Angiotensin II
Angiotensin II is a potent vasoactive octapeptide generated by angiotensin-converting enzyme (ACE) from angiotensin I. As the primary effector of the classical renin-angiotensin-aldosterone system (RAAS), it drives vasoconstriction, aldosterone secretion, sympathetic activation, and cellular growth through AT1 receptor signaling, while AT2 receptor activation mediates opposing vasodilatory and anti-proliferative effects.
Overview
Angiotensin II occupies a central position in cardiovascular physiology as the primary hormonal mediator of the RAAS cascade. The pathway begins with renin — an aspartyl protease released from the juxtaglomerular cells of the kidney in response to decreased renal perfusion pressure, sympathetic stimulation, or reduced sodium delivery to the macula densa. Renin cleaves the liver-derived substrate angiotensinogen to produce the decapeptide angiotensin I, which is then converted to the octapeptide angiotensin II by angiotensin-converting enzyme (ACE), a zinc metallopeptidase predominantly expressed on the pulmonary vascular endothelium.
While Ang II is the classical effector of the RAAS, the system is now understood as a dual-axis framework. The ACE/Ang II/AT1 axis drives vasoconstriction, sodium retention, inflammation, and fibrosis, while the counter-regulatory ACE2/Angiotensin-(1-7)/MAS axis promotes vasodilation, natriuresis, and tissue protection. The balance between these two arms determines cardiovascular and renal homeostasis.
The pharmacological importance of Ang II is underscored by the fact that ACE inhibitors and ARBs — drugs that either reduce Ang II production or block its AT1 receptor — are among the most prescribed medications worldwide for hypertension, heart failure, diabetic nephropathy, and post-myocardial infarction management. Paradoxically, synthetic Ang II itself has also been approved as a therapeutic agent (Giapreza) for refractory vasodilatory shock.
Mechanism of Action
Angiotensin II exerts its biological effects through two principal receptor subtypes with largely opposing actions:
AT1 Receptor (Angiotensin Type 1)
The AT1 receptor is a G protein-coupled receptor (GPCR) that mediates the classical cardiovascular and renal effects of Ang II:
- Vasoconstriction: AT1 activation stimulates phospholipase C (PLC), generating inositol trisphosphate (IP3) and diacylglycerol (DAG). IP3 triggers calcium release from intracellular stores, driving smooth muscle contraction and vasoconstriction. This is the primary mechanism of Ang II-mediated blood pressure elevation.
- Aldosterone secretion: Ang II acts on the adrenal zona glomerulosa to stimulate aldosterone synthesis and release, promoting renal sodium reabsorption and potassium excretion, expanding extracellular fluid volume.
- Sympathetic potentiation: AT1 activation enhances norepinephrine release from sympathetic nerve terminals and inhibits vagal tone, amplifying adrenergic cardiovascular effects.
- ADH release: Stimulates vasopressin (ADH) secretion from the posterior pituitary, further promoting water retention.
- Cellular proliferation and hypertrophy: AT1 signaling activates MAPK/ERK, JAK/STAT, and NF-kB pathways, driving cardiomyocyte hypertrophy, vascular smooth muscle cell proliferation, and fibroblast activation.
- Pro-inflammatory effects: NF-kB activation increases expression of adhesion molecules (VCAM-1, ICAM-1), chemokines (MCP-1), and pro-inflammatory cytokines (TNF-alpha, IL-6, IL-1beta), promoting vascular inflammation and atherosclerosis.
- Pro-fibrotic effects: AT1 signaling stimulates TGF-beta1 expression and collagen synthesis, driving cardiac, renal, and vascular fibrosis.
- Oxidative stress: NADPH oxidase activation generates superoxide radicals, contributing to endothelial dysfunction, lipid oxidation, and NO scavenging.
AT2 Receptor (Angiotensin Type 2)
The AT2 receptor opposes many AT1-mediated effects and is predominantly expressed during fetal development, with lower but functionally significant adult expression:
- Vasodilation: AT2 activation stimulates bradykinin and nitric oxide (NO) release, producing endothelium-dependent vasodilation.
- Anti-proliferative effects: Promotes apoptosis of vascular smooth muscle cells and inhibits neointimal formation after vascular injury.
- Anti-fibrotic signaling: Opposes TGF-beta1/Smad signaling, reducing extracellular matrix deposition.
- Neuronal regeneration: AT2 activation promotes neurite outgrowth and nerve regeneration, an area of active investigation.
Metabolic Fates
Ang II is rapidly degraded by several peptidases into bioactive metabolites:
- ACE2 cleaves the C-terminal phenylalanine to produce Angiotensin-(1-7), the primary effector of the counter-regulatory MAS receptor axis.
- Aminopeptidase A removes the N-terminal aspartate to generate Angiotensin III (Ang 2-8), which retains AT1/AT2 activity with preferential central and adrenal effects.
- Aminopeptidase N further degrades Ang III to Angiotensin IV (Ang 3-8), which activates the AT4 receptor (IRAP) to enhance cognition and renal blood flow.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Blood Pressure Regulation and Hypertension
Ang II is the most potent endogenous vasoconstrictor identified in the cardiovascular system, approximately 40 times more potent than norepinephrine on a molar basis. The RAAS cascade was first elucidated by Tigerstedt and Bergman (1898) with the discovery of renin, and the isolation of Ang II by Skeggs et al. (1956) established the biochemical basis for what would become one of the most therapeutically targeted pathways in medicine. Chronic Ang II infusion in animal models produces sustained hypertension, cardiac hypertrophy, renal injury, and vascular remodeling — recapitulating the organ damage seen in human hypertensive disease [1].
"Ang II is not merely a vasoconstrictor hormone; it is a growth factor, a pro-inflammatory cytokine, and a pro-fibrotic mediator whose chronic activation drives target organ damage independent of its hemodynamic effects." — Ruiz-Ortega M et al. (2001) [2]
Cardiac Remodeling and Heart Failure
Chronic Ang II exposure drives pathological cardiac remodeling through AT1-mediated cardiomyocyte hypertrophy, fibroblast proliferation, and interstitial fibrosis. In post-myocardial infarction models, Ang II promotes adverse left ventricular remodeling, chamber dilation, and progressive heart failure. The landmark clinical trials of ACE inhibitors (CONSENSUS, SOLVD) and ARBs (Val-HeFT, CHARM) demonstrated that blocking Ang II signaling reduces morbidity and mortality in heart failure, validating decades of basic research on Ang II-mediated cardiac pathology [3].
"Blockade of the renin-angiotensin system remains a cornerstone of heart failure therapy, reflecting the central role of angiotensin II in disease progression." — Pfeffer MA et al. (1992) [4]
Renal Physiology and Nephropathy
In the kidney, Ang II preferentially constricts the efferent arteriole, maintaining glomerular filtration pressure during states of reduced renal perfusion. While this is an appropriate short-term compensatory response, chronic efferent constriction produces glomerular hypertension, podocyte injury, proteinuria, and progressive glomerulosclerosis. Ang II also directly stimulates mesangial cell proliferation, proximal tubular sodium reabsorption, and tubulointerstitial fibrosis. The RENAAL and IDNT trials demonstrated that ARBs slow the progression of diabetic nephropathy, confirming the pathogenic role of Ang II in renal disease [5].
Vascular Inflammation and Atherosclerosis
Ang II is a potent pro-inflammatory mediator in the vasculature. Through AT1-mediated NF-kB activation, it upregulates adhesion molecules, chemokines, and cytokines that recruit monocytes and macrophages to the vessel wall, promoting atherosclerotic plaque formation. Ang II also stimulates NADPH oxidase-derived reactive oxygen species (ROS), which oxidize LDL, impair endothelial NO bioavailability, and accelerate endothelial dysfunction. ApoE-knockout mice infused with Ang II develop accelerated atherosclerosis and abdominal aortic aneurysms [6].
Giapreza: Synthetic Angiotensin II for Refractory Hypotension
In a notable therapeutic paradox, synthetic Ang II (Giapreza/LJPC-501) was approved by the FDA in December 2017 for the treatment of refractory vasodilatory shock — a condition in which patients remain hypotensive despite high-dose catecholamine vasopressors and fluid resuscitation. The pivotal ATHOS-3 trial demonstrated that Ang II infusion achieved a mean arterial pressure (MAP) response in 69.9% of patients versus 23.4% with placebo, with reduced catecholamine requirements [7].
"Angiotensin II is an effective vasopressor in patients with vasodilatory shock who are not responding to high doses of conventional vasopressors." — Khanna A et al. (2017) [7]
The pharmacologic rationale for Giapreza rests on the observation that critically ill patients with vasodilatory shock often have impaired endogenous RAAS activation, depleted Ang II levels, and catecholamine resistance. Exogenous Ang II provides an alternative vasopressor mechanism through AT1 receptor-mediated vasoconstriction independent of adrenergic receptors.
COVID-19 and ACE2 Research
The COVID-19 pandemic brought Ang II biology to global attention when SARS-CoV-2 was identified as using ACE2 as its cellular entry receptor. Viral binding and internalization of ACE2 reduces the conversion of Ang II to protective Angiotensin-(1-7), resulting in unopposed Ang II accumulation. Elevated plasma Ang II levels in critically ill COVID-19 patients correlated with viral load, lung injury severity, and mortality [8].
"Plasma angiotensin II levels in COVID-19 patients were markedly elevated and linearly associated with viral load and degree of lung injury." — Liu Y et al. (2020) [8]
This mechanistic insight prompted therapeutic strategies including recombinant soluble ACE2 (to restore Ang II degradation and act as a viral decoy), exogenous Angiotensin-(1-7) (TXA127), and ARBs to block the deleterious effects of excess Ang II. The role of RAAS dysregulation in COVID-19 pathophysiology has fundamentally advanced understanding of the Ang II/ACE2 axis beyond traditional cardiovascular medicine [9].
Organ Fibrosis
Ang II is a master driver of fibrosis across multiple organs. Through AT1-mediated TGF-beta1 upregulation, Ang II activates Smad2/3 signaling, promotes myofibroblast differentiation, and stimulates collagen and fibronectin deposition. This fibrotic mechanism operates in cardiac fibrosis (contributing to diastolic dysfunction and arrhythmia), renal fibrosis (driving CKD progression), hepatic fibrosis (in non-alcoholic steatohepatitis), and pulmonary fibrosis. ARBs and ACE inhibitors have demonstrated anti-fibrotic effects in clinical trials across these organ systems, confirming Ang II as a central mediator of pathological tissue remodeling [10].
Central Nervous System Effects
Ang II acts within the brain to regulate thirst, salt appetite, sympathetic outflow, and vasopressin release. Circumventricular organs — regions lacking a complete blood-brain barrier — express AT1 receptors that sense circulating Ang II. Additionally, a local brain RAS produces Ang II independently of the systemic circulation. Central Ang II signaling through the subfornical organ and paraventricular nucleus drives sympathetic activation, contributing to neurogenic hypertension. Angiotensin III may be the primary effector of the brain RAS in some pathways [11].
Safety Profile
Endogenous Ang II is a normal physiological hormone, and its effects are dose-dependent and context-dependent. Exogenous Ang II (Giapreza) has been studied in clinical trials and ICU settings with the following safety observations:
- Thromboembolic events: The ATHOS-3 trial reported higher rates of arterial and venous thromboembolism in the Ang II group (12.9%) versus placebo (5.1%). This is consistent with Ang II's known prothrombotic effects including PAI-1 upregulation and tissue factor expression.
- Peripheral ischemia: Digital and distal extremity ischemia can occur with Ang II infusion, reflecting its potent vasoconstrictive properties.
- Hypertension: Excessive Ang II infusion produces severe hypertension; careful dose titration and monitoring are required.
- Tachyphylaxis: Prolonged Ang II exposure can produce AT1 receptor downregulation, though this is not typically clinically significant during acute ICU use.
- End-organ effects: Chronic Ang II exposure (as in untreated hypertension) produces cardiac hypertrophy, renal damage, vascular remodeling, and fibrosis — the rationale for ACE inhibitor and ARB therapy.
Clinical Research Protocols
- Giapreza (ICU vasopressor): Initial infusion at 20 ng/kg/min, titrated in increments of up to 15 ng/kg/min every 5 minutes to a maximum of 80 ng/kg/min to achieve target MAP ≥ 75 mmHg.
- Pharmacology research: Doses of 1-10 ng/kg/min IV infusion used in research settings for hemodynamic studies.
- Ang II challenge test: Historically used to assess adrenal responsiveness; 5-25 ng/kg/min IV for 20-60 minutes.
- Duration: Typically 24-72 hours in ICU settings; prolonged use warrants monitoring for thromboembolic complications.
Subpopulation Research
- Septic shock patients: ATHOS-3 demonstrated efficacy in catecholamine-resistant vasodilatory shock. Post-hoc analyses suggest particular benefit in patients with low endogenous Ang II levels and high Ang I/Ang II ratios (indicating impaired ACE activity) [7].
- COVID-19 patients: Elevated Ang II levels correlate with disease severity due to ACE2 depletion by SARS-CoV-2 [8].
- Heart failure patients: Chronic RAAS activation with elevated Ang II drives disease progression; ACE inhibitors and ARBs are first-line therapies [3, 4].
- Diabetic nephropathy: Ang II-mediated efferent arteriolar constriction and mesangial proliferation contribute to progressive renal injury; ARBs are renoprotective [5].
- African American populations: Higher prevalence of low-renin hypertension may affect response to RAAS-targeting therapies, though Ang II sensitivity is preserved.
Pharmacokinetic Profile
Angiotensin II — Pharmacokinetic Curve
Intravenous infusion (Giapreza)Ongoing & Future Research
- Biomarker-guided Giapreza use: The Ang I/Ang II ratio (renin activity marker) is being investigated to identify patients most likely to benefit from exogenous Ang II in shock.
- AT2 receptor agonists: Compound 21 (C21) and other selective AT2 agonists are in development to exploit the protective arm of Ang II signaling without AT1-mediated harm.
- Biased AT1 ligands: Beta-arrestin-biased AT1 agonists (TRV027) that activate cardioprotective signaling while avoiding harmful Gq-mediated pathways have entered clinical trials.
- Ang II in organ transplantation: Machine perfusion with Ang II is being explored to maintain vascular tone and reduce ischemia-reperfusion injury in donor organs.
- RAAS profiling in precision medicine: Comprehensive RAAS peptide profiling (Ang I, Ang II, Ang-(1-7), Ang III, Ang IV) as a tool for personalized cardiovascular therapy selection.
Quick Start
- Route
- Intravenous infusion (Giapreza)
Molecular Structure
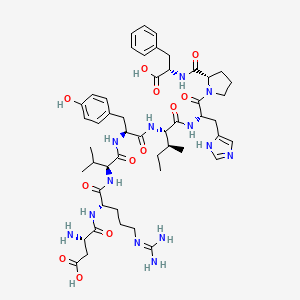
- Formula
- C50H71N13O12
- Weight
- 1046.18 Da
- CAS
- 4474-91-3
- PubChem CID
- 172198
- Exact Mass
- 1045.5345 Da
- LogP
- -1.7
- TPSA
- 409 Ų
- H-Bond Donors
- 13
- H-Bond Acceptors
- 15
- Rotatable Bonds
- 29
- Complexity
- 1980
Identifiers (SMILES, InChI)
InChI=1S/C50H71N13O12/c1-5-28(4)41(47(72)59-36(23-31-25-54-26-56-31)48(73)63-20-10-14-38(63)45(70)60-37(49(74)75)22-29-11-7-6-8-12-29)62-44(69)35(21-30-15-17-32(64)18-16-30)58-46(71)40(27(2)3)61-43(68)34(13-9-19-55-50(52)53)57-42(67)33(51)24-39(65)66/h6-8,11-12,15-18,25-28,33-38,40-41,64H,5,9-10,13-14,19-24,51H2,1-4H3,(H,54,56)(H,57,67)(H,58,71)(H,59,72)(H,60,70)(H,61,68)(H,62,69)(H,65,66)(H,74,75)(H4,52,53,55)/t28-,33-,34-,35-,36-,37-,38-,40-,41-/m0/s1
CZGUSIXMZVURDU-JZXHSEFVSA-NResearch Indications
Vasodilatory Shock (FDA-Approved)
FDA-approved (2017) as Giapreza for increasing blood pressure in adults with septic or distributive shock. ATHOS-3 trial showed significant MAP increase vs. placebo. Acts via AT1 receptor on vascular smooth muscle causing vasoconstriction.
Used as rescue vasopressor when patients fail to achieve adequate MAP with first-line vasopressors and fluid resuscitation. Provides a distinct mechanism of action independent of catecholamine pathways.
Hemodynamic Support
Particularly effective in patients with elevated plasma renin concentration, suggesting benefit in states of endogenous renin-angiotensin system activation. Half-life under 1 minute allows precise titration.
Emerging evidence for use in vasoplegia following cardiopulmonary bypass, where catecholamine-refractory vasodilation is common. Clinical experience supports its role as adjunctive vasopressor.
Research Protocols
intravenous Injection
Administered via intravenous injection.
What to Expect
What to Expect
Giapreza (ICU vasopressor): Initial infusion at 20 ng/kg/min, titrated in increments of up to 15 ng/kg/min every 5 minutes to a maximum of 80...
Duration: Typically 24-72 hours in ICU settings; prolonged use warrants monitoring for thromboembolic complications.
Ang II challenge test: Historically used to assess adrenal responsiveness; 5-25 ng/kg/min IV for 20-60 minutes.
Continued use as directed
Quality Indicators
What to look for
- Naturally occurring compound
Frequently Asked Questions
References (14)
- [9]Verdecchia P et al. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur J Intern Med (2020)
- [14]Povlsen AL et al. Counter-regulatory renin-angiotensin system in cardiovascular disease. Nat Rev Cardiol (2023)
- [5]Brenner BM et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med (2001)
- [6]Daugherty A et al. Angiotensin II promotes atherosclerotic lesions and aneurysms in apolipoprotein E-deficient mice. J Clin Invest (2000)
- [7]
- [8]Liu Y et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci (2019)
- [10]Mezzano SA et al. Renin-angiotensin system activation and interstitial inflammation in human diabetic nephropathy. Kidney Int Suppl (2003)
- [11]
- [13]Patel VB et al. Role of the ACE2/Angiotensin 1-7 axis of the renin-angiotensin system in heart failure. Circ Res (2016)
- [12]
- [4]Pfeffer MA et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med (1992)
- [1]Crowley SD et al. Angiotensin II causes hypertension and cardiac hypertrophy through its receptors in the kidney. Proc Natl Acad Sci U S A (2006)
- [2]Ruiz-Ortega M et al. Molecular mechanisms of angiotensin II-induced vascular injury. Curr Hypertens Rep (2003)
- [3]The SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med (1991)
Angiotensin-(1-7)
Angiotensin-(1-7) is a seven-amino acid endogenous peptide (Asp-Arg-Val-Tyr-Ile-His-Pro) generated primarily through the enzymatic cleavage of angiotensin II by
Angiotensin III
Angiotensin III (Ang III, also designated Ang 2-8 or des-Asp1-angiotensin II) is a naturally occurring heptapeptide (Arg-Val-Tyr-Ile-His-Pro-Phe) generated by t