metabolic Pathways:: Knowledge of human metabolism and degradation pathways is incomplete
- Phenolic Compound Concerns:: Potential for environmental contamination and degradation products requires consideration
- Safety Assessment:: More clinical data needed to establish comprehensive safety profile
References (4)
- [4]Carvacrol: From Ancient Flavoring to Neuromodulatory Agent
→ Beyond its traditional use as a flavoring agent, carvacrol shows neuromodulatory properties and potential therapeutic applications in neurological conditions.
- [3]Carvacrol—A Natural Phenolic Compound with Antimicrobial Properties
→ Research confirms carvacrol's potent antimicrobial effects against numerous bacterial and fungal strains, demonstrating efficacy both alone and in combination with other compounds for controlling pathogens.
- [1]Carvacrol and human health: A comprehensive review
→ Carvacrol demonstrates multiple bioactivities including antimicrobial, antioxidant, and anti-inflammatory properties that make it potentially useful for clinical applications across various health conditions.
- [2]Therapeutic application of carvacrol: A comprehensive review
→ Carvacrol exerts strong anti-inflammatory effects by inducing antioxidant enzymes (SOD, GPx, GR, CAT), preventing lipid peroxidation, and reducing pro-inflammatory cytokine levels while modulating LPS-induced immune responses.
Cartalax
Cartalax is a synthetic tripeptide (Ala-Glu-Asp) classified as a polyfunctional bioregulatory peptide with primary effects on fibroblasts across multiple tissue
Casein Protein
Casein is a slow-digesting protein derived from milk that provides a sustained release of amino acids over several hours. It is commonly used as a dietary suppl