RANTES / CCL5-Derived Peptides
RANTES (Regulated upon Activation, Normal T cell Expressed and Secreted), officially designated CCL5, is a 68-amino acid CC chemokine that signals through CCR1, CCR3, and CCR5 receptors to orchestrate immune cell trafficking. CCL5/CCR5 signaling plays dual roles in the tumor microenvironment — promoting anti-tumor immunity through T cell recruitment while also facilitating tumor-promoting inflammation and metastasis. CCR5 is a major HIV-1 co-receptor, and CCL5-derived antagonists including Met-RANTES have informed the development of CCR5 inhibitors such as maraviroc. Cancer immunotherapy applications targeting the CCL5/CCR5 axis are under active investigation.
Overview
CCL5 belongs to the CC chemokine subfamily, characterized by two adjacent cysteine residues near the N-terminus. It is produced by a wide array of cell types including T lymphocytes (particularly CD8+ memory T cells), macrophages, platelets, synovial fibroblasts, tubular epithelial cells, and certain tumor cells. CCL5 signals through three seven-transmembrane G-protein coupled receptors: CCR1 (monocytes, neutrophils), CCR3 (eosinophils, basophils, Th2 cells), and CCR5 (monocytes, macrophages, Th1 cells, activated T cells, dendritic cells, NK cells). Additionally, CCL5 binds glycosaminoglycans (GAGs) on endothelial surfaces, creating immobilized chemokine gradients essential for leukocyte arrest and transmigration under flow conditions.
The structure of CCL5 includes a flexible N-terminal region (residues 1-9) critical for receptor activation, followed by an N-loop that contacts the receptor extracellular domains, and a three-stranded antiparallel beta-sheet with a C-terminal alpha-helix. At low concentrations (<100 nM), CCL5 exists as dimers; at higher concentrations, it forms tetramers and higher-order oligomers that bind GAGs with increased affinity. This oligomerization is functionally important: obligate monomer CCL5 variants (E66S mutant) retain receptor binding but show impaired in vivo chemotactic activity, demonstrating that oligomer-GAG interactions are essential for physiological function.
Met-RANTES, created by adding a methionine residue to the N-terminus, was the first chemokine-derived receptor antagonist. This modification converts CCL5 from a CCR5 agonist to a potent antagonist, blocking HIV-1 entry and leukocyte recruitment. Met-RANTES served as proof-of-concept for targeting CCR5, directly informing the development of small-molecule CCR5 antagonists including maraviroc (the first FDA-approved CCR5 inhibitor in 2007) and the broader concept of chemokine receptor blockade in disease.
Mechanism of Action
CCL5 and its derivatives activate multiple signaling pathways through CCR receptors:
-
Gi/o-coupled chemotaxis signaling: CCR5 activation by CCL5 couples to pertussis toxin-sensitive Gi proteins, activating PLC-beta (generating IP3/calcium flux and DAG/PKC), PI3K-gamma (generating PIP3, activating Akt and Rac GTPases), and releasing Gbetagamma subunits that drive cytoskeletal reorganization. The resulting actin polymerization, pseudopod extension, and integrin activation produce directional cell migration along the chemokine gradient. Oppermann, M. (2004) — Cell. Signal.
-
HIV-1 co-receptor function of CCR5: R5-tropic HIV-1 gp120, after binding CD4, undergoes conformational change exposing the V3 loop that engages CCR5's N-terminus and extracellular loops. This triggers gp41-mediated membrane fusion. CCL5/RANTES blocks HIV entry through two mechanisms: direct competitive inhibition of gp120-CCR5 binding, and receptor internalization reducing surface CCR5 density. The CCR5-delta32 mutation (32bp deletion producing a truncated, non-functional receptor) confers near-complete resistance to R5-tropic HIV-1 in homozygotes and is found at ~10% allele frequency in northern European populations. Cocchi, F. et al. (1995) — Science
-
Met-RANTES antagonism: The N-terminal methionine addition blocks the conformational change required for CCR5 activation. Met-RANTES binds CCR5 with affinity similar to native CCL5 but fails to trigger Gi coupling, calcium mobilization, or chemotaxis. Met-RANTES potently inhibits HIV-1 infection of macrophages and PBMCs at nanomolar concentrations. Proudfoot, A. E. et al. (1996) — J. Biol. Chem.
-
Tumor-promoting CCL5/CCR5 signaling: In the tumor microenvironment, CCL5/CCR5 signaling on tumor cells activates mTOR, NF-kappaB, and STAT3, promoting proliferation, survival, and epithelial-mesenchymal transition (EMT). CCL5 produced by mesenchymal stem cells (MSCs) recruited to tumors acts in a paracrine fashion to enhance breast cancer metastasis. In pancreatic cancer, tumor-derived CCL5 recruits immunosuppressive Tregs and myeloid-derived suppressor cells (MDSCs) to the tumor microenvironment. Karnoub, A. E. et al. (2007) — Nature
-
Anti-tumor CCL5 signaling: Paradoxically, CCL5 is also critical for anti-tumor immunity. Tumor-produced CCL5 recruits CD8+ cytotoxic T lymphocytes, NK cells, and dendritic cells to the tumor microenvironment, enabling immune recognition and killing. CCL5 expression correlates with improved survival in colorectal cancer, melanoma, and ovarian cancer when associated with Th1-polarized immune infiltrates. The Batf3-dependent conventional dendritic cell 1 (cDC1) population is recruited by CCL5 and XCL1, and its presence is essential for checkpoint immunotherapy responsiveness. Spranger, S. et al. (2017) — Cell
-
Platelet-derived CCL5: Platelets are a major source of CCL5, released from alpha-granules upon activation. Platelet-derived CCL5 deposited on inflamed endothelium promotes monocyte arrest and transmigration. CCL5 heterodimer formation with CXCL4 (platelet factor 4) creates a synergistic signal for monocyte recruitment in atherosclerosis. von Hundelshausen, P. et al. (2005) — J. Exp. Med.
Research
Met-RANTES and Chemokine-Derived Antagonists
Met-RANTES, synthesized by Proudfoot et al. in 1996, demonstrated that minimal N-terminal modification of chemokines could convert agonists into receptor antagonists — a principle that revolutionized chemokine biology. Met-RANTES blocked M-tropic HIV-1 infection at IC50 values of approximately 5-50 nM, comparable to native RANTES but without inducing the pro-inflammatory signaling. Proudfoot, A. E. et al. (1996) — J. Biol. Chem. Subsequent engineering produced AOP-RANTES (aminooxypentane-RANTES), which was 50-100 fold more potent than Met-RANTES as an HIV inhibitor and acted primarily through profound CCR5 internalization and intracellular sequestration. PSC-RANTES, a further-optimized analog, achieved picomolar HIV-inhibitory potency and was explored as a topical microbicide candidate. These chemokine-derived antagonists proved the concept that eventually led to small-molecule CCR5 antagonists (maraviroc, vicriviroc, cenicriviroc).
Maraviroc Repurposing in Oncology
Maraviroc, originally developed for HIV, is being investigated for cancer applications based on the pro-tumorigenic CCR5 signaling axis. The MARACON trial (phase 1) evaluated maraviroc in metastatic colorectal cancer patients with CCR5-positive liver metastases. Maraviroc treatment repolarized tumor-associated macrophages from M2 (immunosuppressive) to M1 (anti-tumor) phenotype and was associated with objective anti-tumor activity in some patients. Halama, N. et al. (2016) — Cancer Discov. This trial was notable as one of the first demonstrations that CCR5 blockade could modify the immune microenvironment of established human tumors. Ongoing studies are evaluating maraviroc in combination with checkpoint inhibitors (anti-PD-1) and with chemotherapy in breast, colorectal, and hepatocellular cancers.
CCL5 in Graft-Versus-Host Disease and Transplantation
CCL5/CCR5 signaling contributes to allograft rejection and graft-versus-host disease (GVHD) by recruiting alloreactive T cells and inflammatory monocytes to transplanted tissues. CCR5-delta32 recipient genotype is associated with improved renal allograft survival. In GVHD, CCL5 levels correlate with disease severity, and CCR5 blockade with maraviroc has been investigated for GVHD prophylaxis. A phase 2 trial by Reshef et al. demonstrated that maraviroc added to standard GVHD prophylaxis (tacrolimus + methotrexate) reduced the incidence of visceral acute GVHD after reduced-intensity allogeneic stem cell transplantation. Reshef, R. et al. (2012) — N. Engl. J. Med.
CCL5 in Atherosclerosis
CCL5 plays a central role in atherogenesis through recruitment of monocytes and T cells to arterial plaques. Platelet-deposited CCL5 on activated endothelium promotes monocyte rolling, arrest, and transmigration. The CCL5-CXCL4 heterodimer is particularly potent at promoting atherosclerotic monocyte recruitment, and disruption of this heterodimer with cyclic peptide inhibitors (MKEY) reduced atherosclerosis in ApoE-knockout mice. Koenen, R. R. et al. (2009) — Nat. Med. CCR5-delta32 carriers have a modestly reduced risk of cardiovascular events in some epidemiological studies, supporting CCR5 as a therapeutic target in cardiovascular inflammation.
CCR5 as HIV-1 Co-receptor and CCL5 as Suppressive Factor
The 1995 discovery by Cocchi et al. that CCL5 (RANTES), MIP-1alpha (CCL3), and MIP-1beta (CCL4) are the major HIV-suppressive factors produced by CD8+ T cells was transformative for HIV biology. These beta-chemokines suppressed R5-tropic HIV-1 replication by competing with viral gp120 for CCR5 binding and by inducing CCR5 internalization. This discovery, combined with the identification of CCR5 as the HIV co-receptor and the protective CCR5-delta32 polymorphism, validated CCR5 as a drug target. Cocchi, F. et al. (1995) — Science The clinical translation culminated in maraviroc (Selzentry/Celsentri), a small-molecule CCR5 allosteric antagonist approved in 2007 for treatment-experienced patients with R5-tropic HIV-1. Maraviroc binds within the CCR5 transmembrane helical bundle, inducing a conformational change that prevents gp120 engagement without directly competing at the chemokine binding site.
CCL5/CCR5 in the Tumor Microenvironment — Dual Roles
The role of CCL5/CCR5 in cancer is context-dependent and represents one of the most nuanced signaling axes in tumor immunology:
Pro-tumorigenic effects: In breast cancer, CCR5 is expressed on tumor cells, and CCL5 produced by tumor-infiltrating macrophages and bone marrow-derived MSCs promotes invasion, metastasis, and chemoresistance. Velasco-Velazquez et al. demonstrated that maraviroc reduced breast cancer cell invasion in vitro and metastasis in mouse models, suggesting CCR5 antagonism as a therapeutic strategy. Velasco-Velazquez, M. et al. (2012) — J. Natl. Cancer Inst. In pancreatic cancer, CCL5 recruits immunosuppressive Tregs that express CCR5, shifting the immune balance toward tolerance. In hepatocellular carcinoma, CCR5 signaling promotes tumor cell proliferation through mTOR activation.
Anti-tumorigenic effects: CCL5 is a critical component of the T cell-inflamed tumor microenvironment gene signature that predicts response to immune checkpoint inhibitors (anti-PD-1, anti-CTLA-4). Tumors lacking CCL5/CXCL9/CXCL10 expression exhibit "cold" immune microenvironments with poor T cell infiltration and checkpoint inhibitor resistance. The cDC1 dendritic cell population, essential for cross-priming anti-tumor CD8+ T cell responses, is recruited by tumor-produced CCL5 and XCL1. Loss of these chemokines through tumor-intrinsic WNT/beta-catenin pathway activation correlates with immune exclusion. Spranger, S. et al. (2017) — Cell
Safety Profile
Native CCL5 is an endogenous chemokine and is not administered therapeutically. Safety considerations apply to CCL5-derived antagonists and CCR5-targeting agents:
Maraviroc (CCR5 antagonist): Generally well tolerated in HIV clinical trials and post-marketing surveillance. Common adverse effects include cough, upper respiratory infections, musculoskeletal symptoms, abdominal pain, and dizziness. Hepatotoxicity (elevated transaminases) has been reported rarely; an allergic component (rash, eosinophilia, elevated IgE) may precede hepatic injury. A theoretical concern with CCR5 blockade is increased susceptibility to infections (particularly West Nile virus, given CCR5's role in neuroinflammation) and impaired anti-tumor immunity. However, long-term follow-up of HIV patients on maraviroc and epidemiological studies of CCR5-delta32 carriers have not demonstrated significant immunodeficiency beyond HIV susceptibility changes. QT prolongation has not been observed at therapeutic doses.
Met-RANTES and chemokine-derived antagonists: Met-RANTES was primarily studied in vitro and in animal models. As a modified chemokine protein, immunogenicity (anti-drug antibodies) is a potential concern for chronic administration. Local injection site reactions may occur. Systemic CCR5 blockade risks are similar to maraviroc.
CCR5 blockade in oncology: The dual role of CCL5/CCR5 in the tumor microenvironment creates a theoretical risk that CCR5 antagonism could impair anti-tumor T cell trafficking in some contexts, particularly in "hot" tumors where CCL5 is mediating beneficial immune infiltration. Patient selection based on tumor immune phenotyping (CCR5 expression on tumor cells vs. immune cells) is likely necessary to avoid this paradox.
CCR5-delta32 homozygosity considerations: While protective against R5-tropic HIV, CCR5-delta32 homozygotes show increased susceptibility to West Nile virus encephalitis, suggesting that chronic CCR5 blockade may carry similar risk in endemic areas.
Clinical Research Protocols
- Maraviroc for HIV (approved): 300 mg orally twice daily (with or without food). Dose adjustment with CYP3A4 inhibitors (150 mg BID with protease inhibitors) or inducers (600 mg BID with efavirenz). Tropism testing (Trofile or genotypic) mandatory before initiation to confirm R5-tropic virus.
- Maraviroc for colorectal cancer (MARACON protocol): 300 mg orally twice daily for 4 weeks pre-operatively in patients with CCR5-positive liver metastases. Pre- and post-treatment biopsies for immune phenotyping (macrophage polarization M1/M2, T cell infiltration).
- Maraviroc for GVHD prophylaxis: 300 mg orally twice daily starting day -2 through day +30 after allogeneic stem cell transplantation, combined with standard tacrolimus + methotrexate prophylaxis.
- CCL5/CCR5 biomarker assessment in tumors: Multiplex immunohistochemistry for CCR5, CCL5, CD8, CD4, FoxP3 (Tregs), CD68/CD163 (macrophage polarization). RNA expression: CCL5 in tumor immune gene signature panels (e.g., T cell-inflamed GEP score used for pembrolizumab response prediction).
- Met-RANTES (research only): Preclinical dosing: 10-100 μg subcutaneous or intraperitoneal in mouse models. Not currently in clinical development due to availability of small-molecule CCR5 antagonists.
Pharmacokinetic Profile
RANTES / CCL5-Derived Peptides — Pharmacokinetic Curve
Not used therapeutically as native peptide; derivatives and CCR5 antagonists administered orally (maraviroc) or by injection (Met-RANTES, investigational)Quick Start
- Route
- Not used therapeutically as native peptide; derivatives and CCR5 antagonists administered orally (maraviroc) or by injection (Met-RANTES, investigational)
Molecular Structure
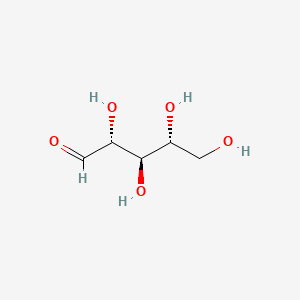
- Formula
- C5H10O5
- Weight
- 150.13 Da
- PubChem CID
- 5311110
- Exact Mass
- 150.0528 Da
- LogP
- -2.3
- TPSA
- 98 Ų
- H-Bond Donors
- 4
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 4
- Complexity
- 104
Identifiers (SMILES, InChI)
InChI=1S/C5H10O5/c6-1-3(8)5(10)4(9)2-7/h1,3-5,7-10H,2H2/t3-,4+,5-/m0/s1
PYMYPHUHKUWMLA-LMVFSUKVSA-NResearch Protocols
subcutaneous Injection
- Met-RANTES (research only): Preclinical dosing: 10-100 μg subcutaneous or intraperitoneal in mouse models.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Met-RANTES (research only) | 10-100 μg | Per protocol | —(Route: Subcutaneous Injection, Intraperitoneal) |
oral
Clinical Research Protocols - Maraviroc for HIV (approved): 300 mg orally twice daily (with or without food). - Maraviroc for colorectal cancer (MARACON protocol): 300 mg orally twice daily for 4 weeks pre-operatively in patients with CCR5-positive liver metastases.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Maraviroc for HIV (approved) | 300 mg, 150 mg, 600 mg | Twice daily | —(Route: Oral) |
| Maraviroc for colorectal cancer (MARACON protocol) | 300 mg | Twice daily | 4 weeks(Route: Oral) |
| Maraviroc for GVHD prophylaxis | 300 mg | Twice daily | —(Route: Oral) |
topical
PSC-RANTES, a further-optimized analog, achieved picomolar HIV-inhibitory potency and was explored as a topical microbicide candidate.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 150 mg, 600 mg | Per protocol | — |
intraperitoneal Injection
- Met-RANTES (research only): Preclinical dosing: 10-100 μg subcutaneous or intraperitoneal in mouse models.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Met-RANTES (research only) | 10-100 μg | Per protocol | —(Route: Subcutaneous Injection, Intraperitoneal) |
What to Expect
What to Expect
Effects begin within hours of administration based on half-life of ~3-4 hours (serum); extended by GAG binding and oligomerization
Maraviroc for colorectal cancer (MARACON protocol): 300 mg orally twice daily for 4 weeks pre-operatively in patients with CCR5-positive liver...
Due to short half-life (~3-4 hours (serum); extended by GAG binding and oligomerization), effects are expected per-dose; consistent daily...
Regular administration schedule required; effects are dose-dependent and do not persist between doses
Quality Indicators
What to look for
- Human clinical trials conducted
- Well-established safety profile
- Extensive peer-reviewed research base
Caution
- Injection site reactions reported
Red flags
- Potential carcinogenicity concerns
- Liver toxicity concerns reported
Frequently Asked Questions
References (6)
- [3]Spranger, S. et al — Tumor-Residing Batf3 Dendritic Cells Are Required for Effector T Cell Trafficking and Adoptive T Cell Therapy Cell (2017)
- [2]Halama, N. et al — Tumoral Immune Cell Exploitation in Colorectal Cancer Metastases Can Be Targeted Effectively by Anti-CCR5 Therapy in Cancer Patients Cancer Discov. (2016)
- [1]Cocchi, F. et al — Identification of RANTES, MIP-1alpha, and MIP-1beta as the major HIV-suppressive factors produced by CD8+ T cells Science (1995)
- [5]Aldinucci, D. & Casagrande, N — The CCL5/CCR5 axis in cancer progression: current knowledge and future perspectives Cancers (2023)
- [4]Suarez-Carmona, M. et al — CCR5 in cancer immunotherapy: the story so far and future perspectives J. Immunother. Cancer (2022)
- [6]Reshef, R. et al — Blockade of lymphocyte chemotaxis in visceral graft-versus-host disease N. Engl. J. Med. (2012)
R-Alpha Lipoic Acid
R-Alpha Lipoic Acid (R-ALA) is the naturally occurring, biologically active form of alpha-lipoic acid, a powerful antioxidant and mitochondrial cofactor. It fun
Rapamycin
Rapamycin (Sirolimus) is an mTOR (mechanistic target of rapamycin) inhibitor originally developed as an immunosuppressant for organ transplant patients. It work