EGF (Epidermal Growth Factor)
EGF is a 53-amino acid polypeptide that binds the EGF receptor to stimulate cell proliferation, differentiation, and survival, with applications in wound healing and anti-aging skincare.
Overview
EGF was the first growth factor to be isolated and characterized, identified from mouse submaxillary gland extracts by Stanley Cohen at Vanderbilt University. Human EGF is produced by platelets, macrophages, and monocytes and is found in saliva, urine, milk, and plasma. Endogenous EGF levels decline with age, contributing to slower wound healing and reduced epidermal turnover. Recombinant human EGF (rhEGF) has been developed for therapeutic use, including FDA-cleared wound care products for diabetic foot ulcers and burn injuries. In cosmetics, biosynthetic EGF is incorporated into serums and creams to promote skin renewal and reduce visible signs of aging.
Mechanism of Action
EGF binds to the extracellular domain of the EGF receptor (EGFR/ErbB1/HER1), a transmembrane receptor tyrosine kinase. Ligand binding induces receptor dimerization and autophosphorylation, activating three major downstream signaling cascades: the RAS-RAF-MEK-ERK (MAPK) pathway driving cell proliferation, the PI3K-AKT pathway promoting cell survival, and the JAK-STAT pathway regulating gene transcription. These pathways collectively stimulate keratinocyte and fibroblast proliferation, accelerate epithelial migration across wound beds, increase production of extracellular matrix components, and suppress apoptosis in damaged tissue Carpenter & Cohen (1990)45534-0). The three intramolecular disulfide bonds maintain the structural integrity required for receptor binding affinity.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
This calculator is provided for informational and educational purposes only. It is not intended as medical advice, diagnosis, or treatment guidance. Always consult a qualified healthcare professional before preparing or administering any substance. PepGuide assumes no liability for decisions made based on these calculations.
Research
Breast Milk EGF
Human breast milk contains EGF at concentrations of 30-100 ng/mL in mature milk and up to 200 ng/mL in colostrum — concentrations 500-2000-fold higher than plasma levels. This EGF is remarkably stable, resisting degradation by gastric acid and intestinal proteases in the neonatal gut. Playford et al. (2000) demonstrated that breast milk EGF stimulates intestinal epithelial proliferation and differentiation in neonatal animal models, contributing to gut maturation. Formula-fed infants lack this EGF source, which may partly explain the higher incidence of NEC, infectious diarrhea, and necrotizing enterocolitis in formula-fed versus breastfed premature infants. The addition of recombinant EGF to infant formula has been proposed but not yet implemented clinically.
Ulcer Healing
EGF accelerates the healing of gastric and duodenal ulcers through stimulation of epithelial cell proliferation, migration, and angiogenesis at ulcer margins. Konturek et al. (1988) showed that intragastric administration of EGF accelerates healing of cysteamine-induced duodenal ulcers in rats, with efficacy comparable to cimetidine. In human studies, recombinant hEGF administered topically (via endoscopic spray) accelerated gastric ulcer healing rates compared to antisecretory therapy alone. However, the development of proton pump inhibitors (PPIs), which achieve near-complete acid suppression and >90% ulcer healing rates, reduced clinical interest in EGF-based ulcer therapy.
Experimental Colitis
EGF administration (systemic or rectal) reduces mucosal injury and promotes healing in experimental colitis models. Sinha et al. (2003) demonstrated that recombinant hEGF enemas significantly reduced disease activity and histological injury scores in DSS-induced colitis, with evidence of enhanced epithelial proliferation and reduced apoptosis. In a pilot clinical study of left-sided ulcerative colitis, Sinha et al. (2003) reported that EGF enemas (5 micrograms) in addition to oral mesalamine achieved remission in 83% of patients versus 8% in the placebo group — a striking result that has not been followed by larger confirmatory trials.
Wound Healing
EGF is one of the most clinically validated growth factors for wound repair. Topical rhEGF accelerates healing of diabetic foot ulcers, chronic wounds, and burn injuries by promoting re-epithelialization and granulation tissue formation. Heberprot-P, a formulation of rhEGF for intralesional injection, has been used in over 300,000 diabetic foot ulcer patients in Cuba and other countries, significantly reducing amputation rates Berlanga-Acosta et al. (2013). Clinical trials have demonstrated 40-70% improvement in wound closure rates compared to standard care.
Skin Regeneration and Anti-Aging
Topical EGF stimulates epidermal cell turnover, increases skin thickness, and improves elasticity. Clinical studies using EGF-containing creams have shown measurable reduction in wrinkle depth and improvement in skin texture over 8-12 weeks of use. EGF promotes synthesis of hyaluronic acid and collagen in the dermis, counteracting age-related thinning. Post-procedure applications (after laser resurfacing or chemical peels) accelerate recovery and reduce downtime Shin et al. (2012).
Hair Growth
EGF signaling plays a role in hair follicle cycling. Research has shown that EGF can stimulate proliferation of outer root sheath keratinocytes and promote the anagen (growth) phase of the hair cycle. Topical formulations containing EGF have demonstrated increased hair density and thickness in clinical studies, though results vary and the mechanism is complex, involving crosstalk with Wnt/beta-catenin signaling pathways Jang (2005).
Corneal Healing
EGF accelerates corneal epithelial wound healing following injury, surgery, or chemical burns. Eye drops containing rhEGF have been studied for post-LASIK recovery, corneal abrasion repair, and dry eye syndrome. The corneal epithelium is particularly responsive to EGF due to high EGFR expression, and topical application promotes rapid re-epithelialization without stromal scarring Pastor & Calonge (1992)90004-d).
Salivary EGF and Upper GI Protection
The salivary glands are a major source of luminal EGF for the upper GI tract. Human saliva contains EGF at concentrations of 1-5 ng/mL, and continuous swallowing delivers approximately 10-30 micrograms of EGF to the esophageal and gastric mucosa daily. Sialoadenectomy (salivary gland removal) in rodents dramatically impairs gastric ulcer healing, an effect reversed by exogenous EGF administration. Konturek et al. (1991) demonstrated that salivary EGF accelerates gastric ulcer healing through stimulation of mucosal cell proliferation and angiogenesis. The clinical relevance is illustrated by the observation that xerostomia (dry mouth) from Sjogren syndrome or radiation therapy is associated with increased susceptibility to esophagitis and peptic ulceration.
Brunner's Gland Secretion
Brunner's glands, located in the duodenal submucosa, secrete an alkaline, mucin-rich fluid containing high concentrations of EGF. This Brunner's gland EGF provides local protection to the duodenal mucosa at the pyloric-duodenal junction — the region exposed to the highest acid load from gastric emptying. Poulsen et al. (1986) demonstrated that Brunner's gland secretion is stimulated by vagal activation, secretin, and luminal acid, creating a feed-forward protective mechanism. Brunner's gland hyperplasia is commonly observed in duodenal ulcer disease, interpreted as an adaptive increase in EGF production.
Necrotizing Enterocolitis (NEC)
NEC is the most common and devastating gastrointestinal emergency in premature neonates, characterized by acute intestinal inflammation, necrosis, and perforation. EGF deficiency is implicated in NEC pathogenesis, as premature infants have lower salivary EGF production, reduced EGFR expression in the intestinal mucosa, and lack the breast milk EGF that full-term breastfed infants receive. Dvorak et al. (2002) demonstrated that enteral EGF administration significantly reduces NEC incidence and severity in neonatal rat models by preserving mucosal barrier integrity, reducing bacterial translocation, and suppressing inflammatory cytokine production. Shin et al. (2000) showed that EGF prevents NEC-associated intestinal apoptosis through EGFR-mediated PI3K/Akt activation. A clinical pilot study by Sullivan et al. (2007) administering recombinant hEGF orally to extremely premature infants demonstrated safety and tolerability, with trends toward improved feeding tolerance, though the study was underpowered for NEC prevention endpoints.
Safety Profile
Topical EGF has a well-established safety profile in cosmetic and wound care applications. No significant systemic absorption occurs through intact skin, and local adverse effects are rare. However, EGF's role in stimulating cell proliferation has raised theoretical concerns regarding its use in individuals with a history of or predisposition to cancer. EGFR is overexpressed in many carcinomas, and chronic stimulation of this pathway could theoretically promote tumor growth. Regulatory bodies in several countries have debated the inclusion of EGF in cosmetics, and South Korea's MFDS has issued guidelines on its use. Current evidence does not demonstrate carcinogenic risk from topical cosmetic concentrations, but the debate has led some brands to avoid EGF in favor of alternative growth factors. Injectable formulations require clinical oversight due to the more direct mitogenic effects at higher local concentrations.
Post-Procedure Recovery
EGF-containing formulations are increasingly used following dermatological procedures including laser resurfacing, microneedling, chemical peels, and dermabrasion. Applied immediately after treatment, EGF accelerates re-epithelialization of the treated surface, reducing erythema duration and minimizing infection risk during the vulnerable healing window. Split-face studies have demonstrated faster recovery on EGF-treated sides compared to standard post-procedure care, with reduced crusting and earlier return to normal appearance Hou et al. (2020).
Cosmetic Controversy
EGF's mechanism of action -- promoting cell proliferation through EGFR activation -- has generated ongoing debate in the cosmetics industry. Since EGFR is overexpressed in many epithelial cancers and anti-EGFR therapies (cetuximab, erlotinib) are established cancer treatments, some researchers and regulatory bodies have questioned whether topical EGF application could theoretically promote or accelerate skin cancer development. Proponents counter that topical concentrations are orders of magnitude below those needed for systemic EGFR activation, that the stratum corneum limits penetration, and that decades of topical EGF use have not produced epidemiological signals of increased cancer risk. The European Scientific Committee on Consumer Safety (SCCS) has reviewed EGF and similar growth factors without issuing a ban, though several Asian regulatory bodies have imposed concentration limits.
Pharmacokinetic Profile
EGF (Epidermal Growth Factor) — Pharmacokinetic Curve
Topical, IntradermalQuick Start
- Route
- Topical, Intradermal
Molecular Structure
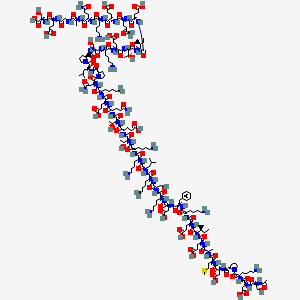
- Formula
- C257H381N73O83S7
- Weight
- 6 Da
- CAS
- 62253-63-8
- PubChem CID
- 16132341
- Exact Mass
- 4962.4930 Da
- LogP
- -43.5
- TPSA
- 2250 Ų
- H-Bond Donors
- 72
- H-Bond Acceptors
- 88
- Rotatable Bonds
- 180
- Complexity
- 12200
Identifiers (SMILES, InChI)
InChI=1S/C212H350N56O78S/c1-16-106(7)166(261-190(323)131(64-75-160(292)293)231-172(305)109(10)228-174(307)134(78-91-347-15)245-196(329)140(98-165(302)303)255-201(334)147-53-39-88-266(147)209(342)135(52-29-38-87-221)250-197(330)139(97-164(300)301)254-198(331)143(100-269)229-113(14)276)204(337)246-129(62-73-158(288)289)187(320)233-118(47-24-33-82-216)179(312)252-137(94-114-42-19-18-20-43-114)194(327)253-138(96-163(298)299)195(328)237-121(50-27-36-85-219)181(314)258-144(101-270)199(332)239-119(48-25-34-83-217)178(311)251-136(92-104(3)4)193(326)236-116(45-22-31-80-214)175(308)235-122(51-28-37-86-220)189(322)263-168(110(11)273)207(340)249-133(66-77-162(296)297)192(325)264-169(111(12)274)206(339)248-126(58-69-152(224)279)184(317)243-128(61-72-157(286)287)186(319)234-120(49-26-35-84-218)180(313)256-142(95-153(225)280)211(344)268-90-40-54-148(268)202(335)257-141(93-105(5)6)210(343)267-89-41-55-149(267)203(336)259-145(102-271)200(333)238-117(46-23-32-81-215)177(310)244-132(65-76-161(294)295)191(324)265-170(112(13)275)208(341)262-167(107(8)17-2)205(338)247-130(63-74-159(290)291)188(321)241-125(57-68-151(223)278)183(316)242-127(60-71-156(284)285)185(318)232-115(44-21-30-79-213)176(309)240-124(56-67-150(222)277)173(306)227-108(9)171(304)226-99-154(281)230-123(59-70-155(282)283)182(315)260-146(103-272)212(345)346/h18-20,42-43,104-112,115-149,166-170,269-275H,16-17,21-41,44-103,213-221H2,1-15H3,(H2,222,277)(H2,223,278)(H2,224,279)(H2,225,280)(H,226,304)(H,227,306)(H,228,307)(H,229,276)(H,230,281)(H,231,305)(H,232,318)(H,233,320)(H,234,319)(H,235,308)(H,236,326)(H,237,328)(H,238,333)(H,239,332)(H,240,309)(H,241,321)(H,242,316)(H,243,317)(H,244,310)(H,245,329)(H,246,337)(H,247,338)(H,248,339)(H,249,340)(H,250,330)(H,251,311)(H,252,312)(H,253,327)(H,254,331)(H,255,334)(H,256,313)(H,257,335)(H,258,314)(H,259,336)(H,260,315)(H,261,323)(H,262,341)(H,263,322)(H,264,325)(H,265,324)(H,282,283)(H,284,285)(H,286,287)(H,288,289)(H,290,291)(H,292,293)(H,294,295)(H,296,297)(H,298,299)(H,300,301)(H,302,303)(H,345,346)/t106-,107-,108-,109-,110+,111+,112+,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,140-,141-,142-,143-,144-,145-,146-,147-,148-,149-,166-,167-,168-,169-,170-/m0/s1
UGPMCIBIHRSCBV-XNBOLLIBSA-NResearch Indications
Gastrointestinal Healing
Purified EGF heals small intestinal ulceration by stimulating cell migration from ulcer margins and granulation tissue formation. Over 11 clinical reports support GI administration for mucosal damage.
IBD patients show significantly lower serum EGF levels correlated with mucosal damage. EGF supplementation promotes epithelial development, wound healing, and angiogenesis in affected tissues.
EGF protects intestinal barrier function by regulating tight junction components, enhancing mucin secretion, and decreasing pathogen colonization through EGFR signaling pathways.
Mucosal Protection
EGF and TGF-α stimulate mucosal cell restitution and proliferation, provide gastroprotection, and inhibit gastric acid secretion. EGF accumulates in lesion areas to locally stimulate healing.
HB-EGF acts as an effective mitogen, anti-apoptotic protein, and vasodilator in NEC models, showing significant protective effects in intestinal ischemia/reperfusion injury.
Research Protocols
topical
Topical rhEGF accelerates healing of diabetic foot ulcers, chronic wounds, and burn injuries by promoting re-epithelialization and granulation tissue formation. Skin Regeneration and Anti-Aging Topical EGF stimulates epidermal cell turnover, increases skin thickness, and improves elasticity.
intradermal Injection
Administered via intradermal injection.
oral
No systemic toxicity has been observed with oral or rectal EGF administration at therapeutic doses. The short half-life (~8 minutes) limits systemic exposure after oral administration, reducing the risk of extra-intestinal mitogenic effects.
subcutaneous Injection
Administered via subcutaneous injection.
Interactions
Peptide Interactions
Both EGF and BPC-157 promote gastrointestinal mucosal healing through complementary mechanisms. EGF activates EGFR-mediated epithelial proliferation while BPC-157 upregulates growth factor expression and angiogenesis. Combined use accelerates ulcer healing and mucosal regeneration in preclinical models (Sikiric et al., 2018; Playford & Wright, 1996).
PPIs reduce gastric acid secretion, creating a less hostile environment for EGF-mediated mucosal repair. EGF is acid-labile, so gastric acid suppression preserves its bioactivity in the GI lumen. No pharmacokinetic interaction; mechanistically complementary for peptic ulcer management (Konturek et al., 1991).
What to Expect
What to Expect
Effects begin within hours of administration based on half-life of ~1-2 hours (serum)
Clinical studies using EGF-containing creams have shown measurable reduction in wrinkle depth and improvement in skin texture over 8-12 weeks of use.
Due to short half-life (~1-2 hours (serum)), effects are expected per-dose; consistent daily administration maintains therapeutic levels
Clinical studies using EGF-containing creams have shown measurable reduction in wrinkle depth and improvement in skin texture over 8-12 weeks of use.
Regular administration schedule required; effects are dose-dependent and do not persist between doses
Quality Indicators
What to look for
- Well-established safety profile
- Multiple peer-reviewed studies available
Red flags
- Potential carcinogenicity concerns
Frequently Asked Questions
References (10)
- [8]
- [9]
- [10]
- [11]
- [13]
- [14]
- [15]Kim et al — EGF-loaded nanoparticle hydrogels for enhanced diabetic wound healing Int. J. Biol. Macromol. (2023)
- [16]Park et al — Efficacy of topical recombinant human EGF for aging skin: a double-blind randomized controlled trial J. Cosmet. Dermatol. (2022)
- [17]Wang et al — Advanced delivery systems for EGF in chronic wound management: a systematic review Adv. Drug Deliv. Rev. (2024)
- [18]Li et al — EGF-functionalized silk fibroin scaffolds for full-thickness skin regeneration Biomaterials (2023)
Ecnoglutide (XW003)
Ecnoglutide (XW003) is a long-acting glucagon-like peptide-1 (GLP-1) receptor agonist developed by Sciwind Biosciences in collaboration with Gan & Lee Pharmaceu
Elastin Tripeptide
**Elastin tripeptides** are short peptide fragments derived from or modeled on the repetitive sequences found in tropoelastin, the soluble precursor of elastin.